Get your patient on Propranolol Hydrochloride - Propranolol Hydrochloride tablet (Propranolol Hydrochloride)
Propranolol Hydrochloride - Propranolol Hydrochloride tablet prescribing information
INDICATIONS AND USAGE
Hypertension
Propranolol hydrochloride tablets are indicated in the management of hypertension. They may be used alone or used in combination with other antihypertensive agents, particularly a thiazide diuretic. Propranolol hydrochloride tablets are not indicated in the management of hypertensive emergencies.
Angina Pectoris Due to Coronary Atherosclerosis
Propranolol hydrochloride tablets are indicated to decrease angina frequency and increase exercise tolerance in patients with angina pectoris.
Atrial Fibrillation
Propranolol hydrochloride tablets are indicated to control ventricular rate in patients with atrial fibrillation and a rapid ventricular response.
Myocardial Infarction
Propranolol hydrochloride tablets are indicated to reduce cardiovascular mortality in patients who have survived the acute phase of myocardial infarction and are clinically stable.
Migraine
Propranolol hydrochloride tablets are indicated for the prophylaxis of common migraine headache. The efficacy of propranolol in the treatment of a migraine attack that has started has not been established, and propranolol is not indicated for such use.
Essential Tremor
Propranolol hydrochloride tablets are indicated in the management of familial or hereditary essential tremor. Familial or essential tremor consists of involuntary, rhythmic, oscillatory movements, usually limited to the upper limbs. It is absent at rest, but occurs when the limb is held in a fixed posture or position against gravity and during active movement. Propranolol hydrochloride tablets, USP causes a reduction in the tremor amplitude but not in the tremor frequency. Propranolol hydrochloride tablets are not indicated for the treatment of tremor associated with Parkinsonism.
Hypertrophic Subaortic Stenosis
Propranolol hydrochloride tablets improve NYHA functional class in symptomatic patients with hypertrophic subaortic stenosis.
Pheochromocytoma
Propranolol hydrochloride tablets are indicated as an adjunct to alpha-adrenergic blockade to control blood pressure and reduce symptoms of catecholamine-secreting tumors.
DOSAGE AND ADMINISTRATION
General
Because of the variable bioavailability of propranolol, the dose should be individualized based on response.
Hypertension
The usual initial dosage is 40 mg propranolol hydrochloride twice daily, whether used alone or added to a diuretic. Dosage may be increased gradually until adequate blood pressure control is achieved. The usual maintenance dosage is 120 mg to 240 mg per day. In some instances a dosage of 640 mg a day may be required. The time needed for full antihypertensive response to a given dosage is variable and may range from a few days to several weeks.
While twice-daily dosing is effective and can maintain a reduction in blood pressure throughout the day, some patients, especially when lower doses are used, may experience a modest rise in blood pressure toward the end of the 12-hour dosing interval. This can be evaluated by measuring blood pressure near the end of the dosing interval to determine whether satisfactory control is being maintained throughout the day. If control is not adequate, a larger dose, or 3-times-daily therapy may achieve better control.
Angina Pectoris
Total daily doses of 80 mg to 320 mg propranolol hydrochloride when administered orally, twice a day, three times a day, or four times a day, have been shown to increase exercise tolerance and to reduce ischemic changes in the ECG. If treatment is to be discontinued, reduce dosage gradually over a period of several weeks. (See WARNINGS )
Atrial Fibrillation
The recommended dose is 10 mg to 30 mg propranolol hydrochloride three or four times daily before meals and at bedtime.
Myocardial Infarction
In the Beta-Blocker Heart Attack Trial (BHAT), the initial dose was 40 mg t.i.d., with titration after 1- month to 60 mg to 80 mg t.i.d. as tolerated. The recommended daily dosage is 180 mg to 240 mg propranolol hydrochloride per day in divided doses. Although a t.i.d. regimen was used in BHAT and a q.i.d. regimen in the Norwegian Multicenter Trial, there is a reasonable basis for the use of either a t.i.d. or b.i.d. regimen (see PHARMACODYNAMICS AND CLINICAL EFFECTS ). The effectiveness and safety of daily dosages greater than 240 mg for prevention of cardiac mortality have not been established. However, higher dosages may be needed to effectively treat coexisting diseases such as angina or hypertension (see above).
