Rubraca - Rucaparib tablet, Film Coated prescribing information
INDICATIONS AND USAGE
RUBRACA is a poly (ADP-ribose) polymerase (PARP) inhibitor indicated:
Ovarian cancer
- for the maintenance treatment of adult patients with a deleterious BRCA mutation (germline and/or somatic)-associated recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in a complete or partial response to platinum-based chemotherapy. (1.1 )
Prostate cancer
- for the treatment of adult patients with a deleterious BRCA mutation (germline and/or somatic)-associated metastatic castration-resistant prostate cancer (mCRPC) who have been treated with androgen receptor-directed therapy. Select patients for therapy based on an FDA-approved companion diagnostic for RUBRACA. (1.2 , 2.1 )
Maintenance Treatment of BRCA -mutated Recurrent Ovarian Cancer
RUBRACA is indicated for the maintenance treatment of adult patients with a deleterious BRCA mutation (germline and/or somatic)-associated recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in a complete or partial response to platinum-based chemotherapy.
BRCA -mutated Metastatic Castration-Resistant Prostate Cancer
RUBRACA is indicated for the treatment of adult patients with a deleterious BRCA mutation (germline and/or somatic)-associated metastatic castration-resistant prostate cancer (mCRPC) who have been treated with androgen receptor-directed therapy. Select patients for therapy based on an FDA-approved companion diagnostic for RUBRACA [see Dosage and Administration (2.1 )].
DOSAGE AND ADMINISTRATION
- Recommended dose is 600 mg orally twice daily with or without food. (2.2 )
- Continue treatment until disease progression or unacceptable toxicity. (2.2 )
- For adverse reactions, consider interruption of treatment or dose reduction. (2.3 )
- Patients receiving RUBRACA for mCRPC should also receive a gonadotropin-releasing hormone (GnRH) analog concurrently or should have had bilateral orchiectomy. (2.2 )
Patient Selection
Maintenance Treatment of BRCA -mutated Recurrent Ovarian Cancer
Select patients for the maintenance treatment of recurrent ovarian cancer with RUBRACA based on the presence of a deleterious BRCA mutation (germline and/or somatic) [see Clinical Studies (14.1 )].
An FDA-approved test for the detection of deleterious germline and/or somatic BRCA mutations is not currently available.
Treatment of BRCA -mutated mCRPC after Androgen Receptor-directed Therapy
Select patients for the treatment of mCRPC with RUBRACA based on the presence of a deleterious BRCA mutation (germline and/or somatic) in plasma specimens [see Clinical Studies (14.2 )]. A negative result from a plasma specimen does not mean that the patient’s tumor is negative for BRCA mutations. Should the plasma specimen have a negative result, consider performing further genomic testing using tumor specimens as clinically indicated.
Information on the FDA-approved tests for the detection of a BRCA mutation in patients with ovarian cancer or with prostate cancer is available at: http://www.fda.gov/CompanionDiagnostics.
Recommended Dose
The recommended dose of Rubraca is 600 mg (two 300 mg tablets) taken orally twice daily with or without food, for a total daily dose of 1,200 mg. (2.2 )
Continue treatment until disease progression or unacceptable toxicity. (2.2 )
For adverse reactions, consider interruption of treatment or dose reduction. (2.3 )
Patients receiving RUBRACA for mCRPC should also receive a gonadotropin-releasing hormone (GnRH) analog concurrently or should have had bilateral orchiectomy. (2.2 )
Dose Modifications for Adverse Reactions
To manage adverse reactions, consider interruption of treatment or dose reduction. Recommended Rubraca dose modifications for adverse reactions are indicated in Table 1 .
| Dose Reduction | Dose |
| Starting Dose | 600 mg twice daily (two 300 mg tablets) |
| First Dose Reduction | 500 mg twice daily (two 250 mg tablets) |
| Second Dose Reduction | 400 mg twice daily (two 200 mg tablets) |
| Third Dose Reduction | 300 mg twice daily (one 300 mg tablet) |
DOSAGE FORMS AND STRENGTHS
- Tablets (200 mg): blue, round, immediate-release, film-coated, debossed with “C2”.
- Tablets (250 mg): white, diamond, immediate-release, film-coated, debossed with “C25”.
- Tablets (300 mg): yellow, oval, immediate-release, film-coated, debossed with “C3”.
USE IN SPECIFIC POPULATIONS
- Lactation: Advise women not to breastfeed. (8.2 )
Pregnancy
Risk Summary
Based on findings from animal studies and its mechanism of action, Rubraca can cause fetal harm when administered to pregnant women. There are no available data in pregnant women to inform the drug-associated risk. In an animal reproduction study, administration of rucaparib to pregnant rats during organogenesis resulted in embryo-fetal death at maternal exposures that were 0.04 times the AUC 0-24h in patients receiving the recommended dose of 600 mg twice daily [see Data] . Apprise pregnant women of the potential risk to a fetus.
The background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
In a dose range-finding embryo-fetal development study, pregnant rats received oral doses of 50, 150, 500, or 1000 mg/kg/day of rucaparib during the period of organogenesis. Post-implantation loss (100% early resorptions) was observed in all animals at doses greater than or equal to 50 mg/kg/day (with maternal systemic exposures approximately 0.04 times the human exposure at the recommended dose based on AUC 0-24h ).
Lactation
Risk Summary
There is no information regarding the presence of rucaparib in human milk, or on its effects on milk production or the breast-fed child. Because of the potential for serious adverse reactions in breast-fed children from Rubraca, advise lactating women not to breastfeed during treatment with Rubraca and for 2 weeks following the last dose.
Females and Males of Reproductive Potential
Rubraca can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1 )] .
Pregnancy Testing
Pregnancy testing is recommended for females of reproductive potential prior to initiating Rubraca.
Contraception
Females
Advise females of reproductive potential to use effective contraception during treatment and for 6 months following the last dose of Rubraca [see Use in Specific Populations (8.1 )] .
Males
Based on findings in genetic toxicity and animal reproduction studies, advise male patients with female partners of reproductive potential or who are pregnant to use effective methods of contraception during treatment and for 3 months following last dose of Rubraca. Advise male patients not to donate sperm during therapy and for 3 months following the last dose of Rubraca [see Use in Specific Populations (8.1 ) and Nonclinical Toxicology (13.1 )].
Pediatric Use
The safety and effectiveness of Rubraca in pediatric patients have not been established.
Geriatric Use
Of the 937 patients with ovarian cancer who received Rubraca in clinical trials including ARIEL3, 41% were age 65 or older and 10% were 75 years or older. No major differences in safety were observed between younger and older patients with ovarian cancer.
Of the 209 patients with mCRPC who received Rubraca in TRITON2, 77% were age 65 or older and 33% were 75 years or older. No major differences in safety were observed between younger and older patients with mCRPC.
Renal Impairment
No dosage modification is recommended for patients with mild to moderate renal impairment (creatinine clearance [CLcr] between 30 and 89 mL/min, as estimated by the Cockcroft-Gault method) [see Clinical Pharmacology (12.3 )]. Rubraca has not been studied in patients with CLcr < 30 mL/min or patients on dialysis.
Hepatic Impairment
No dosage modification is recommended for patients with mild to moderate hepatic impairment (total bilirubin ≤ 3 x upper limit of normal [ULN] or AST > ULN) [see Clinical Pharmacology (12.3 )] . Rubraca has not been studied in patients with severe hepatic impairment (total bilirubin > 3 x ULN and any AST).
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
- Myelodysplastic Syndrome/Acute Myeloid Leukemia (MDS/AML): MDS/AML occurred in patients exposed to RUBRACA, and some cases were fatal. Monitor patients for hematological toxicity at baseline and monthly thereafter. Interrupt or reduce the dose based on severity of reaction. Discontinue if MDS/AML is confirmed. (2.3 , 5.1 )
- Embryo-Fetal Toxicity: RUBRACA can cause fetal harm. Advise of the potential risk to a fetus and to use effective contraception. (5.2 , 8.1 , 8.3 )
Myelodysplastic Syndrome/Acute Myeloid Leukemia
Myelodysplastic Syndrome (MDS)/Acute Myeloid Leukemia (AML) occur in patients treated with RUBRACA, and are potentially fatal adverse reactions. In 2141 treated patients with ovarian and prostate cancer [see Adverse Reactions (6.1 )], MDS/AML occurred in 34 patients (1.6%), including those in long term follow-up. Of these, 14 occurred during treatment or during the 28-day safety follow-up (0.7%). The duration of RUBRACA treatment prior to the diagnosis of MDS/AML ranged from < 2 months to approximately 72 months. The cases were typical of secondary MDS/cancer therapy-related AML; in all cases, patients had received previous platinum-containing chemotherapy regimens and/or other DNA damaging agents.
In ARIEL3, of patients with a germline and/or somatic BRCA mutation treated with RUBRACA, MDS/AML occurred in 9 out of 129 (7%) patients treated with RUBRACA and 4 out of 66 (6%) patients treated with placebo. The duration of therapy with RUBRACA in patients who developed secondary MDS/cancer therapy-related AML varied from 1.2 to 4.7 years.
In TRITON3, MDS/AML occurred in 2 out of 201 patients (1%) with a BRCA mutation treated with RUBRACA. The duration of therapy with RUBRACA in patients who developed secondary MDS/cancer therapy-related AML varied from 1.4 to 2.3 years.
Do not start Rubraca until patients have recovered from hematological toxicity caused by previous chemotherapy (≤ Grade 1). Monitor complete blood counts for cytopenia at baseline and monthly thereafter for clinically significant changes during treatment. For prolonged hematological toxicities (> 4 weeks), interrupt Rubraca or reduce dose according to Table 1 [see Dosage and Administration (2.3 )] and monitor blood counts weekly until recovery. If the levels have not recovered to Grade 1 or less after 4 weeks or if MDS/AML is suspected, refer the patient to a hematologist for further investigations, including bone marrow analysis and blood sample for cytogenetics. If MDS/AML is confirmed, discontinue Rubraca.
Embryo-Fetal Toxicity
Rubraca can cause fetal harm when administered to a pregnant woman based on its mechanism of action and findings from animal studies. In an animal reproduction study, administration of rucaparib to pregnant rats during the period of organogenesis resulted in embryo-fetal death at exposures that were 0.04 times the AUC 0-24h in patients receiving the recommended human dose of 600 mg twice daily. Apprise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment and for 6 months following the last dose of Rubraca [see Use in Specific Populations (8.1 , 8.3 ) and Clinical Pharmacology (12.1 )].
Based on findings from genetic toxicity and animal reproduction studies, advise male patients with female partners of reproductive potential or who are pregnant to use effective contraception during treatment and for 3 months following the last dose of Rubraca [see Use in Specific Populations (8.1 , 8.3 )].
ADVERSE REACTIONS
The following serious adverse reactions are discussed elsewhere in the labeling:
- Myelodysplastic Syndrome/Acute Myeloid Leukemia [see Warnings and Precautions (5.1 )].
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The pooled safety population for patients with ovarian and prostate cancer in the WARNINGS AND PRECAUTIONS section reflect exposure to RUBRACA at 600 mg orally twice daily in 2141 patients treated on clinical trials including ARIEL3, TRITON2, and TRITON3. In ARIEL3 among the patients who received RUBRACA, 57% of patients were exposed for 6 months or longer and 33% were exposed for greater than one year. In TRITON3 among the patients with BRCA-mutated mCRPC who received RUBRACA, 66% were exposed for 6 months or longer and 34% were exposed for greater than one year.
In the pooled safety population for patients with ovarian cancer, the most common adverse reactions in ≥ 10% of patients were nausea (68%), asthenia/fatigue (65%), anemia/hemoglobin decreased (45%), AST/ALT increased (39%), vomiting (36%), diarrhea (29%), decreased appetite (27%), thrombocytopenia/platelet count decreased (25%), dysgeusia (24%), neutropenia/neutrophil count decreased (21%), blood creatinine increased (17%), dyspnea (16%), dizziness (14%), dyspepsia (11%), photosensitivity reaction (10%), and leukopenia/white blood cell count decreased (10%).
In the pooled safety population for patients with BRCA -mutated mCRPC (N=373) from TRITON2 and TRITON3, the most common adverse reactions in ≥ 10% of patients were fatigue/asthenia (61%), musculoskeletal pain (52%), nausea (52%), anemia/decreased hemoglobin (49%), decreased appetite (35%), increased ALT/AST (31%), constipation (30%), diarrhea (27%), vomiting (26%), thrombocytopenia/decreased platelet count (21%), dyspnea (20%), increased blood creatinine (19%), edema (19%), dizziness (19%), weight decreased (16%), abdominal pain (15%), dysgeusia (15%), rash (13%), neuropathy peripheral (13%), urinary tract infection (13%), cough (12%), headache (12%), hemorrhage (12%), neutropenia/decreased neutrophil count (12%), and photosensitivity reaction (10%).
Maintenance Treatment of BRCA -mutated Recurrent Ovarian Cancer
The safety of RUBRACA for the maintenance treatment of patients with BRCA -mutated recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer was investigated in ARIEL3, a randomized (2:1), double-blind, placebo-controlled study in which 195 patients with a deleterious BRC A mutation received either RUBRACA 600 mg orally twice daily (n=129) or placebo (n=66) until disease progression or unacceptable toxicity. The median duration of study treatment was 13.6 months (range: < 1 month to 39 months) for patients who received RUBRACA and 5.5 months for patients who received placebo.
Dose interruptions due to an adverse reaction of any grade occurred in 67% of patients receiving RUBRACA and 14% of those receiving placebo; dose reductions due to an adverse reaction occurred in 57% of RUBRACA patients and 6% of placebo patients. The most frequent adverse reactions leading to dose interruption or dose reduction of RUBRACA were thrombocytopenia (25%), anemia (19%), ALT/AST increased (16%), fatigue/asthenia (14%), and nausea (10%). Discontinuation due to adverse reactions occurred in 15% of RUBRACA patients and 5% of placebo patients. Specific adverse reactions that most frequently led to discontinuation in patients treated with RUBRACA were thrombocytopenia (4%), nausea (3%), and anemia (2%). Table 2 describes the adverse reactions occurring in ≥20% of patients; while Table 3 describes the laboratory abnormalities occurring in ≥25% of patients occurring in ARIEL3.
a National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE version 4.03) | ||||
b Consists of grouped related terms that reflect the medical concept of the adverse reaction | ||||
| Adverse reactions | RUBRACA N=129 | Placebo N=66 | ||
| Grades 1-4 a % | Grades 3-4 % | Grades 1-4 a % | Grades 3-4 % | |
| Gastrointestinal Disorders | ||||
| Nausea | 79 | 2 | 29 | 0 |
| Abdominal pain/distention b | 48 | 3 | 49 | 2 |
| Constipation | 39 | 4 | 36 | 2 |
| Vomiting | 37 | 4 | 14 | 0 |
| Diarrhea | 34 | 2 | 18 | 0 |
| Stomatitis b | 28 | 0.8 | 12 | 0 |
| General Disorders and Administration Site Conditions | ||||
| Fatigue/asthenia | 74 | 9 | 52 | 5 |
| Skin and Subcutaneous Tissue Disorders | ||||
| Rash b | 45 | 0 | 23 | 0 |
| Nervous System Disorders | ||||
| Dysgeusia | 33 | 0 | 6 | 0 |
| Headache | 22 | 0 | 15 | 2 |
| Investigations | ||||
| ALT/AST elevation | 33 | 16 | 3 | 0 |
| Blood and Lymphatic System Disorders | ||||
| Anemia | 41 | 26 | 6 | 0 |
| Thrombocytopenia | 35 | 6 | 3 | 0 |
| Neutropenia | 22 | 8 | 6 | 0 |
| Respiratory, Thoracic, and Mediastinal Disorders | ||||
| Nasopharyngitis/Upper respiratory tract infection b | 29 | 0 | 20 | 2 |
| Metabolism and Nutrition Disorders | ||||
| Decreased appetite | 23 | 2 | 14 | 0 |
Adverse reactions occurring < 20% of patients treated with RUBRACA include insomnia (19%), dyspnea (17%), dizziness (15%), pyrexia (15%), dyspepsia (12%), peripheral edema (12%), and depression (11%).
Table 3. Laboratory Abnormalities in ≥ 25% of Patients with BRCA-mutated Ovarian Cancer in ARIEL3
a Patients were allowed to enter clinical studies with laboratory values of CTCAE Grade 1. | ||||
b NCI CTCAE version 4.03. | ||||
| Laboratory Parameter a | RUBRACA N=129 | Placebo N=66 | ||
| Grades 1-4 b % | Grades 3-4 % | Grades 1-4 b % | Grades 3-4 % | |
| Chemistry | ||||
| Increase in creatinine | 96 | 0 | 89 | 0 |
| Increase in ALT | 67 | 11 | 6 | 0 |
| Increase in AST | 59 | 1 | 6 | 0 |
| Increase in cholesterol | 39 | 3 | 20 | 0 |
| Increase in Alkaline Phosphatase | 39 | 0 | 3 | 0 |
| Hematology | ||||
| Decrease in hemoglobin | 61 | 18 | 14 | 2 |
| Decrease in platelets | 47 | 2 | 8 | 0 |
| Decrease in leukocytes | 39 | 3 | 23 | 0 |
| Decrease in neutrophils | 38 | 6 | 18 | 2 |
| Decrease in lymphocytes | 33 | 7 | 14 | 2 |
Treatment of BRCA-mutated mCRPC after Androgen Receptor-directed Therapy
TRITON3
The safety of RUBRACA monotherapy was evaluated in patients with BRCA-mutated mCRPC who had progressed following prior treatment with androgen receptor-directed therapy and who had not received treatment with taxane-based chemotherapy in the castration-resistant setting. [see Clinical Studies (14.2 )]. TRITON3 was a randomized, open-label, multi-center study, in which 298 patients with a deleterious BRCA mutation received either RUBRACA 600 mg twice daily (n=201) or physicians choice (n=97) of abiraterone acetate or enzalutamide, or docetaxel until disease progression, unacceptable toxicity, or up to 10 cycles for docetaxel. Among the patients who received RUBRACA, 66% were exposed for 6 months or longer and 34% were exposed for greater than one year.
Serious adverse reactions occurred in 27% of patients receiving RUBRACA. Serious adverse reactions in ≥2% of patients included anemia (4%), pneumonia (4%), urinary tract infection (3%), acute kidney injury (3%), myocardial ischemia/infarction (2%), and pulmonary embolism (2%). Fatal adverse reactions occurred in 1.5% of patients treated with RUBRACA, including cardiac failure, myocardial ischemia, and sepsis (1 patient each).
Permanent discontinuation due to adverse reactions occurred in 14% of patients treated with RUBRACA. Adverse reactions which resulted in permanent discontinuation of RUBRACA in ≥2% of patients included anemia (6%), fatigue/asthenia (3%), thrombocytopenia/platelet count decreased (3%), and nausea (2%).
Dosage interruptions of RUBRACA due to adverse reactions occurred in 53% of patients. Adverse reactions which required dosage interruption in ≥5% of patients included anemia (24%), fatigue/asthenia (10%), nausea (8%), and increase in ALT (5%).
Dose reductions of RUBRACA due to an adverse reaction occurred in 36% of patients. Adverse reactions which required dose reduction in ≥5% of patients included anemia (16%) and fatigue/asthenia (8%).
The most common adverse reactions (≥10%) in patients who received RUBRACA were fatigue/asthenia, musculoskeletal pain, nausea, decreased appetite, diarrhea, constipation, vomiting, dyspnea, dysgeusia, edema, abdominal pain, dizziness, weight decreased, rash, headache, peripheral neuropathy, photosensitivity reaction, and urinary tract infection.
The most common select laboratory abnormalities (≥25%) that worsened from baseline in patients who received RUBRACA were increased ALT, decreased neutrophils, decreased phosphate, decreased hemoglobin, increased AST, increased creatinine, increased glucose, decreased lymphocytes, decreased sodium, decreased platelets, increased calcium.
Tables 4 and 5 summarize the adverse reactions and laboratory abnormalities, respectively, in patients with BRCA- mutated mCRPC in TRITON3.
| a National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE version 5.0) | ||||
| b Includes multiple related terms | ||||
| Adverse Reaction | RUBRACA N=201 | ARPI or Docetaxel N=97 | ||
| Grades 1-4 a (%) | Grades 3-4 (%) | Grades 1-4 a (%) | Grades 3-4 (%) | |
| General disorders and administration site conditions | ||||
| Fatigue/Asthenia | 61 | 7 | 65 | 10 |
| Edema b | 18 | 0 | 20 | 0 |
| Musculoskeletal and connective tissue disorders | ||||
| Musculoskeletal pain b | 53 | 7 | 57 | 8 |
| Gastrointestinal disorders | ||||
| Nausea | 51 | 2 | 22 | 1 |
| Diarrhea | 31 | 2 | 29 | 1 |
| Constipation | 31 | 2 | 17 | 1 |
| Vomiting | 25 | 1 | 10 | 1 |
| Abdominal pain b | 17 | 1 | 10 | 1 |
| Metabolism and nutrition disorders | ||||
| Decreased appetite | 34 | 1 | 20 | 2 |
| Respiratory, thoracic and mediastinal disorders | ||||
| Dyspnea b | 19 | 1 | 13 | 1 |
| Nervous system disorders | ||||
| Dysgeusia b | 18 | 0 | 12 | 0 |
| Dizziness b | 16 | 1 | 9 | 0 |
| Headache b | 12 | 0 | 9 | 0 |
| Peripheral neuropathy b | 12 | 1 | 26 | 1 |
| Investigations | ||||
| Weight decreased | 16 | 1 | 13 | 0 |
| Skin and subcutaneous tissue disorders | ||||
| Rash b | 13 | 1 | 6 | 0 |
| Photosensitivity reaction b | 12 | 1 | 0 | 0 |
| Infections and infestations | ||||
| Urinary tract infection b | 10 | 3 | 4 | 1 |
Clinically relevant adverse reactions occurring in <10% of patients treated with RUBRACA include cough, hemorrhage, fall, hypertension, dyspepsia, pruritus, pyrexia, stomatitis, venous thromboembolism, arrhythmia, hypersensitivity, and febrile neutropenia.
| a Denominator for each laboratory parameter is based on the number of patients with a baseline and post-treatment laboratory value available for 192 to 201 patients in the RUBRACA Arm and 94 to 97 patients in the ARPI or Docetaxel Arm. | ||||
| b NCI CTCAE version 5.0; decrease in phosphate is graded using NCI CTACE Version 4.03 | ||||
| Laboratory Parameter | Rubraca N=201 a | ARPI or Docetaxel N=97 a | ||
| Grades 1-4 b (%) | Grades 3-4 (%) | Grades 1-4 b (%) | Grades 3-4 (%) | |
| Chemistry | ||||
| Increase in ALT | 68 | 4 | 8 | 0 |
| Decrease in phosphate | 64 | 8 | 45 | 5 |
| Increase in AST | 59 | 2 | 7 | 0 |
| Increase in creatinine | 56 | 1 | 16 | 0 |
| Increase in glucose | 45 | 6 | 38 | 3 |
| Decrease in sodium | 35 | 5 | 21 | 4 |
| Increase in calcium | 29 | 3 | 19 | 2 |
| Hematology | ||||
| Decrease in neutrophils | 67 | 9 | 24 | 10 |
| Decrease in hemoglobin | 60 | 23 | 31 | 2 |
| Decrease in lymphocytes | 43 | 14 | 45 | 17 |
| Decrease in platelets | 34 | 6 | 4 | 0 |
TRITON2
The safety of RUBRACA 600 mg twice daily was evaluated in a single arm trial (TRITON2) [see Clinical Studies (14.2)]. TRITON2 enrolled 209 patients with HRR-mutated mCRPC, including 115 with BRCA-mutated mCRPC. Among the patients with BRCA -mutated mCRPC, the median duration of RUBRACA treatment was 6.5 months (range 0.5 to 26.7).
Fatal adverse reactions occurred in 1.7% of patients who received RUBRACA, including acute respiratory distress syndrome and pneumonia (1 patient each).
Permanent discontinuation of RUBRACA due to an adverse reaction occurred in 8% of patients.
Dosage interruption of RUBRACA due to an adverse reaction occurred in 57% of patients.
Dose reductions of RUBRACA occurred in 41% of patients.
Tables 6 and 7 summarize the adverse reactions and laboratory abnormalities, respectively, in patients with BRCA -mutated mCRPC in TRITON2.
| a NCI CTCAE version 4.03. | ||
| b Includes platelet count decreased | ||
| c Includes blister, blood blister, dermatitis, dermatitis contact, eczema, genital rash, palmar-plantar erythrodysaesthesia syndrome, photosensitivity reaction, psoriasis, rash, rash maculo-papular, rash pruritic, skin exfoliation, skin lesion, urticaria | ||
| Adverse Reaction | Rubraca N = 115 | |
| Grades 1-4 a (%) | Grades 3-4 (%) | |
| General disorders and administration site conditions | ||
| Asthenia/Fatigue | 62 | 9 |
| Gastrointestinal disorders | ||
| Nausea | 52 | 3 |
| Constipation | 27 | 1 |
| Vomiting | 22 | 1 |
| Diarrhea | 20 | 0 |
| Blood and lymphatic system disorders | ||
| Anemia | 43 | 25 |
| Thrombocytopenia b | 25 | 10 |
| Metabolism and nutrition disorders | ||
| Decreased appetite | 28 | 2 |
| Skin and subcutaneous tissue disorders | ||
| Rash c | 27 | 2 |
Other clinically relevant adverse reactions that occurred in less than 20% of patients included dyspnea, dizziness, bleeding, urinary tract infection, dysgeusia, dyspepsia, hypersensitivity (including flushing, asthma, choking sensation, periorbital swelling, swelling face, and wheezing), pneumonia, sepsis, ischemic cardiovascular events, renal failure, venous thromboembolism, and stomatitis.
| a Denominator for each laboratory parameter is based on the number of patients with a baseline and post-treatment laboratory value available for 111 to 115 patients. | ||
| b NCI CTCAE version 5.0; decrease in phosphate is graded using NCI CTACE Version 4.03 | ||
| c Grade 3-4 ALT or AST elevation led to drug interruption in 4 patients, of which 1 had dose reduction upon rechallenge. | ||
| Laboratory Parameter | Rubraca N = 115 a | |
| Grades 1-4 b (%) | Grades 3-4 (%) | |
| Clinical Chemistry | ||
| Increase in ALT c | 69 | 5 |
| Decrease in phosphate | 68 | 15 |
| Increase in alkaline phosphatase | 44 | 2 |
| Increase in creatinine | 43 | 2 |
| Increase in triglycerides | 42 | 5 |
| Decrease in sodium | 38 | 3 |
| Hematologic | ||
| Decrease in leukocytes | 69 | 5 |
| Decrease in absolute neutrophil count | 62 | 5 |
DRUG INTERACTIONS
Effect of Rubraca on Other Drugs
Certain CYP1A2, CYP3A, CYP2C9, or CYP2C19 Substrates
Concomitant administration of Rubraca with CYP1A2, CYP3A, CYP2C9, or CYP2C19 substrates can increase the systemic exposure of these substrates [see Clinical Pharmacology (12.3 )] , which may increase the frequency or severity of adverse reactions of these substrates. If concomitant administration is unavoidable between Rubraca and substrates of these enzymes where minimal concentration changes may lead to serious adverse reactions, decrease the substrate dosage in accordance with the approved prescribing information.
If concomitant administration with warfarin (a CYP2C9 substrate) cannot be avoided, consider increasing the frequency of international normalized ratio (INR) monitoring.
DESCRIPTION
Rucaparib is an inhibitor of the mammalian polyadenosine 5'-diphosphoribose polymerase (PARP) enzyme. The chemical name is 8-fluoro-2-{4-[(methylamino)methyl]phenyl}-1,3,4,5-tetrahydro-6H-azepino[5,4,3-cd]indol-6-one ((1S,4R)-7,7-dimethyl-2-oxobicyclo[2.2.1]hept-1-yl)methanesulfonic acid salt. The chemical formula of rucaparib camsylate is C 19 H 18 FN 3 O•C 10 H 16 O 4 S and the relative molecular mass is 555.67 Daltons.
The chemical structure of rucaparib camsylate is shown below:
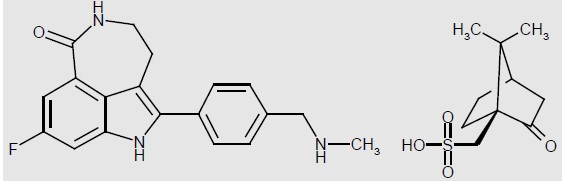
Rucaparib camsylate is a white to pale yellow powder; formulated into a tablet for oral use. Rucaparib shows pH-independent low solubility of approximately 1 mg/mL across the physiological pH range.
Rubraca (rucaparib) tablets contain rucaparib camsylate as the active ingredient. Each 200 mg tablet contains 344 mg rucaparib camsylate equivalent to 200 mg rucaparib free base. Each 250 mg tablet contains 430 mg rucaparib camsylate equivalent to 250 mg rucaparib free base. Each 300 mg tablet contains 516 mg rucaparib camsylate equivalent to 300 mg rucaparib free base.
The inactive ingredients in Rubraca tablets include: microcrystalline cellulose, sodium starch glycolate, colloidal silicon dioxide, and magnesium stearate. The cosmetic blue film coating for 200 mg tablets, cosmetic white film coating for 250 mg tablets, and cosmetic yellow film coating for 300 mg tablets is Opadry II containing polyvinyl alcohol, titanium dioxide, polyethylene glycol/macrogol, and talc. The coating is colorized as blue using brilliant blue aluminum lake and indigo carmine aluminum lake, or yellow using yellow iron oxide.
