Get your patient on Sucralfate - Sucralfate suspension (Sucralfate)
Sucralfate - Sucralfate suspension prescribing information
INDICATIONS AND USAGE
Sucralfate Oral Suspension is indicated in the short-term (up to 8 weeks) treatment of active duodenal ulcer.
DOSAGE AND ADMINISTRATION
Active Duodenal Ulcer: The recommended adult oral dosage for duodenal ulcer is 1 gram (10 mL) four times per day. Sucralfate Oral Suspension should be administered on an empty stomach.
Antacids may be prescribed as needed for relief of pain but should not be taken within one-half hour before or after Sucralfate Oral Suspension.
While healing with sucralfate may occur during the first week or two, treatment should be continued for 4 to 8 weeks unless healing has been demonstrated by x-ray or endoscopic examination.
Elderly: In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy (See PRECAUTIONS , Geriatric Use ).
Call your doctor for medical advice about side effects. You may report side effects to Allergan USA, Inc. at 1-800-678-1605 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
CONTRAINDICATIONS
Sucralfate Oral Suspension is contraindicated for patients with known hypersensitivity reactions to the active substance or to any of the excipients.
ADVERSE REACTIONS
Adverse reactions to sucralfate tablets in clinical trials were minor and only rarely led to discontinuation of the drug. In studies involving over 2,700 patients treated with sucralfate, adverse effects were reported in 129 (4.7%).
Constipation was the most frequent complaint (2%). Other adverse effects reported in less than 0.5% of the patients are listed below by body system:
Gastrointestinal: diarrhea, dry mouth, flatulence, gastric discomfort, indigestion, nausea, vomiting
Dermatological: pruritus, rash
Nervous System: dizziness, insomnia, sleepiness, vertigo
Other: back pain, headache
Post-marketing cases of hypersensitivity have been reported with the use of sucralfate oral suspension, including anaphylactic reactions, dyspnea, lip swelling, edema of the mouth, pharyngeal edema, pruritus, rash, swelling of the face and urticaria.
Cases of bronchospasm, laryngeal edema and respiratory tract edema have been reported with an unknown oral formulation of sucralfate.
Cases of hyperglycemia have been reported with sucralfate.
Bezoars have been reported in patients treated with sucralfate. The majority of patients had underlying medical conditions that may predispose to bezoar formation (such as delayed gastric emptying) or were receiving concomitant enteral tube feedings.
Drug Interactions
Some studies have shown that simultaneous sucralfate administration in healthy volunteers reduced the extent of absorption (bioavailability) of single doses of the following: cimetidine, digoxin, fluoroquinolone antibiotics, ketoconazole, l-thyroxine, phenytoin, quinidine, ranitidine, tetracycline, and theophylline. Subtherapeutic prothrombin times with concomitant warfarin and sucralfate therapy have been reported in spontaneous and published case reports. However, two clinical studies have demonstrated no change in either serum warfarin concentration or prothrombin time with the addition of sucralfate to chronic warfarin therapy.
The mechanism of these interactions appears to be nonsystemic in nature, presumably resulting from sucralfate binding to the concomitant agent in the gastrointestinal tract. In all cases studied to date (cimetidine, ciprofloxacin, digoxin, norfloxacin, ofloxacin, and ranitidine), dosing the concomitant medication 2 hours before sucralfate eliminated the interaction. Due to Sucralfate Oral Suspension's potential to alter the absorption of some drugs, Sucralfate Oral Suspension should be administered separately from other drugs when alterations in bioavailability are felt to be critical. In these cases, patients should be monitored appropriately.
DESCRIPTION
Sucralfate Oral Suspension contains sucralfate and sucralfate is an α-D-glucopyranoside, β-D- fructofuranosyl-, octakis-(hydrogen sulfate), aluminum complex.