Migraine
The initial dose is 80 mg propranolol hydrochloride daily in divided doses. The usual effective dose range is 160 mg to 240 mg per day. The dosage may be increased gradually to achieve optimum migraine prophylaxis. If a satisfactory response is not obtained within four to six weeks after reaching the maximum dose, propranolol hydrochloride therapy should be discontinued. It may be advisable to withdraw the drug gradually over a period of several weeks.
Essential Tremor
The initial dosage is 40 mg propranolol hydrochloride twice daily. Optimum reduction of essential tremor is usually achieved with a dose of 120 mg per day. Occasionally, it may be necessary to administer 240 mg to 320 mg per day.
Hypertrophic Subaortic Stenosis
The usual dosage is 20 mg to 40 mg propranolol hydrochloride three or four times daily before meals and at bedtime.
Pheochromocytoma
The usual dosage is 60 mg propranolol hydrochloride daily in divided doses for three days prior to surgery as adjunctive therapy to alpha-adrenergic blockade. For the management of inoperable tumors, the usual dosage is 30 mg daily in divided doses as adjunctive therapy to alpha-adrenergic blockade.
CONTRAINDICATIONS
Propranolol is contraindicated in 1) cardiogenic shock; 2) sinus bradycardia and greater than first degree block; 3) bronchial asthma; and 4) in patients with known hypersensitivity to propranolol hydrochloride.
ADVERSE REACTIONS
The following adverse events were observed and have been reported in patients using propranolol.
Cardiovascular: Bradycardia; congestive heart failure; intensification of AV block; hypotension; paresthesia of hands; thrombocytopenic purpura; arterial insufficiency, usually of the Raynaud type.
Central Nervous System: Lightheadedness, mental depression manifested by insomnia, lassitude, weakness, fatigue; catatonia; visual disturbances; hallucinations; vivid dreams; an acute reversible syndrome characterized by disorientation for time and place, short-term memory loss, emotional lability, slightly clouded sensorium, and decreased performance on neuropsychometrics. For immediate-release formulations, fatigue, lethargy, and vivid dreams appear dose-related.
Gastrointestinal: Nausea, vomiting, epigastric distress, abdominal cramping, diarrhea, constipation, mesenteric arterial thrombosis, ischemic colitis.
Allergic: Hypersensitivity reactions, including anaphylactic/anaphylactoid reactions, pharyngitis and agranulocytosis; erythematous rash, fever combined with aching and sore throat; laryngospasm, and respiratory distress.
Respiratory: Bronchospasm.
Hematologic: Agranulocytosis, nonthrombocytopenic purpura, thrombocytopenic purpura.
Autoimmune: Systemic lupus erythematosus (SLE).
Skin and mucous membranes: Stevens-Johnson Syndrome, toxic epidermal necrolysis, dry eyes, exfoliative dermatitis, erythema multiforme, urticaria, alopecia, SLE-like reactions, and psoriasiform rashes. Oculomucocutaneous syndrome involving the skin, serous membranes and conjunctivae reported for a beta-blocker (practolol) have not been associated with propranolol.
Genitourinary: Male impotence; Peyronie’s disease.
Drug Interactions
Interactions with Substrates, Inhibitors or Inducers of Cytochrome P-450 Enzymes
Because propranolol’s metabolism involves multiple pathways in the cytochrome P-450 system (CYP2D6,1A2, 2C19), coadministration with drugs that are metabolized by, or effect the activity (induction or inhibition) of one or more of these pathways may lead to clinically relevant drug interactions (see Drug Interactions under PRECAUTIONS ).
Substrates or Inhibitors of CYP2D6
Blood levels and/ or toxicity of propranolol may be increased by coadministration with substrates or inhibitors of CYP2D6, such as amiodarone, cimetidine, delavudin, fluoxetine, paroxetine, quinidine, and ritonavir. No interactions were observed with either ranitidine or lansoprazole.
Substrates or Inhibitors of CYP1A2
Blood levels and/ or toxicity of propranolol may be increased by coadministration with substrates or inhibitors of CYP1A2, such as imipramine, cimetidine, ciprofloxacin, fluvoxamine, isoniazid, ritonavir, theophylline, zileuton, zolmitriptan, and rizatriptan.