CLINICAL PHARMACOLOGY
Mechanism of Action
Rucaparib is an inhibitor of poly (ADP-ribose) polymerase (PARP) enzymes, including PARP-1, PARP-2, and PARP-3, which play a role in DNA repair. In vitro studies have shown that rucaparib-induced cytotoxicity may involve inhibition of PARP enzymatic activity and increased formation of PARP-DNA complexes resulting in DNA damage, apoptosis, and cancer cell death. Increased rucaparib-induced cytotoxicity and anti-tumor activity was observed in tumor cell lines with deficiencies in BRCA1/2 and other DNA repair genes. Rucaparib has been shown to decrease tumor growth in mouse xenograft models of human cancer with or without deficiencies in BRCA .
Pharmacodynamics
The exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of rucaparib has not fully been characterized.
Cardiac Electrophysiology
A positive concentration-QTc relationship was observed in patients who were administered continuous dosages of Rubraca ranging from 40 mg once daily (0.03 times the approved recommended dosage) to 840 mg twice daily (1.4 times the approved recommended dosage). The mean (90% confidence interval [CI]) QTcF increase from baseline at steady state of Rubraca 600 mg twice daily was 14.9 msec (11.1, 18.7 msec).
Pharmacokinetics
The AUC and C max of rucaparib demonstrated linear pharmacokinetics over a dose range from 240 mg to 840 mg twice daily (0.4 times to 1.4 times the approved recommended dosage). The mean (coefficient of variation [CV]) steady-state rucaparib C max is 1,940 ng/mL (54%) and AUC 0-12h is 16,900 h×ng/mL (54%) at the approved recommended dosage. The mean AUC accumulation ratio is 3.5 to 6.2 fold.
Absorption
The median T max (min, max) at the steady state is 1.9 hours (0, 5.98) at the approved recommended dosage. The mean (min, max) absolute bioavailability is 36% (30%, 45%).
Effect of Food
Following a high-fat meal (approximately 800-1000 calories, including approximately 250 calories from carbohydrates, approximately 500-600 calories from fat, approximately 150 calories from protein), the C max was increased by 20%, AUC 0-24h was increased by 38%, and the T max was delayed by 2.5 hours, as compared to fasted conditions [see Dosage and Administration (2.2 )] .
Distribution
The mean apparent volume of distribution is 2300 L (21%).
Rucaparib is 70% bound to human plasma proteins in vitro. Rucaparib preferentially distributed to red blood cells with a blood-to-plasma concentration ratio of 1.8.
Elimination
The mean apparent total clearance at steady state is 44.2 L/h (45%) and the mean terminal elimination half-life is 26 (39%) hours.
Metabolism
In vitro, rucaparib is primarily metabolized by CYP2D6 and to a lesser extent by CYP1A2 and CYP3A4. In addition to CYP-based oxidation, rucaparib also undergoes N-demethylation, N-methylation, and glucuronidation.
Excretion
Following a single oral dose of radiolabeled rucaparib, unchanged rucaparib accounted for 64% of the radioactivity. Rucaparib accounted for 45% and 95% of radioactivity in urine and feces, respectively.
Specific Populations
Age (20 to 86 years), race (White, Black, and Asian), sex, body weight (41 to 171 kg), mild to moderate renal impairment (CLcr ≥ 30 mL/min), mild hepatic impairment (total bilirubin < ULN and AST > ULN or total bilirubin 1 to 1.5 x ULN and any AST), and CYP2D6 or CYP1A2 genotype polymorphisms did not have a clinically meaningful effect on the pharmacokinetics of rucaparib. The effect of severe renal impairment (CLcr 15 to 29 mL/min), end-stage renal disease (CLcr < 15 mL/min), or severe hepatic impairment (total bilirubin > 3 x ULN and any AST) has not been studied.
Hepatic Impairment
Moderate hepatic impairment (total bilirubin > 1.5 to 3 x ULN and any AST) increased rucaparib AUC by 45%, but had no effect on C max compared to patients with normal hepatic function.
Drug Interaction Studies
Clinical Studies and Model-Informed Approaches
Effect of Other Drugs on Rucaparib
Concomitant administration of Rubraca with a proton pump inhibitor had no clinically meaningful effect on steady-state concentrations of rucaparib.
Effect of Rucaparib on Other Drugs
Concomitant administration of Rubraca with rosuvastatin (BCRP substrate) had no clinically meaningful effect on the concentrations of rosuvastatin.
Coadministration of Rubraca with the following substrates increased the C max of each coadministered substrate by ≤ 1.1-fold and increased the AUC of each substrate as follows:
- Caffeine (CYP1A2): by 2.6-fold
- Midazolam (CYP3A4): by 1.4-fold
- Warfarin (CYP2C9): by 1.5-fold
- Omeprazole (CYP2C19): by 1.6-fold
- Digoxin (P-glycoprotein): by 1.2-fold
Concomitant administration of Rubraca with an oral contraceptive containing ethinylestradiol and levonorgestrel (CYP3A substrates): increased ethinylestradiol AUC by 1.4-fold and levonorgestrel AUC by 1.6-fold, but did not have a clinically meaningful effect on their C max .
In Vitro Studies
Cytochrome P450 (CYP) Enzymes : Rucaparib inhibited CYP2C8 and CYP2D6 and induced CYP1A2.
UDP-glucuronosyltransferase (UGT) Enzymes : Rucaparib inhibited UGT1A1.
Transporter Systems : Rucaparib is a substrate of P-gp and BCRP. Rucaparib is not a substrate of OATP1B1, OATP1B3, OAT1, OAT3, or OCT2.
Rucaparib inhibited OATP1B1, OATP1B3, OAT1, OAT3, MATE1, MATE2-K, OCT1, OCT2, and MRP4. Rucaparib did not inhibit MRP2, MRP3, or BSEP.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with rucaparib.
Rucaparib was clastogenic in an in vitro chromosomal aberration assay in cultured human lymphocytes. The clastogenic response in mitotically-stimulated cells was anticipated based on the mechanism of action of rucaparib and indicates potential genotoxicity in humans. Rucaparib was not mutagenic in a bacterial reverse mutation (Ames) test.
Fertility studies with rucaparib have not been conducted. In 3-month repeat-dose general toxicology studies, rucaparib had no effects on male and female reproductive organs at doses up to 100 mg/kg/day and 20 mg/kg/day in rats and dogs, respectively. These dose levels resulted in systemic exposures of approximately 0.3 and 0.09 times the human exposure (AUC 0-24h ), respectively, at the recommended dose.
CLINICAL STUDIES
Maintenance Treatment of BRCA -mutated Recurrent Ovarian Cancer
The efficacy of Rubraca was investigated in ARIEL3 (NCT01968213 ), a double-blind, multicenter clinical trial in which 564 patients with recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer who were in response to platinum-based chemotherapy were randomized (2:1) to receive Rubraca tablets 600 mg orally twice daily (n=375) or placebo (n=189). Treatment was continued until disease progression or unacceptable toxicity. All patients had achieved a response (complete or partial) to their most recent platinum-based chemotherapy. Randomization was stratified by best response to last platinum (complete or partial), time to progression following the penultimate platinum therapy (6 to ≤12 months and >12 months), and tumor biomarker status.
Tumor tissue samples were tested using a clinical trial assay (CTA) (N=564) and an investigational Foundation Medicine tissue test (n=518). Of the samples evaluated with both tests, tumor BRCA (t BRCA ) mutant status was confirmed for 99% (177/178) of t BRCA -positive patients determined by the CTA. Blood samples for 94% (186/196) of the t BRCA patients were evaluated using a central blood germline BRCA test. Based on these results, 70% (130/186) of the t BRCA patients had a germline BRCA mutation and 30% (56/186) had a somatic BRCA mutation. The efficacy results are based on the t BRCA (germline or somatic) subgroup.
The major efficacy outcome was investigator-assessed progression-free survival (PFS) evaluated according to Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1 (v1.1). Overall survival (OS) was an additional outcome measure.
