Get your patient on Sulfamethoxazole And Trimethoprim - Sulfamethoxazole And Trimethoprim tablet (Sulfamethoxazole And Trimethoprim)
Sulfamethoxazole And Trimethoprim - Sulfamethoxazole And Trimethoprim tablet prescribing information
INDICATIONS AND USAGE
To reduce the development of drug-resistant bacteria and maintain the effectiveness of sulfamethoxazole and trimethoprim tablets and other antibacterial drugs, sulfamethoxazole and trimethoprim tablets should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to empiric selection of therapy.
Urinary Tract Infections:
For the treatment of urinary tract infections due to susceptible strains of the following organisms: Escherichia coli , Klebsiella species, Enterobacter species, Morganella morganii , Proteus mirabilis and Proteus vulgaris . It is recommended that initial episodes of uncomplicated urinary tract infections be treated with a single effective antibacterial agent rather than the combination.
Acute Otitis Media:
For the treatment of acute otitis media in pediatric patients due to susceptible strains of Streptococcus pneumoniae or Haemophilus influenzae when in the judgment of the physician sulfamethoxazole and trimethoprim offers some advantage over the use of other antimicrobial agents. To date, there are limited data on the safety of repeated use of sulfamethoxazole and trimethoprim in pediatric patients under two years of age. Sulfamethoxazole and trimethoprim is not indicated for prophylactic or prolonged administration in otitis media at any age.
Acute Exacerbations of Chronic Bronchitis in Adults:
For the treatment of acute exacerbations of chronic bronchitis due to susceptible strains of Streptococcus pneumoniae or Haemophilus influenzae when a physician deems that sulfamethoxazole and trimethoprim could offer some advantage over the use of a single antimicrobial agent.
Shigellosis:
For the treatment of enteritis caused by susceptible strains of Shigella flexneri and Shigella sonnei when antibacterial therapy is indicated.
Pneumocystis jirovecii Pneumonia:
For the treatment of documented Pneumocystis jirovecii pneumonia and for prophylaxis against P. jirovecii pneumonia in individuals who are immunosuppressed and considered to be at an increased risk of developing P. jirovecii pneumonia.
Traveler's Diarrhea in Adults:
For the treatment of traveler's diarrhea due to susceptible strains of enterotoxigenic E. coli .
DOSAGE AND ADMINISTRATION
Sulfamethoxazole and trimethoprim is contraindicated in pediatric patients less than 2 months of age.
Urinary Tract Infections and Shigellosis in Adults and Pediatric Patients, and Acute Otitis Media in Children
Adults: The usual adult dosage in the treatment of urinary tract infections is 1 sulfamethoxazole and trimethoprim DS (double strength) tablet or 2 sulfamethoxazole and trimethoprim tablets every 12 hours for 10 to 14 days. An identical daily dosage is used for 5 days in the treatment of shigellosis.
Children: The recommended dose for children with urinary tract infections or acute otitis media is 40 mg/kg sulfamethoxazole and 8 mg/kg trimethoprim per 24 hours, given in two divided doses every 12 hours for 10 days. An identical daily dosage is used for 5 days in the treatment of shigellosis. The following table is a guideline for the attainment of this dosage:
Children 2 months of age or older:
Weight | Dose-every 12 hours | |
lb | kg | Tablets |
22 | 10 | |
44 | 20 | 1 |
66 | 30 | 1½ |
88 | 40 | 2 or 1 DS tablet |
For Patients with Impaired Renal Function
When renal function is impaired, a reduced dosage should be employed using the following table:
Creatinine Clearance (mL/min) | styleCode="Toprule botrule"> Recommended Dosage Regimen |
Above 30 | Usual standard regimen |
15–30 | ½ the usual regimen |
Below 15 | Use not recommended |
Acute Exacerbations of Chronic Bronchitis in Adults
The usual adult dosage in the treatment of acute exacerbations of chronic bronchitis is 1 sulfamethoxazole and trimethoprim DS (double strength) tablet or 2 sulfamethoxazole and trimethoprim tablets every 12 hours for 14 days.
Pneumocystis Jirovecii Pneumonia Treatment
Adults and Children:
The recommended dosage for treatment of patients with documented Pneumocystis jirovecii pneumonia is 75 to 100 mg/kg sulfamethoxazole and 15 to 20 mg/kg trimethoprim per 24 hours given in equally divided doses every 6 hours for 14 to 21 days. 12 The following table is a guideline for the upper limit of this dosage:
Weight | Dose-every 6 hours | |
lb | kg | Tablets |
18 | 8 | |
35 | 16 | 1 |
53 | 24 | 1½ |
70 | 32 | 2 or 1 DS tablet |
88 | 40 | 2½ |
106 | 48 | 3 or 1½ DS tablets |
141 | 64 | 4 or 2 DS tablets |
176 | 80 | 5 or 2½ DS tablets |
For the lower limit dose (75 mg/kg sulfamethoxazole and 15 mg/kg trimethoprim per 24 hours) administer 75% of the dose in the above table.