Substrates or Inhibitors of CYP2C19
Blood levels and/ or toxicity of propranolol may be increased by coadministration with substrates or inhibitors of CYP2C19, such as fluconazole, cimetidine, fluoxetine, fluvoxamine, tenioposide, and tolbutamide. No interaction was observed with omeprazole.
Inducers of Hepatic Drug Metabolism
Blood levels of propranolol may be decreased by coadministration with inducers such as rifampin, ethanol, phenytoin, and phenobarbital. Cigarette smoking also induces hepatic metabolism and has been shown to increase up to 77% the clearance of propranolol, resulting in decreased plasma concentrations.
Cardiovascular Drugs
Antiarrhythmics
The AUC of propafenone is increased by more than 200% by coadministration of propranolol.
The metabolism of propranolol is reduced by coadministration of quinidine, leading to a two-three fold increased blood concentration and greater degrees of clinical beta-blockade.
The metabolism of lidocaine is inhibited by coadministration of propranolol, resulting in a 25% increase in lidocaine concentrations.
Calcium Channel Blockers
The mean C max and AUC of propranolol are increased respectively, by 50% and 30% by coadministration of nisoldipine and by 80% and 47%, by coadministration of nicardipine.
The mean C max and AUC of nifedipine are increased by 64% and 79%, respectively, by coadministration of propranolol.
Propranolol does not affect the pharmacokinetics of verapamil and norverapamil. Verapamil does not affect the pharmacokinetics of propranolol.
Non-Cardiovascular Drugs
Migraine Drugs
Administration of zolmitriptan or rizatriptan with propranolol resulted in increased concentrations of zolmitriptan (AUC increased by 56% and C max by 37%) or rizatriptan (the AUC and C max were increased by 67% and 75%, respectively).
Theophylline
Coadministration of theophylline with propranolol decreases theophylline oral clearance by 30% to 52%.
Benzodiazepines
Propranolol can inhibit the metabolism of diazepam, resulting in increased concentrations of diazepam and its metabolites. Diazepam does not alter the pharmacokinetics of propranolol.
The pharmacokinetics of oxazepam, triazolam, lorazepam, and alprazolam are not affected by coadministration of propranolol.
Neuroleptic Drugs
Coadministration of long-acting propranolol at doses greater than or equal to 160mg/day resulted in increased thioridazine plasma concentrations ranging from 55% to 369% and increased thioridazine metabolite (mesoridazine) concentrations ranging from 33% to 209%.
Coadministration of chlorpromazine with propranolol resulted in a 70% increase in propranolol plasma level.
Anti-Ulcer Drugs
Coadministration of propranolol with cimetidine, a non-specific CYP450 inhibitor, increased propranolol AUC and C max by 46% and 35%, respectively. Coadministration with aluminum hydroxide gel (1200 mg) may result in a decrease in propranolol concentrations.
Coadministration of metoclopramide with the long-acting propranolol did not have a significant effect on propranolol’s pharmacokinetics.
Lipid Lowering Drugs
Coadministration of cholestyramine or colestipol with propranolol resulted in up to 50% decrease in propranolol concentrations.
Coadministration of propranolol with lovastatin or pravastatin, decreased 18% to 23% the AUC of both, but did not alter their pharmacodynamics. Propranolol did not have an effect on the pharmacokinetics of fluvastatin.
Warfarin
Concomitant administration of propranolol and warfarin has been shown to increase warfarin bioavailability and increase prothrombin time.
Alcohol
Concomitant use of alcohol may increase plasma levels of propranolol.
DESCRIPTION
Propranolol hydrochloride is a synthetic beta-adrenergic receptor blocking agent chemically described as 2-Propanol, 1-[(1-methylethyl)amino]-3-(1-naphthalenyloxy)-, hydrochloride,(±)-. It's molecular and structural formulae are:
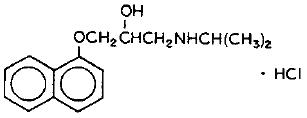
C 16 H 21 NO 2 .HCl
Propranolol hydrochloride is a stable, white, crystalline solid which is readily soluble in water and in ethanol. Its molecular weight is 295.80.
Propranolol hydrochloride tablets, USP are available as 10 mg, 20 mg, 40 mg, 60 mg, and 80 mg tablets for oral administration.
The inactive ingredients contained in propranolol hydrochloride tablets, USP are: lactose monohydrate, corn starch, sodium starch glycolate, magnesium stearate, and povidone. In addition, propranolol hydrochloride tablets, USP 10 mg, 40 mg and 80 mg contain FD &C yellow No.6 Aluminium Lake and Color D&C Yellow No. 10; propranolol hydrochloride tablets, USP 20 mg and 40mg contain FD&C Blue No.1 and propranolol hydrochloride tablets, USP 60 mg contain D&C Red No. 30 Lake.
CLINICAL PHARMACOLOGY
General
Propranolol is a nonselective beta-adrenergic receptor blocking agent possessing no other autonomic nervous system activity. It specifically competes with beta-adrenergic receptor agonist agents for available receptor sites. When access to beta-receptor sites is blocked by propranolol, the chronotropic, inotropic, and vasodilator responses to beta-adrenergic stimulation are decreased proportionately. At dosages greater than required for beta blockade, propranolol also exerts a quinidine-like or anesthetic-like membrane action, which affects the cardiac action potential. The significance of the membrane action in the treatment of arrhythmias is uncertain.
Mechanism of Action
The mechanism of the antihypertensive effect of propranolol has not been established. Factors that may contribute to the antihypertensive action include: (1) decreased cardiac output, (2) inhibition of renin release by the kidneys, and (3) diminution of tonic sympathetic nerve outflow from vasomotor centers in the brain. Although total peripheral resistance may increase initially, it readjusts to or below the pretreatment level with chronic use of propranolol. Effects of propranolol on plasma volume appear to be minor and somewhat variable.
In angina pectoris, propranolol generally reduces the oxygen requirement of the heart at any given level of effort by blocking the catecholamine-induced increases in the heart rate, systolic blood pressure, and the velocity and extent of myocardial contraction. Propranolol may increase oxygen requirements by increasing left ventricular fiber length, end diastolic pressure, and systolic ejection period. The net physiologic effect of beta-adrenergic blockade is usually advantageous and is manifested during exercise by delayed onset of pain and increased work capacity.
Propranolol exerts its antiarrhythmic effects in concentrations associated with beta-adrenergic blockade, and this appears to be its principal antiarrhythmic mechanism of action. In dosages greater than required for beta-blockade, propranolol also exerts a quinidine-like or anesthetic-like membrane action, which affects the cardiac action potential. The significance of the membrane action in the treatment of arrhythmias is uncertain.
The mechanism of the antimigraine effect of propranolol has not been established. Beta-adrenergic receptors have been demonstrated in the pial vessels of the brain.
The specific mechanism of propranolol’s antitremor effects has not been established, but beta-2 (noncardiac) receptors may be involved. A central effect is also possible. Clinical studies have demonstrated that propranolol hydrochloride is of benefit in exaggerated physiological and essential (familial) tremor.
PHARMACOKINETICS AND DRUG METABOLISM
Absorption
Propranolol is highly lipophilic and almost completely absorbed after oral administration. However, it undergoes high first-pass metabolism by the liver and on average, only about 25% of propranolol reaches the systemic circulation. Peak plasma concentrations occur about 1 to 4 hours after an oral dose.
Administration of protein-rich foods increase the bioavailability of propranolol by about 50% with no change in time to peak concentration, plasma binding, half-life, or the amount of unchanged drug in the urine.
Distribution
Approximately 90% of circulating propranolol is bound to plasma proteins (albumin and alpha 1 acid glycoprotein). The binding is enantiomer-selective. The S(-)-enantiomer is preferentially bound to alpha 1 glycoprotein and the R(+)-enantiomer preferentially bound to albumin. The volume of distribution of propranolol is approximately 4 liters/kg.
Propranolol crosses the blood-brain barrier and the placenta, and is distributed into breast milk.
Metabolism and Elimination
Propranolol is extensively metabolized with most metabolites appearing in the urine. Propranolol is metabolized through three primary routes: aromatic hydroxylation (mainly 4-hydroxylation), N-dealkylation followed by further side-chain oxidation, and direct glucuronidation. It has been estimated that the percentage contributions of these routes to total metabolism are 42%, 41% and 17%, respectively, but with considerable variability between individuals. The four major metabolites are propranolol glucuronide, naphthyloxylactic acid and glucuronic acid, and sulfate conjugates of 4- hydroxy propranolol.
In vitro studies have indicated that the aromatic hydroxylation of propranolol is catalyzed mainly by polymorphic CYP2D6. Side-chain oxidation is mediated mainly by CYP1A2 and to some extent by CYP2D6. 4-hydroxy propranolol is a weak inhibitor of CYP2D6.
Propranolol is also a substrate of CYP2C19 and a substrate for the intestinal efflux transporter, p-glycoprotein (p-gp). Studies suggest however that p-gp is not dose-limiting for intestinal absorption of propranolol in the usual therapeutic dose range.
In healthy subjects, no difference was observed between CYP2D6 extensive metabolizers (EMs) and poor metabolizers (PMs) with respect to oral clearance or elimination half-life. Partial clearance of 4-hydroxy propranolol was significantly higher and of naphthyloxyactic acid significantly lower in EMs than PMs.
The plasma half-life of propranolol is from 3 to 6 hours.
Enantiomers
Propranolol is a racemic mixture of two enantiomers, R(+) and S(-). The S(-)-enantiomer is approximately 100 times as potent as the R(+)-enantiomer in blocking beta-adrenergic receptors. In normal subjects receiving oral doses of racemic propranolol, S(-)-enantiomer concentrations exceeded those of the R(+)-enantiomer by 40-90% as a result of stereoselective hepatic metabolism. Clearance of the pharmacologically active S(-)-propranolol is lower than R(+) propranolol after intravenous and oral doses.
Special Populations
Geriatric
In a study of 12 elderly (62-79 years old) and 12 young (25-33 years old) healthy subjects, the clearance of S(-)-enantiomer of propranolol was decreased in the elderly. Additionally, the half-life of both the R(+)- and S(-)-propranolol were prolonged in the elderly compared with the young (11 hours vs. 5 hours).
Clearance of propranolol is reduced with aging due to decline in oxidation capacity (ring oxidation and side-chain oxidation). Conjugation capacity remains unchanged. In a study of 32 patients age 30 to 84 years given a single 20 mg dose of propranolol, an inverse correlation was found between age and the partial metabolic clearances to 4-hydroxypropranolol (40HP-ring oxidation) and to naphthoxylactic acid (NLA-side chain oxidation). No correlation was found between age and the partial metabolic clearance to propranolol glucuronide (PPLG-conjugation).
Gender
In a study of 9 healthy women and 12 healthy men, neither the administration of testosterone nor the regular course of the menstrual cycle affected the plasma binding of the propranolol enantiomers. In contrast, there was a significant, although non-enantioselective diminution of the binding of propranolol after treatment with ethinyl estradiol. These findings are inconsistent with another study, in which administration of testosterone cypionate confirmed the stimulatory role of this hormone on propranolol metabolism and concluded that the clearance of propranolol in men is dependent on circulating concentrations of testosterone. In women, none of the metabolic clearances for propranolol showed any significant association with either estradiol or testosterone.
Race
A study conducted in 12 Caucasian and 13 African-American male subjects taking propranolol, showed that at steady state, the clearance of R(+)- and S(-)-propranolol were about 76% and 53% higher in African-Americans than in Caucasians, respectively.
Chinese subjects had a greater proportion (18% to 45% higher) of unbound propranolol in plasma compared to Caucasians, which was associated with a lower plasma concentration of alpha 1 acid glycoprotein.
Renal Insufficiency
In a study conducted in 5 patients with chronic renal failure, 6 patients on regular dialysis, and 5 healthy subjects, who received a single oral dose of 40 mg of propranolol, the peak plasma concentrations (C max ) of propranolol in the chronic renal failure group were 2 to 3-fold higher (161±41 ng/mL) than those observed in the dialysis patients (47±9 ng/mL) and in the healthy subjects (26±1 ng/mL). Propranolol plasma clearance was also reduced in the patients with chronic renal failure.
Studies have reported a delayed absorption rate and a reduced half-life of propranolol in patients with renal failure of varying severity. Despite this shorter plasma half-life, propranolol peak plasma levels were 3-4 times higher and total plasma levels of metabolites were up to 3 times higher in these patients than in subjects with normal renal function.
Chronic renal failure has been associated with a decrease in drug metabolism via down regulation of hepatic cytochrome P450 activity resulting in a lower "first-pass" clearance.
Propranolol is not significant dialyzable.
Hepatic Insufficiency
Propranolol is extensively metabolized by the liver. In a study conducted in 7 patients with cirrhosis and 9 healthy subjects receiving 80 mg oral propranolol every 8 hours for 7 doses, the steady-state unbound propranolol concentration in patients with cirrhosis was increased 3-fold in comparison to controls. In cirrhosis, the half-life increased to 11 hours compared to 4 hours (see PRECAUTIONS ).
Drug Interactions
Interactions with Substrates, Inhibitors or Inducers of Cytochrome P-450 Enzymes
Because propranolol’s metabolism involves multiple pathways in the cytochrome P-450 system (CYP2D6,1A2, 2C19), coadministration with drugs that are metabolized by, or effect the activity (induction or inhibition) of one or more of these pathways may lead to clinically relevant drug interactions (see Drug Interactions under PRECAUTIONS ).
Substrates or Inhibitors of CYP2D6
Blood levels and/ or toxicity of propranolol may be increased by coadministration with substrates or inhibitors of CYP2D6, such as amiodarone, cimetidine, delavudin, fluoxetine, paroxetine, quinidine, and ritonavir. No interactions were observed with either ranitidine or lansoprazole.
Substrates or Inhibitors of CYP1A2
Blood levels and/ or toxicity of propranolol may be increased by coadministration with substrates or inhibitors of CYP1A2, such as imipramine, cimetidine, ciprofloxacin, fluvoxamine, isoniazid, ritonavir, theophylline, zileuton, zolmitriptan, and rizatriptan.
Substrates or Inhibitors of CYP2C19
Blood levels and/ or toxicity of propranolol may be increased by coadministration with substrates or inhibitors of CYP2C19, such as fluconazole, cimetidine, fluoxetine, fluvoxamine, tenioposide, and tolbutamide. No interaction was observed with omeprazole.
Inducers of Hepatic Drug Metabolism
Blood levels of propranolol may be decreased by coadministration with inducers such as rifampin, ethanol, phenytoin, and phenobarbital. Cigarette smoking also induces hepatic metabolism and has been shown to increase up to 77% the clearance of propranolol, resulting in decreased plasma concentrations.
Cardiovascular Drugs
Antiarrhythmics
The AUC of propafenone is increased by more than 200% by coadministration of propranolol.
The metabolism of propranolol is reduced by coadministration of quinidine, leading to a two-three fold increased blood concentration and greater degrees of clinical beta-blockade.
The metabolism of lidocaine is inhibited by coadministration of propranolol, resulting in a 25% increase in lidocaine concentrations.
Calcium Channel Blockers
The mean C max and AUC of propranolol are increased respectively, by 50% and 30% by coadministration of nisoldipine and by 80% and 47%, by coadministration of nicardipine.
The mean C max and AUC of nifedipine are increased by 64% and 79%, respectively, by coadministration of propranolol.
Propranolol does not affect the pharmacokinetics of verapamil and norverapamil. Verapamil does not affect the pharmacokinetics of propranolol.
Non-Cardiovascular Drugs
Migraine Drugs
Administration of zolmitriptan or rizatriptan with propranolol resulted in increased concentrations of zolmitriptan (AUC increased by 56% and C max by 37%) or rizatriptan (the AUC and C max were increased by 67% and 75%, respectively).
Theophylline
Coadministration of theophylline with propranolol decreases theophylline oral clearance by 30% to 52%.
Benzodiazepines
Propranolol can inhibit the metabolism of diazepam, resulting in increased concentrations of diazepam and its metabolites. Diazepam does not alter the pharmacokinetics of propranolol.
The pharmacokinetics of oxazepam, triazolam, lorazepam, and alprazolam are not affected by coadministration of propranolol.
Neuroleptic Drugs
Coadministration of long-acting propranolol at doses greater than or equal to 160mg/day resulted in increased thioridazine plasma concentrations ranging from 55% to 369% and increased thioridazine metabolite (mesoridazine) concentrations ranging from 33% to 209%.
Coadministration of chlorpromazine with propranolol resulted in a 70% increase in propranolol plasma level.
Anti-Ulcer Drugs
Coadministration of propranolol with cimetidine, a non-specific CYP450 inhibitor, increased propranolol AUC and C max by 46% and 35%, respectively. Coadministration with aluminum hydroxide gel (1200 mg) may result in a decrease in propranolol concentrations.
Coadministration of metoclopramide with the long-acting propranolol did not have a significant effect on propranolol’s pharmacokinetics.
Lipid Lowering Drugs
Coadministration of cholestyramine or colestipol with propranolol resulted in up to 50% decrease in propranolol concentrations.
Coadministration of propranolol with lovastatin or pravastatin, decreased 18% to 23% the AUC of both, but did not alter their pharmacodynamics. Propranolol did not have an effect on the pharmacokinetics of fluvastatin.
Warfarin
Concomitant administration of propranolol and warfarin has been shown to increase warfarin bioavailability and increase prothrombin time.
Alcohol
Concomitant use of alcohol may increase plasma levels of propranolol.
PHARMACODYNAMICS AND CLINICAL EFFECTS
Hypertension
In a retrospective, uncontrolled study, 107 patients with diastolic blood pressure 110 to 150 mmHg received propranolol 120 mg t.i.d. for at least 6 months, in addition to diuretics and potassium, but with no other antihypertensive agent. Propranolol contributed to control of diastolic blood pressure, but the magnitude of the effect of propranolol on blood pressure cannot be ascertained.
Angina Pectoris
In a double-blind, placebo-controlled study of 32 patients of both sexes, aged 32 to 69 years, with stable angina, propranolol 100 mg t.i.d. was administered for 4 weeks and shown to be more effective than placebo in reducing the rate of angina episodes and in prolonging total exercise time.
Atrial Fibrillation
In a report examining the long-term (5-22 months) efficacy of propranolol, 10 patients, aged 27 to 80, with atrial fibrillation and ventricular rate >120 beats per minute despite digitalis, received propranolol up to 30 mg t.i.d. Seven patients (70%) achieved ventricular rate reduction to <100 beats per minute.
Myocardial Infarction
The Beta-Blocker Heart Attack Trial (BHAT) was a National Heart, Lung and Blood Institute-sponsored multicenter, randomized, double-blind, placebo-controlled trial conducted in 31 U.S. centers (plus one in Canada) in 3,837 persons without history of severe congestive heart failure or presence of recent heart failure; certain conduction defects; angina since infarction, who had survived the acute phase of myocardial infarction. Propranolol was administered at either 60 or 80 mg t.i.d. based on blood levels achieved during an initial trial of 40 mg t.i.d. Therapy with propranolol hydrochloride, begun 5 to 21 days following infarction, was shown to reduce overall mortality up to 39 months, the longest period of follow-up. This was primarily attributable to a reduction in cardiovascular mortality. The protective effect of propranolol hydrochloride was consistent regardless of age, sex, or site of infarction. Compared with placebo, total mortality was reduced 39% at 12 months and 26% over an average follow-up period of 25 months. The Norwegian Multicenter Trial in which propranolol was administered at 40 mg q.i.d. gave overall results which support the findings in the BHAT.
Although the clinical trials used either t.i.d. or q.i.d. dosing, clinical, pharmacologic, and pharmacokinetic data provide a reasonable basis for concluding that b.i.d. dosing with propranolol should be adequate in the treatment of postinfarction patients.
Migraine
In a 34-week, placebo-controlled, 4-period, dose-finding crossover study with a double-blind randomized treatment sequence, 62 patients with migraine received propranolol 20 to 80 mg 3 or 4 times daily. The headache unit index, a composite of the number of days with headache and the associated severity of the headache, was significantly reduced for patients receiving propranolol as compared to those on placebo.
Essential Tremor
In a 2 week, double-blind, parallel, placebo-controlled study of 9 patients with essential or familial tremor, propranolol, at a dose titrated as needed from 40-80 mg t.i.d. reduced tremor severity compared to placebo.
Hypertrophic Subaortic Stenosis
In an uncontrolled series of 13 patients with New York Heart Association (NYHA) class 2 or 3 symptoms and hypertrophic subaortic stenosis diagnosed at cardiac catheterization, oral propranolol 40-80 mg t.i.d. was administered and patients were followed for up to 17 months. Propranolol was associated with improved NYHA class for most patients.
Pheochromocytoma
In an uncontrolled series of 3 patients with norepinephrine-secreting pheochromocytoma who were pretreated with an alpha adrenergic blocker (prazosin), perioperative use of propranolol at doses of 40- 80 mg t.i.d. resulted in symptomatic blood pressure control.
HOW SUPPLIED
Propranolol hydrochloride tablets, USP are supplied as follows:
10 mg: Each tablet is an orange colored, round, biconvex tablet, debossed with "P" and "10" on either side of the breakline on one side and plain on the other side.
Each 10 mg tablet contains 10 mg propranolol hydrochloride USP and is supplied in the following packages sizes:
Bottles of 30 tablets NDC 83980-005-13
Bottles of 100 tablets NDC 83980-005-01
Bottles of 500 tablets NDC 83980-005-05
Bottles of 1,000 tablets NDC 83980-005-10
20 mg: Each tablet is a blue colored, round, biconvex tablet, debossed with "P" and "20" on either side of the breakline on one side and plain on the other side.
Each 20 mg tablet contains 20 mg of propranolol hydrochloride USP and is supplied in the following package sizes:
Bottles of 30 tablets NDC 83980-006-13
Bottles of 100 tablets NDC 83980-006-01
Bottles of 500 tablets NDC 83980-006-05
Bottles of 1,000 tablets NDC 83980-006-10
40 mg: Each tablet is a green colored, round, biconvex tablet, debossed with "P" and "40"on either side of the breakline on one side and plain on the other side.
Each 40 mg tablet contains 40 mg of propranolol hydrochloride USP and is supplied in the following package sizes:
Bottles of 30 tablets NDC 83980-007-13
Bottles of 100 tablets NDC 83980-007-01
Bottles of 500 tablets NDC 83980-007-05
Bottles of 1,000 tablets NDC 83980-007-10
60 mg: Each tablet is a pink colored, round, biconvex tablet, debossed with "P" and "60" on either side of the breakline on one side and plain on the other side.
Each 60 mg tablet contains 60 mg of propranolol hydrochloride USP and is supplied in the following package sizes:
Bottles of 30 tablets NDC 83980-008-13
Bottles of 100 tablets NDC 83980-008-01
Bottles of 500 tablets NDC 83980-008-05
Bottles of 1,000 tablets NDC 83980-008-10
80 mg: Each tablet is a yellow colored, round, biconvex tablet, debossed with "P" and "80" on either side of the breakline on one side and plain on the other side.
Each 80 mg tablet contains 80 mg of propranolol hydrochloride USP and is supplied in the following package sizes:
Bottles of 30 tablets NDC 83980-009-13
Bottles of 100 tablets NDC 83980-009-01
Bottles of 500 tablets NDC 83980-009-05
Bottles of 1,000 tablets NDC 83980-009-10
Storage: Store at 20 °C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Protect from light.
Dispense in a well-closed, light-resistant container as defined in USP.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Mechanism of Action
The mechanism of the antihypertensive effect of propranolol has not been established. Factors that may contribute to the antihypertensive action include: (1) decreased cardiac output, (2) inhibition of renin release by the kidneys, and (3) diminution of tonic sympathetic nerve outflow from vasomotor centers in the brain. Although total peripheral resistance may increase initially, it readjusts to or below the pretreatment level with chronic use of propranolol. Effects of propranolol on plasma volume appear to be minor and somewhat variable.
In angina pectoris, propranolol generally reduces the oxygen requirement of the heart at any given level of effort by blocking the catecholamine-induced increases in the heart rate, systolic blood pressure, and the velocity and extent of myocardial contraction. Propranolol may increase oxygen requirements by increasing left ventricular fiber length, end diastolic pressure, and systolic ejection period. The net physiologic effect of beta-adrenergic blockade is usually advantageous and is manifested during exercise by delayed onset of pain and increased work capacity.
Propranolol exerts its antiarrhythmic effects in concentrations associated with beta-adrenergic blockade, and this appears to be its principal antiarrhythmic mechanism of action. In dosages greater than required for beta-blockade, propranolol also exerts a quinidine-like or anesthetic-like membrane action, which affects the cardiac action potential. The significance of the membrane action in the treatment of arrhythmias is uncertain.
The mechanism of the antimigraine effect of propranolol has not been established. Beta-adrenergic receptors have been demonstrated in the pial vessels of the brain.
The specific mechanism of propranolol’s antitremor effects has not been established, but beta-2 (noncardiac) receptors may be involved. A central effect is also possible. Clinical studies have demonstrated that propranolol hydrochloride is of benefit in exaggerated physiological and essential (familial) tremor.