Of the 564 enrolled patients, 196 patients (35%) had a t BRCA mutation. Among the patients who had a tBRCA mutation, the median age was 58 years (range: 42 to 81) for patients receiving Rubraca and 59 years (range: 36 to 84) for those on placebo; the majority were White (84%); and 100% had an ECOG performance status of 0 or 1. All patients had received at least two prior platinum-based chemotherapies (range: 2 to 5). A total of 33% of patients were in complete response (CR) to their most recent therapy. The progression-free interval to penultimate platinum was 6-12 months in 41% of patients and >12 months in 59%. Prior bevacizumab therapy was reported for 22% of patients who received Rubraca and 17% of patients who received placebo. Measurable disease was present at baseline in 32% of patients.
ARIEL3 demonstrated a statistically significant improvement in PFS for patients randomized to Rubraca as compared with placebo in patients who had a t BRCA mutation. Results from a blinded independent radiology review were consistent.
Efficacy results for patients with a t BRCA mutation are summarized in Table 8 and Figure 1 .
| a t BRCA includes all patients with a deleterious germline or somatic BRCA mutation, as determined by the CTA. | ||
| Rubraca N=130 | Placebo N=66 | |
| Progression Free Survival | ||
| Number of events, n (%) | 67 (52%) | 56 (85%) |
| Median in months (95% CI) | 16.6 (13.4-22.9) | 5.4 (3.4-6.7) |
| HR (95% CI) | 0.23 (0.16, 0.34) | |
| p-value | < 0.0001 | |
Figure 1 Kaplan-Meier Curves of Progression-Free Survival in ARIEL3 as Assessed by Investigator: t BRCA Group
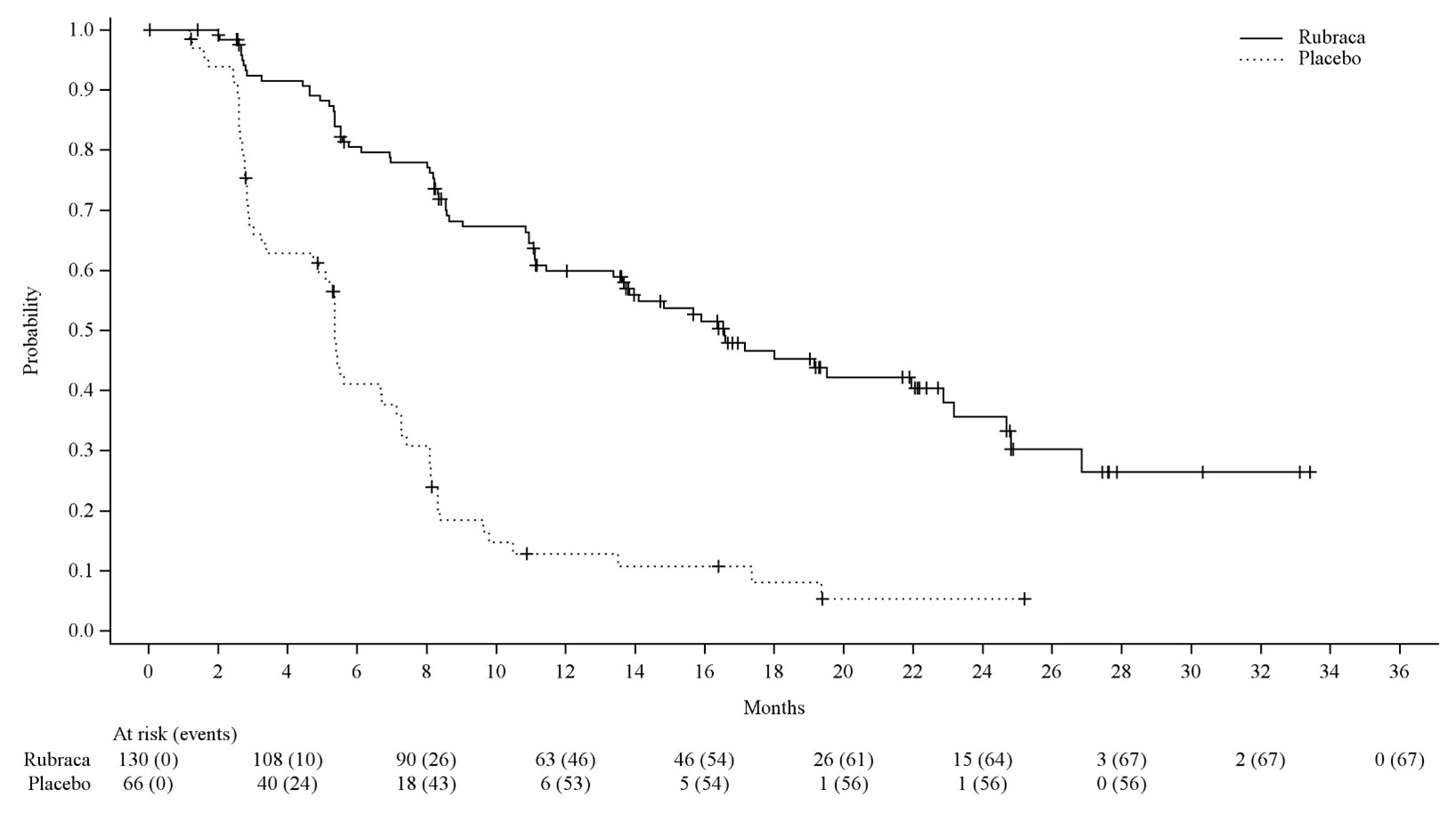
A final OS analysis was conducted after 130 events were observed. Exploratory OS results showed a HR of 0.83 (95% CI: 0.58, 1.19) in the t BRCA subgroup with a median OS of 45.9 months (95% CI: 37.7, 59.6) for patients treated with Rubraca and 47.8 months (95% CI: 43.2, 55.8) for patients on placebo.
BRCA -mutated Metastatic Castration-Resistant Prostate Cancer
TRITON3
The efficacy of RUBRACA was evaluated in TRITON3 (NCT02975934 ), a randomized, open-label, multi-center trial that evaluated RUBRACA compared to an androgen receptor pathway inhibitor (ARPI; enzalutamide or abiraterone acetate) or docetaxel in patients with progressive BRCA or ATM mutated metastatic castrate-resistant prostate cancer (mCRPC). The trial enrolled 405 patients with mCRPC, of whom 302 had BRCA mutations. Patients were required to have progressed on treatment with one ARPI and could not have received prior chemotherapy in the castrate-resistant setting. Prior taxane chemotherapy for castration-sensitive disease was permitted. Patients were required to be surgically or medically castrated with a castrate level of serum testosterone at study entry. BRCA gene mutation status was determined prospectively testing tumor tissue or plasma using Foundation Medicine, Inc. assays or through local testing.
Patients were randomized (2:1) to receive RUBRACA 600 mg twice daily (n=270) or ARPI (enzalutamide or abiraterone acetate; n=60) or docetaxel (n=75). Randomization was stratified by ECOG performance status (0 or 1), hepatic metastases (present or not), and type of mutation ( BRCA 1, BRCA 2, or ATM ). If patients were being treated with androgen deprivation therapy (ADT), therapy was continued throughout the study. Bone-targeted agents were allowed.
Of the 302 patients with BRCA m disease, the median age was 69 years (range: 45 to 92 years); 75% were White, 5% Black, 1.3% Asian, 0.3% American Indian or Alaska Native, 1% multiple races, and 18% unknown; 1.7% were Hispanic/Latino; and baseline ECOG performance status (PS) was 0 (49%) or 1 (51%). Forty percent of patients had bone-only disease; 32% had visceral disease. In the mCSPC setting, 23% of patients had received docetaxel. Among those with BRCA m disease, 40 patients had BRCA 1 mutations, and 262 patients had BRCA 2 mutations.