Prophylaxis:
Adults:
The recommended dosage for prophylaxis in adults is 1 sulfamethoxazole and trimethoprim DS (double strength) tablet daily. 13
Children:
For children, the recommended dose is 750 mg/m 2 /day sulfamethoxazole with 150 mg/m 2 /day trimethoprim given orally in equally divided doses twice a day, on 3 consecutive days per week. The total daily dose should not exceed 1600 mg sulfamethoxazole and 320 mg trimethoprim. 14 The following table is a guideline for the attainment of this dosage in children:
Body Surface Area | Dose-every 12 hours |
(m 2 ) | Tablets |
0.26 | |
0.53 | ½ |
1.06 | 1 |
Traveler's Diarrhea in Adults
For the treatment of traveler's diarrhea, the usual adult dosage is 1 sulfamethoxazole and trimethoprim DS (double strength) tablet or 2 sulfamethoxazole and trimethoprim tablets every 12 hours for 5 days.
CONTRAINDICATIONS
Sulfamethoxazole and trimethoprim is contraindicated in the following situations:
- known hypersensitivity to trimethoprim or sulfonamides
- history of drug-induced immune thrombocytopenia with use of trimethoprim and/or sulfonamides
- documented megaloblastic anemia due to folate deficiency
- pediatric patients less than 2 months of age
- marked hepatic damage
- severe renal insufficiency when renal function status cannot be monitored
- concomitant administration with dofetilide (see PRECAUTIONS ).
ADVERSE REACTIONS
The following adverse reactions associated with the use of sulfamethoxazole and trimethoprim were identified in clinical trials, postmarketing or published reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The most common adverse reactions are gastrointestinal disturbances (nausea, vomiting, anorexia) and allergic skin reactions (such as rash and urticaria). Fatalities and serious adverse reactions, including severe cutaneous adverse reactions (SCARs), including Stevens-Johnson syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms (DRESS), acute febrile neutrophilic dermatosis (AFND), acute generalized erythematous pustulosis (AGEP); fulminant hepatic necrosis; agranulocytosis, aplastic anemia and other blood dyscrasias; acute and delayed lung injury; anaphylaxis and circulatory shock have occurred with the administration of sulfamethoxazole and trimethoprim products (see WARNINGS).
Hematologic: Agranulocytosis, aplastic anemia, thrombocytopenia, leukopenia, neutropenia, hemolytic anemia, megaloblastic anemia, hypoprothrombinemia, methemoglobinemia, eosinophilia, thrombotic thrombocytopenic purpura, idiopathic thrombocytopenic purpura.
Allergic Reactions: Stevens-Johnson syndrome, toxic epidermal necrolysis, anaphylaxis, allergic myocarditis, erythema multiforme, exfoliative dermatitis, angioedema, drug fever, chills, Henoch-Schoenlein purpura, serum sickness-like syndrome, generalized allergic reactions, generalized skin eruptions, photosensitivity, conjunctival and scleral injection, pruritus, urticaria, rash, periarteritis nodosa, systemic lupus erythematosus, drug reaction with eosinophilia and systemic symptoms (DRESS), acute generalized erythematous pustulosis (AGEP), and acute febrile neutrophilic dermatosis (AFND) (see WARNINGS ).
Gastrointestinal: Hepatitis (including cholestatic jaundice and hepatic necrosis), elevation of serum transaminase and bilirubin, pseudomembranous enterocolitis, pancreatitis, stomatitis, glossitis, nausea, emesis, abdominal pain, diarrhea, anorexia.
Genitourinary: Renal failure, interstitial nephritis, BUN and serum creatinine elevation, renal insufficiency, oliguria and anuria, crystalluria and nephrotoxicity in association with cyclosporine.
Metabolic and Nutritional: Hyperkalemia, hyponatremia (see PRECAUTIONS : Electrolyte Abnormalities ) , metabolic acidosis.
Neurologic: Aseptic meningitis, convulsions, peripheral neuritis, ataxia, vertigo, tinnitus, headache.
Psychiatric: Hallucinations, depression, apathy, nervousness.
