Trifluridine - Trifluridine solution prescribing information
INDICATIONS AND USAGE: Trifluridine Ophthalmic Solution is indicated for the treatment of primary keratoconjunctivitis and recurrent epithelial keratitis due to herpes simplex virus, types 1 and 2.
DOSAGE AND ADMINISTRATION: Instill one drop of Trifluridine Ophthalmic Solution onto the cornea of the affected eye every 2 hours while awake for a maximum daily dosage of nine drops until the cornea ulcer has completely re-epithelialized. Following re-epithelialization, treatment for an additional 7 days of one drop every 4 hours while awake for a minimum daily dosage of five drops is recommended.
If there are no signs of improvement after 7 days of therapy or complete re-epithelialization has not occurred after 14 days of therapy, other forms of therapy should be considered. Continuous administration of trifluridine for periods exceeding 21 days should be avoided because of potential ocular toxicity.
CONTRAINDICATIONS: Trifluridine Ophthalmic Solution is contraindicated for patients who develop hypersensitivity reactions or chemical intolerance to trifluridine.
ADVERSE REACTIONS: The most frequent adverse reactions reported during controlled clinical trials were mild, transient burning or stinging upon instillation (4.6%) and palpebral edema (2.8%). Other adverse reactions in decreasing order of reported frequency were superficial punctate keratopathy, epithelial keratopathy, hypersensitivity reaction, stromal edema, irritation, keratitis sicca, hyperemia, and increased intraocular pressure.
DESCRIPTION: Trifluridine Ophthalmic Solution (also known as trifluorothymidine, F 3 TdR, F 3 T), an antiviral drug for topical treatment of epithelial keratitis caused by herpes simplex virus. The chemical name of trifluridine is α,α,α-trifluorothymidine. Trifluridine has the following structural formula.
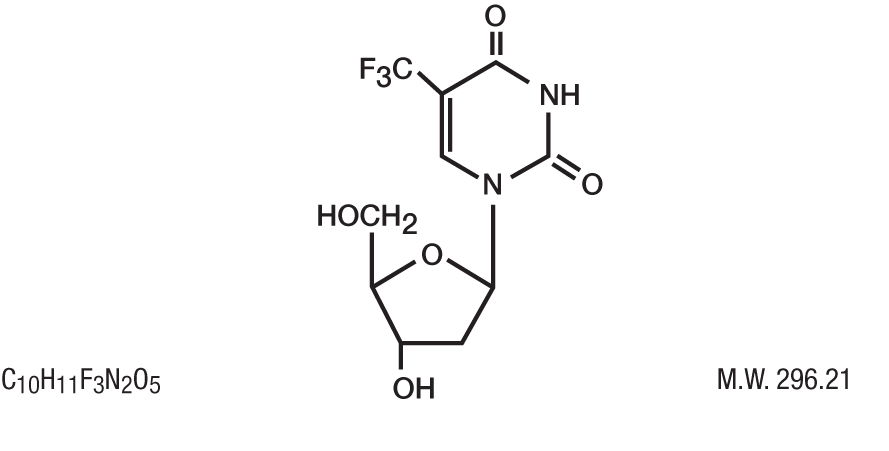
Trifluridine sterile ophthalmic solution contains 1% trifluridine in an aqueous solution with acetic acid and sodium acetate (buffers), sodium chloride, and thimerosal 0.001% (added as a preservative). The pH range is 5.5 to 6.0 and osmolality is approximately 283 mOsm.
CLINICAL PHARMACOLOGY: Trifluridine is a fluorinated pyrimidine nucleoside with in vitro and in vivo activity against herpes simplex virus, types 1 and 2 and vacciniavirus. Some strains of adenovirus are also inhibited in vitro .
Trifluridine is also effective in the treatment of epithelial keratitis that has not responded clinically to the topical administration of idoxuridine or when ocular toxicity or hypersensitivity to idoxuridine has occurred. In a smaller number of patients found to be resistant to topical vidarabine, trifluridine was also effective.
Trifluridine interferes with DNA synthesis in cultured mammalian cells. However, its antiviral mechanism of action is not completely known.
In vitro perfusion studies on excised rabbit corneas have shown that trifluridine penetrates the intact cornea as evidenced by recovery of parental drug and its major metabolite, 5-carboxy-2´-deoxyuridine, on the endothelial side of the cornea. Absence of the corneal epithelium enhances the penetration of trifluridine approximately two-fold.
Intraocular penetration of trifluridine occurs after topical instillation of trifluridine into human eyes. Decreased corneal integrity or stromal or uveal inflammation may enhance the penetration of trifluridine into the aqueous humor. Unlike the results of ocular penetration of trifluridine in vitro , 5-carboxy-2´-deoxyuridine was not found in detectable concentrations within the aqueous humor of the human eye.
Systemic absorption of trifluridine following therapeutic dosing with trifluridine appears to be negligible. No detectable concentrations of trifluridine or 5-carboxy-2´-deoxyuridine were found in the sera of adult healthy normal subjects who had trifluridine instilled into their eyes seven times daily for 14 consecutive days.
Clinical Studies: During a controlled multicenter clinical trial, 92 of 97 (95%) patients (78 of 81 with dendritic and 14 of 16 with geographic ulcers) responded to therapy with trifluridine as evidenced by complete corneal re-epithelialization within the 14-day therapy period. Fifty-six of 75 (75%) patients (49 of 58 with dendritic and 7 of 17 with geographic ulcers) responded to idoxuridine therapy. The mean time to corneal re-epithelialization for dendritic ulcers (6 days) and geographic ulcers (7 days) was similar for both therapies.
In other clinical studies, trifluridine was evaluated in the treatment of herpes simplex virus keratitis in patients who were unresponsive or intolerant to the topical administration of idoxuridine or vidarabine. Trifluridine was effective in 138 of 150 (92%) patients (109 of 114 with dendritic and 29 of 36 with geographic ulcers) as evidenced by corneal re-epithelialization. The mean time to corneal re-epithelialization was 6 days for patients with dendritic ulcers and 12 days for patients with geographic ulcers.
The clinical efficacy of trifluridine in the treatment of stromal keratitis and uveitis due to herpes simplex virus or ophthalmic infections caused by vacciniavirus and adenovirus has not been established by well-controlled clinical trials. Trifluridine has not been shown to be effective in the prophylaxis of herpes simplex virus keratoconjunctivitis and epithelial keratitis by well-controlled clinical trials. Trifluridine is not effective against bacterial, fungal or chlamydial infections of the cornea or nonviral trophic lesions.
HOW SUPPLIED: Trifluridine Ophthalmic Solution, 1% in a 7.5 mL fill packaged in a natural plastic bottle with a natural plastic flat tip and a white plastic closure.
7.5mL- NDC 61314-044-75
Store under refrigeration 2-8°C (36-46°F).