Get your patient on Uvadex - Methoxsalen injection, Solution (Methoxsalen)
Uvadex - Methoxsalen injection, Solution prescribing information
UVADEX ® (methoxsalen) Sterile Solution should be used only by physicians who have special competence in the diagnosis and treatment of cutaneous T-cell lymphoma and who have special training and experience in the THERAKOS ® CELLEX ® Photopheresis System. Please consult the CELLEX ® Operator's Manual before using this product.
INDICATIONS AND USAGE
UVADEX ® (methoxsalen) Sterile Solution is indicated for extracorporeal administration with the THERAKOS ® CELLEX ® Photopheresis System in the palliative treatment of the skin manifestations of Cutaneous T-Cell Lymphoma (CTCL) that is unresponsive to other forms of treatment.
DRUG DOSAGE AND ADMINISTRATION
Each UVADEX ® treatment involves collection of leukocytes, photoactivation, and reinfusion of photoactivated cells. UVADEX ® (methoxsalen) Sterile Solution is supplied in 10 mL vials containing 200 mcg of methoxsalen (concentration of 20 mcg/mL). The THERAKOS ® CELLEX ® Photopheresis System Operator's Manual should be consulted before using this product. UVADEX ® should not be diluted. The contents of the vial should be injected into the THERAKOS ® CELLEX ® Photopheresis System immediately after being drawn up into a syringe. Do not inject directly into patients. The UVADEX ® vial is for single use only. Any UVADEX ® that is not used during a procedure should be immediately discarded. UVADEX ® can adsorb onto PVC and plastics, therefore only THERAKOS ® CELLEX ® photopheresis procedural kits supplied for use with the instrument should be used to administer this medicinal product. Once UVADEX ® is drawn into a plastic syringe it should be immediately injected into the photoactivation bag. UVADEX ® exposed to a plastic syringe for more than one hour should be discarded.
During treatment with the THERAKOS ® CELLEX ® Photopheresis System, the dosage of UVADEX ® for each treatment will be calculated according to the treatment volume.
- The prescribed amount of UVADEX ® should be injected into the recirculation bag prior to the Photoactivation Phase using the formula:
| TREATMENT VOLUME × 0.017 = mL of UVADEX ® for each treatment |
| Example: Treatment volume of 240 mL × 0.017 = 4.1 mL of UVADEX ® |
Frequency/Schedule of Treatment
Normal Treatment Schedule
Treatment is given on two consecutive days every four weeks for a minimum of seven treatment cycles (six months).
Accelerated Treatment Schedule
If the assessment of the patient during the fourth treatment cycle (approximately three months) reveals an increased skin score from the baseline score, the frequency of treatment may be increased to two consecutive treatments every two weeks. If a 25% improvement in the skin score is attained after four consecutive weeks, the regular treatment schedule may resume. Patients who are maintained in the accelerated treatment schedule may receive a maximum of 20 cycles. There is no clinical evidence to show that treatment with UVADEX ® beyond six months or using a different schedule provides additional benefit. In study CTCL 3, 15 of the 17 responses were seen within six months of treatment and only two patients responded to treatment after six months.
CONTRAINDICATIONS
- UVADEX ® (methoxsalen) Sterile Solution is contraindicated in patients exhibiting idiosyncratic or hypersensitivity reactions to methoxsalen, other psoralen compounds or any of the excipients. Patients possessing a specific history of a light sensitive disease state should not initiate methoxsalen therapy. Diseases associated with photosensitivity include lupus erythematosus, porphyria cutanea tarda, erythropoietic protoporphyria, variegate porphyria, xeroderma pigmentosum and albinism.
- UVADEX ® Sterile Solution is contraindicated in patients with aphakia, because of the significantly increased risk of retinal damage due to the absence of lenses.
- Patients should not receive UVADEX ® if they have any contraindications to the photopheresis procedure.
ADVERSE REACTIONS
Side effects of photopheresis (UVADEX ® used with THERAKOS ® Photopheresis Systems) were primarily related to hypotension secondary to changes in extracorporeal volume (>1%). In study CTCL 3 (UVADEX ® ), six serious cardiovascular adverse experiences were reported in five patients (5/51, 10%). Five of these six events were not related to photopheresis and did not interfere with the scheduled photopheresis treatments. One patient (1/51, 2%) with ischemic heart disease had an arrhythmia after the first day of photopheresis that was resolved the next day. Six infections were also reported in five patients. Two of the six events were Hickman catheter infections in one patient, which did not interrupt the scheduled photopheresis. The other four infections were not related to photopheresis and did not interfere with scheduled treatments.
POSTMARKETING
Adverse reactions reported from postmarketing experience include nausea, dysgeusia, photosensitivity reaction, pyrexia and hypersensitivity reactions including anaphylaxis and rash.
Drug Interactions
See Warnings Section.
DESCRIPTION
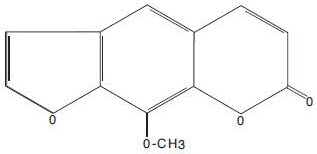
Methoxsalen is a naturally occurring photoactive substance found in the seeds of the Ammi majus (Umbelliferae) plant. It belongs to a group of compounds known as psoralens or furocoumarins. The chemical name of methoxsalen is 9-methoxy-7H-furo[3,2-g][1]-benzopyran-7-one; it has the following structure:
Each mL of UVADEX ® (methoxsalen, 8-methoxypsoralen) Sterile Solution contains methoxsalen 20 mcg, propylene glycol 50 mg, sodium chloride 8 mg, sodium acetate 1.75 mg, ethanol 40.550 mg, glacial acetic acid 1.260 mg, and Water for Injection q.s. to 1.0 mL. Glacial acetic acid and sodium hydroxide are used to adjust the pH of the solution if necessary. UVADEX ® is a clear, colorless to pale yellow liquid.
UVADEX ® is used in combination with the THERAKOS ® CELLEX ® Photopheresis System to extracorporeally treat leukocyte enriched buffy coat.
CLINICAL PHARMACOLOGY
Mechanism of action
The exact mechanism of action of methoxsalen is not known. The best-known biochemical reaction of methoxsalen is with DNA. Methoxsalen, upon photoactivation, conjugates and forms covalent bonds with DNA which leads to the formation of both monofunctional (addition to a single strand of DNA) and bifunctional adducts (crosslinking of psoralen to both strands of DNA). Reactions with proteins have also been described. The formation of photoadducts results in inhibition of DNA synthesis, cell division and epidermal turnover.
For the palliative treatment of Cutaneous T-Cell Lymphoma, Photopheresis consists of removing a portion of the patient's blood and separating the red blood cells from the white cell layer (buffy coat) by centrifugation. The red cells are returned to the patient and the UVADEX ® Sterile Solution is then injected into the instrument and mixed with the buffy coat. The instrument then irradiates this drug-cell mixture with ultraviolet light (UVA light, 320–400 nm) and returns the treated cells to the patient. See the appropriate Operator's Manual for details of this process. Although extracorporeal phototherapy exposes less than 10% of the total body burden of malignant cells to methoxsalen plus light, some patients achieve a complete response. Animal studies suggest that the photopheresis may activate an immune-mediated response against the malignant T-cells.
Use of the THERAKOS ® UVAR and UVAR XTS ® Photopheresis Systems after oral administration of methoxsalen were previously approved for the treatment of Cutaneous T-Cell Lymphoma. Interpatient variability in peak plasma concentration after an oral dose of methoxsalen ranges from 6 to 15 fold. UVADEX ® is injected directly into the separated buffy coat in the instrument in an attempt to diminish this interpatient variability and to improve the exposure of the cells to the drug.
Methoxsalen is reversibly bound to serum albumin and is also preferentially taken up by epidermal cells. Methoxsalen is rapidly metabolized in humans, with approximately 95% of the drug excreted as metabolites in the urine within 24 hours.