Endocrine: The sulfonamides bear certain chemical similarities to some goitrogens, diuretics (acetazolamide and the thiazides) and oral hypoglycemic agents. Cross-sensitivity may exist with these agents. Diuresis and hypoglycemia have occurred.
Musculoskeletal: Arthralgia, myalgia, rhabdomyolysis.
Respiratory: Cough, shortness of breath and pulmonary infiltrates, acute eosinophilic pneumonia, acute and delayed lung injury, interstitial lung disease, acute respiratory failure (see WARNINGS ).
Cardiovascular System: QT prolongation resulting in ventricular tachycardia and torsades de pointes , circulatory shock (see WARNINGS ).
Miscellaneous: Weakness, fatigue, insomnia.
DESCRIPTION
Sulfamethoxazole and trimethoprim is a synthetic antibacterial combination product available in DS (double strength) tablets, each containing 800 mg sulfamethoxazole and 160 mg trimethoprim and in tablets, each containing 400 mg sulfamethoxazole and 80 mg trimethoprim for oral administration.
Sulfamethoxazole, USP is N 1 -(5-methyl-3-isoxazolyl) sulfanilamide; the molecular formula is C 10 H 11 N 3 O 3 S. It is an almost white, odorless, tasteless compound with a molecular weight of 253.28 and the following structural formula:
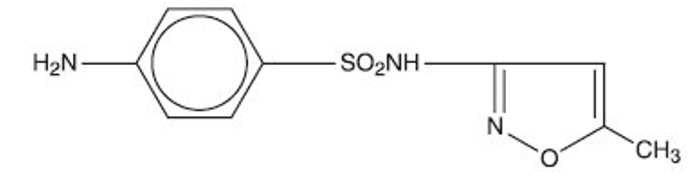
Trimethoprim, USP is 2,4-diamino-5-(3,4,5-trimethoxybenzyl) pyrimidine; the molecular formula is C 14 H 18 N 4 O 3 . It is a white to light yellow, odorless, bitter compound with a molecular weight of 290.3 and the following structural formula:
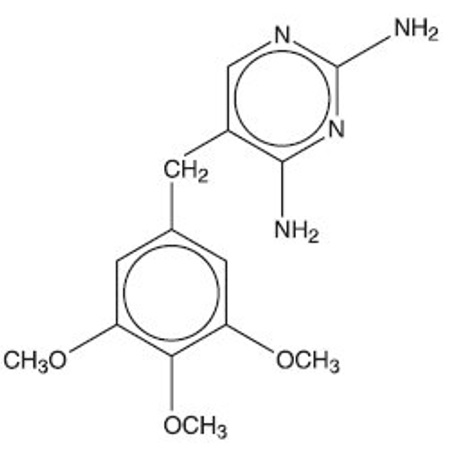
Inactive ingredients: Docusate sodium 85%, magnesium stearate, povidone, pregelatinized starch, and sodium starch glycolate.
CLINICAL PHARMACOLOGY
Sulfamethoxazole and trimethoprim is rapidly absorbed following oral administration. Both sulfamethoxazole and trimethoprim exist in the blood as unbound, protein-bound and metabolized forms; sulfamethoxazole also exists as the conjugated form. Sulfamethoxazole is metabolized in humans to at least 5 metabolites: the N 4 -acetyl-, N 4 -hydroxy-, 5-methylhydroxy-, N 4 -acetyl-5-methylhydroxy- sulfamethoxazole metabolites, and an N-glucuronide conjugate. The formulation of N 4 -hydroxy metabolite is mediated via CYP2C9.
Trimethoprim is metabolized in vitro to 11 different metabolites, of which, five are glutathione adducts and six are oxidative metabolites, including the major metabolites, 1- and 3-oxides and the 3- and 4-hydroxy derivatives.
The free forms of sulfamethoxazole and trimethoprim are considered to be the therapeutically active forms.
In vitro studies suggest that trimethoprim is a substrate of P-glycoprotein, OCT1 and OCT2, and that sulfamethoxazole is not a substrate of P-glycoprotein.
Approximately 70% of sulfamethoxazole and 44% of trimethoprim are bound to plasma proteins. The presence of 10 mg percent sulfamethoxazole in plasma decreases the protein binding of trimethoprim by an insignificant degree; trimethoprim does not influence the protein binding of sulfamethoxazole.
