Get your patient on Pyrazinamide - Pyrazinamide tablet (Pyrazinamide)
Pyrazinamide - Pyrazinamide tablet prescribing information
INDICATIONS AND USAGE:
Pyrazinamide is indicated for the initial treatment of active tuberculosis in adults and children when combined with other antituberculous agents. (The current recommendation of the CDC for drug-susceptible disease is to use a six-month regimen for initial treatment of active tuberculosis, consisting of isoniazid, rifampin and pyrazinamide given for 2 months, followed by isoniazid and rifampin for 4 months. •4 )
(Patients with drug-resistant disease should be treated with regimens individualized to their situation. Pyrazinamide frequently will be an important component of such therapy.)
(In patients with concomitant HIV infection, the physician should be aware of current recommendation of CDC. It is possible these patients may require a longer course of treatment).
It is also indicated after treatment failure with other primary drugs in any form of active tuberculosis.
Pyrazinamide should only be used in conjunction with other effective antituberculous agents.
•See recommendations of Center for Disease Control (CDC) and American Thoracic Society for complete regimen and dosage recommendations. 4
DOSAGE AND ADMINISTRATION:
Pyrazinamide should always be administered with other effective antituberculous drugs. It is administered for the initial 2 months of a 6-month or longer treatment regimen for drug-susceptible patients. Patients who are known or suspected to have drug-resistant disease should be treated with regimens individualized to their situation.
Pyrazinamide frequently will be an important component of such therapy.
Patients with concomitant HIV infection may require longer courses of therapy. Physicians treating such patients should be alert to any revised recommendations from CDC for this group of patients.
Usual dose: Pyrazinamide is administered orally, 15 to 30 mg/kg once daily. Older regimens employed 3 or 4 divided doses daily, but most current recommendations are for once a day. Three grams per day should not be exceeded. The CDC recommendations do not exceed 2 g per day when given as a daily regimen (see table).
Alternatively, a twice weekly dosing regimen (50 to 75 mg/kg twice weekly based on lean body weight) has been developed to promote patient compliance with a regimen on an outpatient basis. In studies evaluating the twice weekly regimen, doses of pyrazinamide in excess of 3 g twice weekly have been administered. This exceeds the recommended maximum 3 g/daily dose. However, an increased incidence of adverse reactions has not been reported.
This table is taken from the CDC-American Thoracic Society joint recommendations. 4
Drug | Daily Dose • | |
Children | Adults | |
Isoniazid | 10 to 20 mg/kg PO or IM | 5 mg/kg PO or IM |
Rifampin | 10 to 20 mg/kg PO | 10 mg/kg PO |
Pyrazinamide | 15 to 30 mg/kg PO | 15 to 30 mg/kg PO |
Streptomycin | 20 to 40 mg/kg IM | 15 mg/kg IM |
Ethambutol |
PO |
PO |
Drug | Maximal Daily Dose in Children and Adults |
Isoniazid | 300 mg |
Rifampin | 600 mg |
Pyrazinamide | 2 g |
Streptomycin | 1 g •• |
Ethambutol |
|
Drug | Twice Weekly Dose | |
Children | Adults | |
Isoniazid | 20 to 40 mg/kg Max. 900 mg | 15 mg/kg Max. 900 mg |
Rifampin | 10 to 20 mg/kg Max. 600 mg | 10 mg/kg Max. 600 mg |
Pyrazinamide | 50 to 70 mg/kg | 50 to 70 mg/kg |
Streptomycin | 25 to 30 mg/kg IM | 25 to 30 mg/kg IM |
Ethambutol | 50 mg/kg | 50 mg/kg |
Definition of abbreviations: PO = perorally; IM = intramuscularly.
CONTRAINDICATIONS:
Pyrazinamide is contraindicated in persons:
- with severe hepatic damage.
- who have shown hypersensitivity to it.
- with acute gout.
ADVERSE REACTIONS:
General:
Fever, porphyria and dysuria have rarely been reported. Gout (see PRECAUTIONS : ).
Gastrointestinal:
The principal adverse effect is a hepatic reaction (see WARNINGS : ). Hepatotoxicity appears to be dose related, and may appear at any time during therapy. GI disturbances including nausea, vomiting and anorexia have also been reported.
Hematologic and Lymphatic:
Thrombocytopenia and sideroblastic anemia with erythroid hyperplasia, vacuolation of erythrocytes and increased serum iron concentration have occurred rarely with this drug. Adverse effects on blood clotting mechanisms have also been rarely reported.
Other:
Mild arthralgia and myalgia have been reported frequently. Hypersensitivity reactions including rashes, urticaria, and pruritis have been reported. Fever, acne, photosensitivity, porphyria, dysuria and interstitial nephritis have been reported rarely.
DESCRIPTION:
Pyrazinamide, the pyrazine analogue of nicotinamide, is an antituberculous agent. It is a white crystalline powder, stable at room temperature, and sparingly soluble in water. Pyrazinamide has the following structural formula:
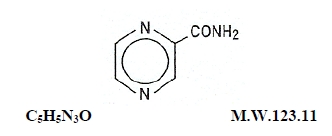
Each pyrazinamide tablet for oral administration contains 500 mg of pyrazinamide and the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, dibasic calcium phosphate (dihydrate), microcrystalline cellulose, and stearic acid.
CLINICAL PHARMACOLOGY:
Pyrazinamide is well absorbed from the GI tract and attains peak plasma concentrations within 2 hours. Plasma concentrations generally range from 30 to 50 mcg/mL with doses of 20 to 25 mg/kg. It is widely distributed in body tissues and fluids including the liver, lungs and cerebrospinal fluid (CSF). The CSF concentration is approximately equal to concurrent steady-state plasma concentrations in patients with inflamed meninges. 1 Pyrazinamide is approximately 10% bound to plasma proteins. 2 The half-life (t 1/2) of pyrazinamide is 9 to 10 hours in patients with normal renal and hepatic function. The plasma half-life may be prolonged in patients with impaired renal or hepatic function. Pyrazinamide is hydrolyzed in the liver to its major active metabolite, pyrazinoic acid. Pyrazinoic acid is hydroxylated to the main excretory product, 5-hydroxypyrazinoic acid. 3
Approximately 70% of an oral dose is excreted in the urine, mainly by glomerular filtration within 24 hours. 3
Pyrazinamide may be bacteriostatic or bactericidal against Mycobacterium tuberculosis depending on the concentration of the drug attained at the site of infection. The mechanism of action is unknown. In vitro and in vivo the drug is active only at a slightly acidic pH.
HOW SUPPLIED:
Pyrazinamide Tablets USP contain pyrazinamide 500 mg. They are supplied as white, round, scored tablets, debossed "VP/012" in containers of 60 tablets, NDC# 61748-012-06, in containers of 90 tablets, NDC# 61748-012-09, in containers of 100 tablets, NDC# 61748-012-01, in containers of 500 tablets, NDC# 61748-012-05, and in hospital unit-dose cartons of 100 tablets (in strips of 10 tablets per strip), NDC# 61748-012-11.
Storage: Store in a well-closed container at controlled room temperature 15°C to 30°C (59°F to 86°F).
Dispense in a well-closed container with a child resistant closure.
Rx only
Distributed by:
Akorn Operating Company LLC
Gurnee, IL 60031 Manufactured by:
MIKART, LLC
Atlanta, GA 30318
Rev. 06/22
Code 590B00