The major efficacy outcome measure was radiographic progression-free survival (rPFS) as determined by independent radiology review (IRR) using RECIST version 1.1 and Prostate Cancer Clinical Trials Working Group 3 (PCWG3) (bone) criteria. Additional efficacy outcome measures included overall survival (OS) and IRR-assessed objective response rate (ORR).
TRITON3 demonstrated a statistically significant improvement in IRR-assessed rPFS for RUBRACA compared to ARPI or docetaxel in patients with BRCA m and in the overall population. In an exploratory analysis in the subgroup of 103 (25%) patients with ATM mutations, the rPFS hazard ratio was 0.95 (95% CI: 0.59, 1.52) and the OS hazard ratio was 1.21 (95% CI: 0.77, 1.90), indicating that the improvement in the overall population was primarily attributed to the results seen in the subgroup of patients with BRCA m. Of patients with BRCA m who were randomized to receive ARPI or docetaxel (n=101), 52% crossed over to receive RUBRACA after disease progression.
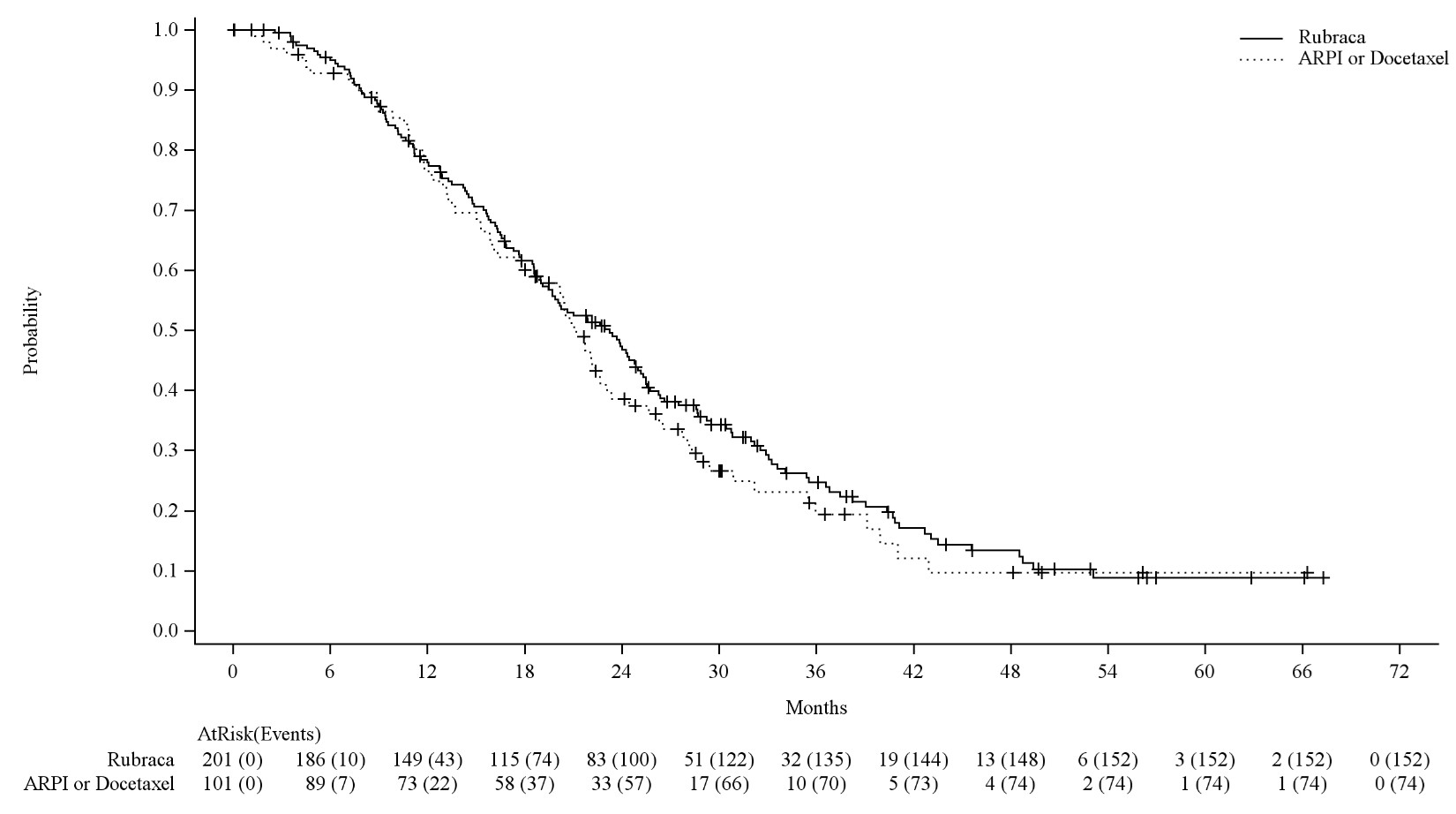
Efficacy results of the BRCA m population in TRITON3 are provided in Table 9, Figure 2 and Figure 3.
| Endpoint | Rubraca N=201 f | ARPI or Docetaxel N=101 f |
| Radiographic Progression Free Survival a | ||
| Number of events, n (%) | 115 (57%) | 67 (66%) |
| Median in months (95% CI) | 11.2 (9.2-13.8) | 6.4 (5.4-8.3) |
| Hazard ratiob (95% CI) | 0.50 (0.36, 0.69) | |
| p-value c | < 0.0001 | |
| Overall Survival d | ||
| Number of events, n (%) | 152 (76%) | 74 (73%) |
| Median in months (95% CI) | 23.2 (19.1, 25.2) | 21.2 (18.0, 23.1) |
| Hazard ratio b (95% CI) | 0.91 (0.68, 1.20) | |
| p-value c | NS | |
| Confirmed Overall Response Rate a,e | ||
| N=82 | N=41 | |
| Overall response rate (95% CI) | 45.1 (34.1, 56.5) | 17.1 (7.2, 32.1) |
| Number of complete responses, n (%) | 10 (12.2) | 2 (4.9) |
| Number of partial responses, n (%) | 27 (32.9) | 5 (12.2) |
| NS=Not significant | ||
| a Based on IRR assessment. | ||
| b Cox proportional hazards model stratified by ECOG Status (0 or 1) and type of mutation ( BRCA 1 or BRCA 2). | ||
| c P-value based on the stratified log-rank test | ||
| d Based on pre-specified final analysis. | ||
| e Based on patients with measurable disease at baseline per IRR assessment. Responses are based on soft tissue and bone lesion assessment. | ||
| f Based on the BRCA mutation status used at the randomization. This excludes one patient with a reported ATM mutation at randomization but found to have a BRCA2 mutation based on the verified source. | ||
Figure 2 Kaplan-Meier Curves of rPFS from the BRCAm Subgroup of the TRITON3 Study (IRR-assessed)
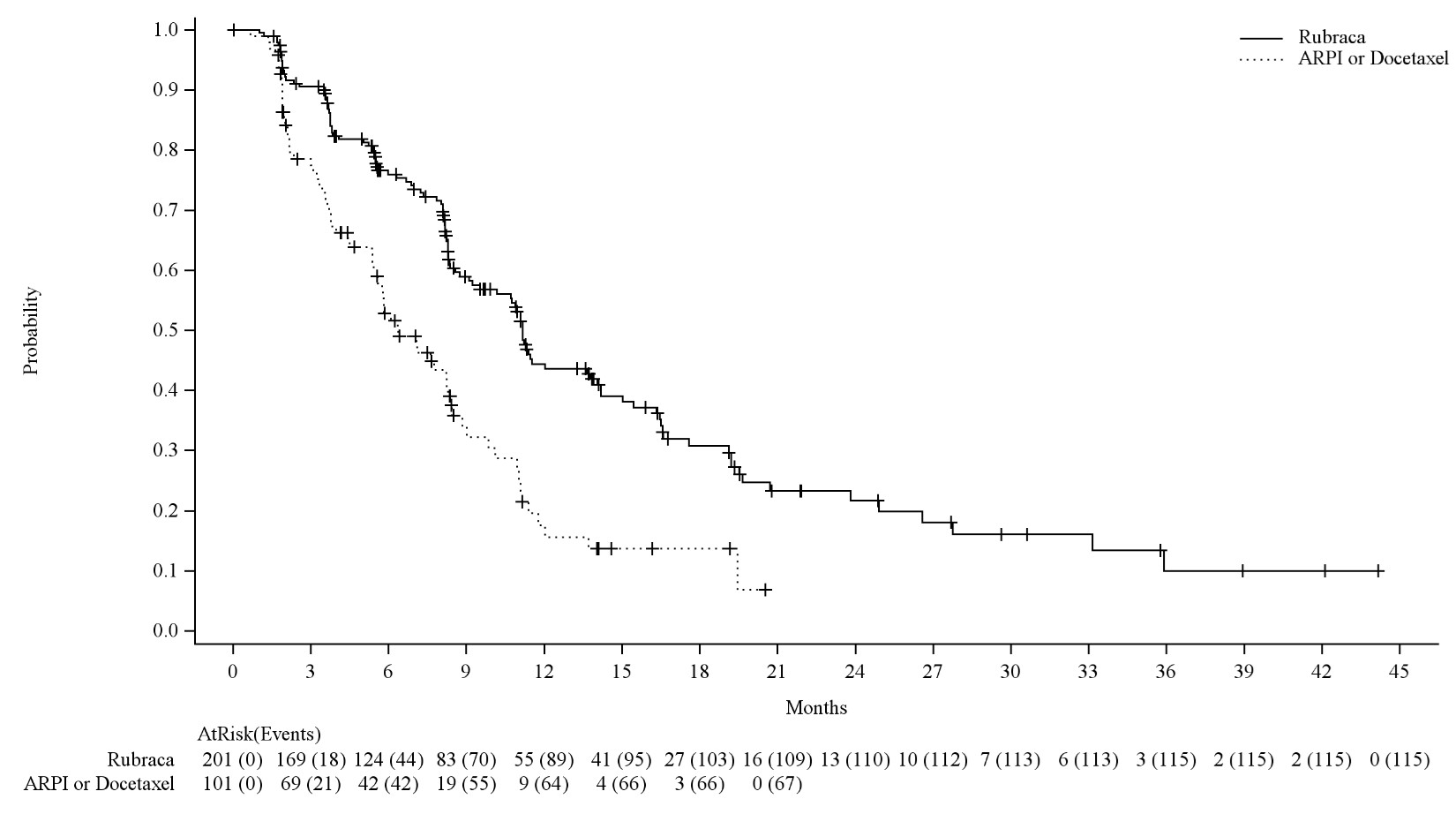
Figure 3 Kaplan-Meier Curves of Final OS from the BRCAm Subgroup of the TRITON3 Study
Exploratory subgroup analysis of rPFS and OS for patients with BRCA1m and BRCA2m (including one patient with a BRCA2 mutation mischaracterized as having an ATM mutation at randomization) were performed. The rPFS and OS hazard ratios were 1.01 (95% CI: 0.48, 2.13) and 0.96 (95% CI: 0.46, 2.00) respectively in the subgroup with BRCA1 mutations (n=40) and 0.46 (95% CI: 0.33, 0.65) and 0.92 (95% CI: 0.68, 1.24) respectively in the subgroup with BRCA2 mutations (n=263).
TRITON2