Peak blood levels for the individual components occur 1 to 4 hours after oral administration. The mean serum half-lives of sulfamethoxazole and trimethoprim are 10 and 8 to 10 hours, respectively. However, patients with severely impaired renal function exhibit an increase in the half-lives of both components, requiring dosage regimen adjustment (see DOSAGE AND ADMINISTRATION section). Detectable amounts of sulfamethoxazole and trimethoprim are present in the blood 24 hours after drug administration. During administration of 800 mg sulfamethoxazole and 160 mg trimethoprim b.i.d., the mean steady-state plasma concentration of trimethoprim was 1.72 mcg/mL. The steady-state mean plasma levels of free and total sulfamethoxazole were 57.4 mcg/mL and 68.0 mcg/mL, respectively. These steady-state levels were achieved after three days of drug administration. 1 Excretion of sulfamethoxazole and trimethoprim is primarily by the kidneys through both glomerular filtration and tubular secretion. Urine concentrations of both sulfamethoxazole and trimethoprim are considerably higher than are the concentrations in the blood. The average percentage of the dose recovered in urine from 0 to 72 hours after a single oral dose of sulfamethoxazole and trimethoprim is 84.5% for total sulfonamide and 66.8% for free trimethoprim. Thirty percent of the total sulfonamide is excreted as free sulfamethoxazole, with the remaining as N 4 -acetylated metabolite. 2 When administered together as sulfamethoxazole and trimethoprim, neither sulfamethoxazole nor trimethoprim affects the urinary excretion pattern of the other.
Both sulfamethoxazole and trimethoprim distribute to sputum, vaginal fluid and middle ear fluid; trimethoprim also distributes to bronchial secretion, and both pass the placental barrier and are excreted in human milk
Pharmacokinetics in Pediatric Patients
A simulation conducted with data from a pharmacokinetic study in 153 infants and children demonstrated that mean steady state AUC and maximum plasma concentration of trimethoprim and sulfamethoxazole would be comparable between pediatric patients 2 months to 18 years receiving 8/40 (trimethoprim/ sulfamethoxazole) mg/kg/day divided every 12 hours and adult patients receiving 320/1600 (trimethoprim/ sulfamethoxazole) mg/day.
Pharmacokinetics in Geriatric Patients
The pharmacokinetics of sulfamethoxazole 800 mg and trimethoprim 160 mg were studied in 6 geriatric subjects (mean age: 78.6 years) and 6 young healthy subjects (mean age: 29.3 years) using a non-US approved formulation. Pharmacokinetic values for sulfamethoxazole in geriatric subjects were similar to those observed in young adult subjects. The mean renal clearance of trimethoprim was significantly lower in geriatric subjects compared with young adult subjects (19 mL/h/kg vs. 55 mL/h/kg). However, after normalizing by body weight, the apparent total body clearance of trimethoprim was on average 19% lower in geriatric subjects compared with young adult subjects. 3
Microbiology
Mechanism of Action
Sulfamethoxazole inhibits bacterial synthesis of dihydrofolic acid by competing with para-aminobenzoic acid (PABA). Trimethoprim blocks the production of tetrahydrofolic acid from dihydrofolic acid by binding to and reversibly inhibiting the required enzyme, dihydrofolate reductase. Thus, sulfamethoxazole and trimethoprim blocks two consecutive steps in the biosynthesis of nucleic acids and proteins essential to many bacteria.
Resistance
In vitro studies have shown that bacterial resistance develops more slowly with both sulfamethoxazole and trimethoprim in combination than with either sulfamethoxazole or trimethoprim alone.
Antimicrobial Activity
Sulfamethoxazole and trimethoprim have been shown to be active against most isolates of the following microorganisms, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section.
Aerobic gram-positive bacteria:
Streptococcus pneumoniae
Aerobic gram-negative bacteria:
Escherichia coli (including susceptible enterotoxigenic strains implicated in traveler's diarrhea) Klebsiella species Enterobacter species Haemophilus influenzae Morganella morganii Proteus mirabilis Proteus vulgaris Shigella flexneri Shigella sonnei
Other Microorganisms:
Pneumocystis jirovecii
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC .
HOW SUPPLIED
Sulfamethoxazole and Trimethoprim Tablets, USP are supplied as follows:
Sulfamethoxazole and Trimethoprim Tablets, USP 400 mg/80 mg are white to off white, round, scored tablets, debossed with “CE” on one side and “103” on the other, supplied in bottles of 90 (NDC 62135-897-90) and 180 (62135-897-18).
Sulfamethoxazole and Trimethoprim (DS) Tablets, USP 800 mg/160 mg are white to off white, oval, scored tablets, debossed with “CE” on one side and “104” on the other, supplied in bottles of 90 (NDC 62135-898-90) and 180 (NDC 62135-898-18).
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined