Terbutaline Sulfate - Terbutaline Sulfate tablet prescribing information
WARNING: TOCOLYSIS
Oral terbutaline sulfate has not been approved and should not be used for acute or maintenance tocolysis. In particular, terbutaline sulfate should not be used for maintenance tocolysis in the outpatient or home setting. Serious adverse reactions, including death, have been reported after administration of terbutaline sulfate to pregnant women. In the mother, these adverse reactions include increased heart rate, transient hyperglycemia, hypokalemia, cardiac arrhythmias, pulmonary edema and myocardial ischemia. Increased fetal heart rate and neonatal hypoglycemia may occur as a result of maternal administration [see CONTRAINDICATIONS , Tocolysis ].
INDICATIONS AND USAGE
Terbutaline sulfate is indicated for the prevention and reversal of bronchospasm in patients 12 years of age and older with asthma and reversible bronchospasm associated with bronchitis and emphysema.
DOSAGE AND ADMINISTRATION
Adults
The usual oral dose of terbutaline sulfate for adults is 5 mg administered at approximately six-hour intervals, three times daily, during the hours the patient is usually awake. If side effects are particularly disturbing, the dose may be reduced to 2.5 mg three times daily, and still provide a clinically significant improvement in pulmonary function. The total dose within 24 hours should not exceed 15 mg.
Children
Terbutaline sulfate is not recommended for use in children below the age of 12 years. A dosage of 2.5 mg three times daily is recommended for children 12 to 15 years of age. The total dose within 24 hours should not exceed 7.5 mg.
If a previously effective dosage regimen fails to provide the usual relief, medical advice should be sought immediately as this is often a sign of seriously worsening asthma that would require reassessment of therapy.
CONTRAINDICATIONS
ADVERSE REACTIONS
Adverse reactions observed with terbutaline sulfate are similar to those commonly seen with other sympathomimetic amines. All of these reactions are generally transient in nature and usually do not require treatment. The frequency of these side effects appears to diminish with continued therapy.
The following table lists the adverse reactions seen in 199 patients treated with terbutaline sulfate tablets during six double-blind crossover studies and four double-blind parallel studies (short- and long-term) performed in the United States.
Reaction | % | |
Nervous System | ||
Nervousness | 35.0 | |
Tremor | 15.0 | |
Somnolence | 5.5 | |
Dizziness | 3.5 | |
Anxiety | 1.0 | |
Insomnia | 1.5 | |
Cardiovascular | ||
Palpitations | 5.0 | |
Tachycardia | 3.5 | |
Extrasystoles ventricular | 1.5 | |
Vasodilations | 1.0 | |
Digestive | ||
Nausea | 3.0 | |
Dry mouth | 1.5 | |
Body as a Whole | ||
Headache | 7.5 | |
Asthenia | 2.0 | |
Skin and Appendages | ||
Sweating | 1.0 | |
The following adverse effects each occurred in fewer than 1% of patients: hallucinations, rash, paresthesia, hypertonia, (muscle cramps), vomiting.
There have been rare reports of elevations in liver enzymes and of hypersensitivity vasculitis.
DESCRIPTION
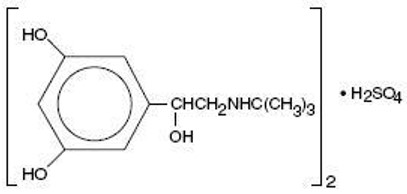
Terbutaline sulfate USP is a beta-adrenergic agonist bronchodilator available as tablets of 2.5 mg (2.05 mg of the free base) and 5 mg (4.1 mg of the free base) for oral administration. Terbutaline sulfate is ±-α-[( tert –butylamino) methyl]-3,5-dihydroxybenzyl alcohol sulfate (2:1) (salt). The molecular formula is (C 12 H 19 NO 3 ) 2 • H 2 SO 4 and the structural formula is
Terbutaline sulfate USP is a white to gray-white crystalline powder. It is odorless or has a faint odor of acetic acid. It is soluble in water and in 0.1N hydrochloric acid, slightly soluble in methanol, and insoluble in chloroform. Its molecular weight is 548.65.
Inactive Ingredients: anhydrous lactose, pregelatinized corn starch, microcrystalline cellulose, povidone, and magnesium stearate.
