Get your patient on Silver Sulfadiazine - Silver Sulfadiazine cream (Silver Sulfadiazine)
Silver Sulfadiazine - Silver Sulfadiazine cream prescribing information
INDICATIONS AND USAGE
Silver sulfadiazine cream, USP 1% is a topical antimicrobial drug indicated as an adjunct for the prevention and treatment of wound sepsis in patients with second and third degree burns.
DOSAGE AND ADMINISTRATION
Prompt institution of appropriate regimens for care of the burned patient is of prime importance and includes the control of shock and pain. The burn wounds are then cleansed and debrided; silver sulfadiazine cream, USP 1% is then applied under sterile conditions. The burn areas should be covered with silver sulfadiazine cream, USP 1% at all times. The cream should be applied once to twice daily to a thickness of approximately one sixteenth of an inch. Whenever necessary, the cream should be reapplied to any areas from which it has been removed by patient activity. Administration may be accomplished in minimal time because dressings are not required. However, if individual patient requirements make dressings necessary, they may be used. Reapply immediately after hydrotherapy. Treatment with silver sulfadiazine cream, USP 1% should be continued until satisfactory healing has occurred or until the burn site is ready for grafting. The drug should not be withdrawn from the therapeutic regimen while there remains the possibility of infection except if a significant adverse reaction occurs.
CONTRAINDICATIONS
Silver sulfadiazine cream, USP 1% is contraindicated in patients who are hypersensitive to silver sulfadiazine or any of the other ingredients in the preparation. Because sulfonamide therapy is known to increase the possibility of kernicterus, silver sulfadiazine cream, USP 1% should not be used on pregnant women approaching or at term, on premature infants, or on newborn infants during the first 2 months of life.
ADVERSE REACTIONS
Several cases of transient leukopenia have been reported in patients receiving silver sulfadiazine therapy. 1,2,3 Leukopenia associated with silver sulfadiazine administration is primarily characterized by decreased neutrophil count. Maximal white blood cell depression occurs within two to four days of initiation of therapy. Rebound to normal leukocyte levels follows onset within two to three days. Recovery is not influenced by continuation of silver sulfadiazine therapy. An increased incidence has been seen in patients treated concurrently with cimetidine. Other infrequently occurring events include skin necrosis, erythema multiforme, skin discoloration, burning sensation, rashes, and interstitial nephritis. Reduction in bacterial growth after application of topical antibacterial agents has been reported to permit spontaneous healing of deep partial-thickness burns by preventing conversion of the partial thickness to full thickness by sepsis. However, reduction in bacterial colonization has caused delayed separation, in some cases necessitating escharotomy in order to prevent contracture.
DESCRIPTION
Silver sulfadiazine cream, USP 1% is a soft, white, water dispersible cream containing the antimicrobial agent silver sulfadiazine in micronized form for topical application. Each gram of silver sulfadiazine cream contains 10mg of micronized silver sulfadiazine. This active agent has the following structural formula:
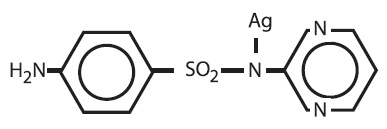
Silver sulfadiazine cream contains 1% w/w silver sulfadiazine. The vehicle in which the active ingredient is dispersed consists of water, stearyl alcohol, white petrolatum, polyoxyl 40 stearate, propylene glycol, isopropyl myristate, and sorbitan monooleate with 0.3% methylparaben as a preservative.
CLINICAL PHARMACOLOGY
Silver sulfadiazine has broad antimicrobial activity. It is bactericidal for many gram-negative and gram-positive bacteria as well as being effective against yeast. Results from in vitro testing are listed below. Sufficient data have been obtained to demonstrate that silver sulfadiazine will inhibit bacteria that are resistant to other antimicrobial agents and that the compound is superior to sulfadiazine. Studies utilizing radioactive micronized silver sulfadiazine, electron microscopy, and biochemical techniques have revealed that the mechanism of action of silver sulfadiazine on bacteria differs from silver nitrate and sodium sulfadiazine. Silver sulfadiazine acts only on the cell membrane and cell wall to produce its bactericidal effect.
| Genus and Species | 50 µg/mL | 100 µg/mL |
| 130/130 | 130/130 |
| 7/7 | 7/7 |
| 48/50 | 50/50 |
| 24/24 | 24/24 |
| 53/54 | 54/54 |
| 63/63 | 63/63 |
| 27/28 | 28/28 |
| 53/53 | 53/53 |
| 10/10 | 10/10 |
| 2/2 | 2/2 |
| 2/2 | 2/2 |
| 1/1 | 1/1 |
| 10/10 | 10/10 |
| 10/11 | 11/11 |
| 100/101 | 100/101 |
| 51/51 | 51/51 |
| 4/4 | 4/4 |
| 52/53 | 52/53 |
| 2/2 | 2/2 |
| 0/2 | 2/2 |
| 43/50 | 50/50 |
Silver sulfadiazine is not a carbonic anhydrase inhibitor and may be useful in situations where such agents are contraindicated.
HOW SUPPLIED
| Silver sulfadiazine cream, USP 1% - white to off-white cream | NDC Number | Size |
| 67877-124-20 | 20g tube | |
| 67877-124-25 | 25g tube | |
| 67877-124-05 | 50g tube | |
| 67877-124-85 | 85g tube | |
| 67877-124-50 | 50g jar | |
| 67877-124-40 | 400g jar | |
| 67877-124-10 | 1000g jar | |
Store at room temperature. 15°-30°C (59°-86°F).
Manufactured for: Ascend Laboratories, LLC Parsippany, NJ 07054 Manufactured by: Crown Laboratories, Inc. Johnson City, TN 37604
Product Information: 1-877-272-7901 Revised: DEC 2018 P1840.05