The efficacy of Rubraca was investigated in TRITON2 (NCT02952534 ), an ongoing multi-center, single arm clinical trial in patients with BRCA -mutated mCRPC who had been treated with androgen receptor-directed therapy and taxane-based chemotherapy. There were 115 patients with either germline or somatic BRCA mutations enrolled in TRITON2, of whom 62 patients had measurable disease at baseline by independent radiology review (IRR). Patients received Rubraca 600 mg orally twice daily until disease progression or unacceptable toxicity. Patients also received concomitant GnRH analog or had prior bilateral orchiectomy. Objective response rate (ORR) and duration of response (DOR) were assessed in patients with measurable disease by blinded IRR and by the investigator according to modified RECIST v1.1/ Prostate Cancer Working Group 3 (PCWG3) criteria.
For the 62 patients with measurable disease at baseline, the median age was 73 years (range 52 to 88); the majority were White (73%) and 10% were Black; and 98% of patients had an ECOG performance status of 0 or 1. All patients had received at least one prior androgen receptor-directed therapy, 34% had received 2 prior androgen receptor-directed therapies and 2% had received 3 prior androgen receptor-directed therapies, and all patients also received prior taxane chemotherapy. Eighteen percent of patients had lung and 21% had liver metastases at baseline. Twenty-four percent had metastases to lymph nodes alone. Forty percent had 10 or more bone lesions at baseline.
All 62 patients had a deleterious somatic or germline BRCA mutation detected from either central plasma (26%), central tissue (32%), or local (42%) testing. Of the 62 patients, 66% had a somatic BRCA mutation, 34% had a germline BRCA mutation, 85% had a BRCA2 mutation, and 15% had a BRCA1 mutation.
The major efficacy outcomes of the study were confirmed ORR by IRR using modified RECIST v1.1/PCWG3 criteria and DOR. Efficacy results of TRITON2 are provided in Table 10. The ORR by IRR was similar in patients with germline versus somatic BRCA mutation.
| NE = not evaluable | |
| a Defined per modified RECIST v1.1 criteria and with no confirmed bone progression per PCWG3. | |
| b The range for the DOR was 1.7-24+ months. Fifteen of the 27 (56%) patients with a confirmed objective response had a DOR of ≥6 months. | |
| Rubraca (N = 62) | |
| Confirmed Objective Response Rate (95% CI) a | 44% (31, 57) |
| Median DOR in months (95% CI) b | NE (6.4, NE) |
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Rubraca is available as 200 mg, 250 mg, and 300 mg tablets.
200 mg Tablets:
- Blue, round, and debossed with “C2” on one side
- Supplied in bottles of 60 tablets (NDC:82154-0783-1)
250 mg Tablets:
- White, diamond, and debossed with “C25” on one side
- Supplied in bottles of 60 tablets (NDC:82154-0784-1)
300 mg Tablets:
- Yellow, oval, and debossed with “C3” on one side
- Supplied in bottles of 60 tablets (NDC:82154-0785-1)
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [ see USP Controlled Room Temperature ].
Mechanism of Action
Rucaparib is an inhibitor of poly (ADP-ribose) polymerase (PARP) enzymes, including PARP-1, PARP-2, and PARP-3, which play a role in DNA repair. In vitro studies have shown that rucaparib-induced cytotoxicity may involve inhibition of PARP enzymatic activity and increased formation of PARP-DNA complexes resulting in DNA damage, apoptosis, and cancer cell death. Increased rucaparib-induced cytotoxicity and anti-tumor activity was observed in tumor cell lines with deficiencies in BRCA1/2 and other DNA repair genes. Rucaparib has been shown to decrease tumor growth in mouse xenograft models of human cancer with or without deficiencies in BRCA .