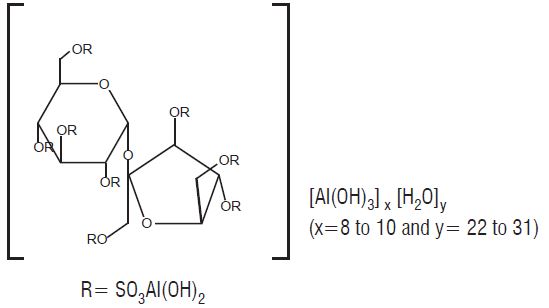
Sucralfate Oral Suspension for oral administration contains 1 g of sucralfate per 10 mL. Sucralfate Oral Suspension also contains: colloidal silicon dioxide, cherry flavor (containing artificial flavors, benzyl alcohol, lactic acid and maltodextrin), FD&C Red #40, glycerin, methylcellulose, methylparaben, microcrystalline cellulose, purified water, simethicone emulsion and sorbitol solution. Therapeutic category: antiulcer.
CLINICAL PHARMACOLOGY
Sucralfate is only minimally absorbed from the gastrointestinal tract. The small amounts of the sulfated disaccharide that are absorbed are excreted primarily in the urine.
Although the mechanism of sucralfate's ability to accelerate healing of duodenal ulcers remains to be fully defined, it is known that it exerts its effect through a local, rather than systemic, action. The following observations also appear pertinent:
1. Studies in human subjects and with animal models of ulcer disease have shown that sucralfate forms an ulcer-adherent complex with proteinaceous exudate at the ulcer site.
2.000000000000000e+00 In vitro , a sucralfate-albumin film provides a barrier to diffusion of hydrogen ions.
3. In human subjects, sucralfate given in doses recommended for ulcer therapy inhibits pepsin activity in gastric juice by 32%.
In vitro , sucralfate adsorbs bile salts.
These observations suggest that sucralfate's antiulcer activity is the result of formation of an ulcer-adherent complex that covers the ulcer site and protects it against further attack by acid, pepsin, and bile salts. There are approximately 14 to 16 mEq of acid-neutralizing capacity per 1 g dose of sucralfate.
CLINICAL TRIALS
In a multicenter, double-blind, placebo-controlled study of Sucralfate Oral Suspension, a dosage regimen of 1 gram (10 mL) four times daily was demonstrated to be superior to placebo in ulcer healing.
| Results from Clinical Trials Healing Rates for Acute Duodenal Ulcer | ||||
| Treatment | n | Week 2 Healing Rates | Week 4 Healing Rates | Week 8 Healing Rates |
| Sucralfate Oral Suspension | 145 | 23(16%)• | 66(46%) † | 95(66%) ‡ |
| Placebo | 147 | 10(7%) | 39(27%) | 58(39%) |
• P =0.016 † P =0.001 ‡ P =0.0001
Equivalence of sucralfate oral suspension to sucralfate tablets has not been demonstrated.
HOW SUPPLIED
Sucralfate Oral Suspension 1 g/10 mL is a light pink to pink suspension supplied in the following:
- NDC 64380-149-01: 420 mL bottles
- NDC 64380-149-03: 5 x 10 mL unit-dose cups in a tray.
- NDC 64380-149-04: 10 x 10 mL unit-dose cups in a tray, 3 trays per carton.
- NDC 64380-149-05: 10 x 10 mL unit-dose cups in a tray, 4 trays per carton.
- NDC 64380-149-06: 10 x 10 mL unit-dose cups in a tray, 5 trays per carton.
- NDC 64380-149-07: 10 x 10 mL unit-dose cups in a tray, 10 trays per carton.
SHAKE WELL BEFORE USING AVOID FREEZING .
Store at controlled room temperature 20-25°C (68-77°F) [see USP].
Rx Only
Manufactured by:
Strides Pharma Science Limited.
Bengaluru – 562106, India
Distributed by:
Strides Pharma Inc.
Bridgewater, NJ 088107
Revised : 07/2025