Systemic administration of methoxsalen followed by UVA exposure leads to cell injury. The most obvious manifestation of this injury after skin exposure is delayed erythema, which may not begin for several hours and peaks at 48–72 hours. The inflammation is followed over several days to weeks, by repair which is manifested by increased melanization of the epidermis and thickening of the stratum corneum.
The total dose of methoxsalen delivered in UVADEX ® is substantially lower (approximately 200 times) than that used with oral administration. More than 80% of blood samples collected 30 minutes after reinfusion of the photoactivated buffy coat had methoxsalen levels below detection limits of the assay (<10 ng/ml), and the mean plasma methoxsalen concentration was approximately 25 ng/ml.
CLINICAL STUDIES
Three single-arm studies were performed to evaluate the effectiveness of photopheresis in the treatment of the skin manifestations of Cutaneous T-Cell Lymphoma (CTCL). In the first study (CTCL 1), 39 patients were treated with the oral formulation of methoxsalen in conjunction with the UVAR Photopheresis System. The second study (CTCL 2) was a 5-year post approval follow-up of 57 CTCL patients that was conducted to evaluate long-term safety. This study also used the oral dosage formulation of methoxsalen. In the third study (CTCL 3), 51 patients were treated with the UVADEX ® formulation of methoxsalen in conjunction with the UVAR Photopheresis System. In study CTCL 3, 86% of the patients were Caucasian, the median age was 62 years, and the average number of prior therapies was 4.3.
In study CTCL 1, prednisone up to 10 mg/day was permitted in addition to topical steroids. In CTCL 2, there was no concomitant medication restriction. In CTCL 3, topical steroids were permitted only for the treatment of fissures on the soles of the feet and the palms of hands. All other steroids, topical or systemic, were prohibited.
In all three studies, patients were initially treated on two consecutive days every four to five weeks. If the patient did not respond after four cycles, treatment was accelerated to two consecutive days every other week. If the patient did not respond after four cycles at the accelerated schedule, the patient was treated on two consecutive days every week. If the patient still did not respond after four cycles of weekly treatments, the schedule was increased to three consecutive days every week for three cycles. In study CTCL 3, 15 of the 17 responses were seen within six months of treatment. Only two patients responded to treatment after six months. Clinical experience does not extend beyond this treatment frequency and there is no evidence to show that treatment with UVADEX ® beyond six months or using a different schedule provided additional benefit.
Overall skin scores were used in the clinical studies of photopheresis to assess the patient's response to treatment. The patient's baseline skin score was used for comparison with subsequent scores. A 25% reduction in skin score maintained for four consecutive weeks was considered a successful response to photopheresis therapy. Table 1 indicates the percent of successful responses within six months of beginning therapy for all patients who received at least one course of photopheresis. Only patients with patch plaque, extensive plaque and erythrodermic disease were enrolled in these studies. No patients with disease in the tumor phase were treated. There are no data available regarding the efficacy of UVADEX ® in patients with disease in the tumor phase.
| Study | Response % Within Six Months |
|---|---|
| CTCL 3 (UVADEX ® ) | 17/51 (33) |
| CTCL 2 (oral methoxsalen) | 16/57 (28) |
| CTCL 1 (oral methoxsalen) | 21/39 (54) |
Although the response rate with UVADEX ® in CTCL 3 was similar to with oral methoxsalen in CTCL 2, the possibility that UVADEX ® is inferior in efficacy to oral methoxsalen cannot be excluded due to the design and size of the clinical trials. The higher response rate with oral methoxsalen in CTCL 1 may be partly due to patients receiving more treatments (mean of 64 in CTCL 1, 31 in CTCL 2, and 20 in CTCL 3), and to the administration of systemic steroids in CTCL 1.
Retrospective analyses of three clinical benefit parameters from the Body Area Severity Scores in CTCL 3 suggested a correlation between skin score response and improvement in edema, scaling and resolution of fissures.