CLINICAL PHARMACOLOGY
In vitro and in vivo pharmacologic studies have demonstrated that terbutaline exerts a preferential effect on beta 2 -adrenergic receptors. While it is recognized that beta2-adrenergic receptors are the predominant receptors in bronchial smooth muscle, data indicate that there is a population of beta2-receptors in the human heart, existing in a concentration between 10% to 50%. The precise function of these receptors has not been established [see WARNINGS ]. In controlled clinical studies in patients given terbutaline sulfate orally, proportionally greater changes occurred in pulmonary function parameters than in heart rate or blood pressure. While this suggests a relative preference for the beta 2 -receptors in man, the usual cardiovascular effects commonly associated with other sympathomimetic agents were also observed with terbutaline sulfate.
The pharmacologic effects of beta-adrenergic agonists, including terbutaline, are at least in part attributable to stimulation through beta-adrenergic receptors of intracellular adenyl cyclase, the enzyme which catalyzes the conversion of adenosine triphosphate (ATP) to cyclic 3’, 5’-adenosine monophosphate (cAMP). Increased cAMP levels are associated with relaxation of bronchial smooth muscle and inhibition of release of mediators of immediate hypersensitivity from cells, especially from mast cells.
Controlled clinical studies have shown that terbutaline sulfate relieves bronchospasm in chronic obstructive pulmonary disease by significantly increasing pulmonary function (e.g., an increase of 15% or more in FEV 1 and in FEF 25%-75% ). After administration of terbutaline sulfate tablets, a measurable change in flow rate usually occurs within 30 minutes, and a clinically significant improvement in pulmonary function occurs within 60 to 120 minutes. The maximum effect usually occurs within 120 to 180 minutes. Terbutaline sulfate also produces a clinically significant decrease in airway and pulmonary resistance, which persists for 4 hours or longer. Significant bronchodilator action (as measured by airway resistance, FEF 25%-75% or PEFR) has also been demonstrated for up to 8 hours in some studies.
In studies comparing the effectiveness of terbutaline sulfate with that of ephedrine for up to 3 months, both drugs maintained a significant improvement in pulmonary function throughout this period of treatment.
Preclinical
Studies in laboratory animals (minipigs, rodents, and dogs) have demonstrated the occurrence of cardiac arrhythmias and sudden death (with histologic evidence of myocardial necrosis) when beta-agonists and methylxanthines were administered concurrently. The clinical significance of these findings is unknown.
Pharmacokinetics
Oral administration of 5-mg terbutaline sulfate tablets or 5 mg terbutaline sulfate in solution in 17 healthy, adult, male subjects, resulted in mean (SD) peak plasma terbutaline concentration of 8.3 (3.9) and 8.6 (3.6) ng/mL, which were observed at median (range) times of 2 (1 to 3) and 1.5 (0.5 to 3.0) hours after dosing. The mean (SD) AUC (0-48) values were 54.6 (26.8) and 53.1 (23.5) hr•ng/mL, and corresponded to a bioavailability of 103% for the tablet relative to the solution.
After oral administration of terbutaline, 51 to 62 mcg/kg of body weight, to 3 healthy male subjects, peak serum levels of 3.1 to 6.2 ng/mL were observed 1 to 3 hours later. In the same study, after 3 days only 30% to 50% of the dose was recovered from urine and the remainder from the feces, which may indicate poor absorption.
After an oral dose to asthmatic patients, the elimination half-life of terbutaline was approximately 3.4 hours.
In comparison to oral dosing, subcutaneous administration of 0.5 mg of terbutaline sulfate to 17 healthy, adult, male subjects resulted in a mean (SD) peak plasma terbutaline concentration of 9.6 (3.6) ng/mL, which was observed at a median (range) time of 0.5 (0.08 to 1.0) hours after dosing. The mean (SD) AUC (0-48) and total body clearance values were 29.4 (14.2) hr•ng/mL, and 311 (112) mL/min, respectively. The terminal half-life was determined in 9 of the 17 subjects and had a mean (SD) of 5.7 (2.0) hours.
About 90% of the drug was excreted in the urine at 96 hours after subcutaneous administration, with about 60% of this being unchanged drug. The sulfate conjugate is a major metabolite of terbutaline, and urinary excretion is the primary route of elimination.
There are no reports of any clinical pharmacokinetic studies investigating dose proportionality, effect of food, or special population studies with terbutaline.
HOW SUPPLIED
Terbutaline sulfate tablets, USP are packaged in bottles of 90 tablets. Descriptions of the 2.5 and 5 mg tablets follow:
Tablets 2.5 mg—round, white, scored (imprinted LCI over 1318)
Bottles of 90 NDC 62135-524-90
Tablets 5 mg—round, white, scored (imprinted LCI over 1311)
Bottles of 90 NDC 62135-525-90
Store at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined in the USP with a child-resistant closure.
Manufactured for: Chartwell RX, LLC. Congers, NY 10920
L71336 Revised 02/2023