HOW SUPPLIED
UVADEX ® (methoxsalen) Sterile Solution 20 mcg/mL in 10 mL amber glass vials (NDC 64067-216-01), and cartons of 12 vials (NDC 64067-216-01). One vial of 10 mL contains 200 micrograms methoxsalen. The drug product must be stored between 59°F (15°C) and 86°F (30°C)
Mechanism of action
The exact mechanism of action of methoxsalen is not known. The best-known biochemical reaction of methoxsalen is with DNA. Methoxsalen, upon photoactivation, conjugates and forms covalent bonds with DNA which leads to the formation of both monofunctional (addition to a single strand of DNA) and bifunctional adducts (crosslinking of psoralen to both strands of DNA). Reactions with proteins have also been described. The formation of photoadducts results in inhibition of DNA synthesis, cell division and epidermal turnover.
For the palliative treatment of Cutaneous T-Cell Lymphoma, Photopheresis consists of removing a portion of the patient's blood and separating the red blood cells from the white cell layer (buffy coat) by centrifugation. The red cells are returned to the patient and the UVADEX ® Sterile Solution is then injected into the instrument and mixed with the buffy coat. The instrument then irradiates this drug-cell mixture with ultraviolet light (UVA light, 320–400 nm) and returns the treated cells to the patient. See the appropriate Operator's Manual for details of this process. Although extracorporeal phototherapy exposes less than 10% of the total body burden of malignant cells to methoxsalen plus light, some patients achieve a complete response. Animal studies suggest that the photopheresis may activate an immune-mediated response against the malignant T-cells.
Use of the THERAKOS ® UVAR and UVAR XTS ® Photopheresis Systems after oral administration of methoxsalen were previously approved for the treatment of Cutaneous T-Cell Lymphoma. Interpatient variability in peak plasma concentration after an oral dose of methoxsalen ranges from 6 to 15 fold. UVADEX ® is injected directly into the separated buffy coat in the instrument in an attempt to diminish this interpatient variability and to improve the exposure of the cells to the drug.
Methoxsalen is reversibly bound to serum albumin and is also preferentially taken up by epidermal cells. Methoxsalen is rapidly metabolized in humans, with approximately 95% of the drug excreted as metabolites in the urine within 24 hours.
Systemic administration of methoxsalen followed by UVA exposure leads to cell injury. The most obvious manifestation of this injury after skin exposure is delayed erythema, which may not begin for several hours and peaks at 48–72 hours. The inflammation is followed over several days to weeks, by repair which is manifested by increased melanization of the epidermis and thickening of the stratum corneum.
The total dose of methoxsalen delivered in UVADEX ® is substantially lower (approximately 200 times) than that used with oral administration. More than 80% of blood samples collected 30 minutes after reinfusion of the photoactivated buffy coat had methoxsalen levels below detection limits of the assay (<10 ng/ml), and the mean plasma methoxsalen concentration was approximately 25 ng/ml.
Uvadex - Methoxsalen injection, Solution PubMed™ news
- Journal Article • 2026 AprThe Human Gut Microbiome Metabolizes Diverse Bioactive Coumarins via α,β-Unsaturated Lactone Reduction.
- Journal Article • 2026 FebXanthotoxin-triggered HR51 suppression derepresses detoxification genes to drive metabolic adaptation in Spodoptera litura.
- Journal Article • 2026 JanPhytochemistry and Allelopathic Properties of Invasive Heracleum sosnowskyi Aqueous Extracts Against Lettuce (Lactuca sativa L.), Perennial Ryegrass (Lolium perenne L.), Timothy (Phleum pratense L.) and White Clover (Trifolium repens L.).
- Journal Article • 2025 DecMethoxsalen deteriorative effects on the testicular parenchyma and testosterone synthesis gene expression in male rats.
- Journal Article • 2025 DecInhibition of CYP2A6-mediated nicotine metabolism: A potential strategy for smoking cessation therapy.