Promethazine Hydrochloride And Dextromethorphan Hydrobromide - Promethazine Hydrochloride And Dextromethorphan Hydrobromide solution prescribing information
Promethazine Hydrochloride and Dextromethorphan Hydrobromide Oral Solution should not be used in pediatric patients less than 2 years of age because of the potential for fatal respiratory depression.
Postmarketing cases of respiratory depression, including fatalities, have been reported with use of promethazine in pediatric patients less than 2 years of age. A wide range of weight-based doses of promethazine have resulted in respiratory depression in these patients.
Caution should be exercised when administering promethazine to pediatric patients 2 years of age and older. It is recommended that the lowest effective dose of promethazine be used in pediatric patients 2 years of age and older and concomitant administration of other drugs with respiratory depressant effects be avoided.
INDICATIONS AND USAGE
Promethazine Hydrochloride and Dextromethorphan Hydrobromide Oral Solution is indicated for the temporary relief of coughs and upper respiratory symptoms associated with allergy or the common cold.
DOSAGE AND ADMINISTRATION
It is important that Promethazine Hydrochloride and Dextromethorphan Hydrobromide Oral Solution is measured with an accurate measuring device (see PRECAUTIONS – Information for Patients ). A household teaspoon is not an accurate measuring device and could lead to overdosage, especially when half a teaspoon is to be measured. It is strongly recommended that an accurate measuring device be used. A pharmacist can provide an appropriate device and can provide instructions for measuring the correct dose.
Promethazine Hydrochloride and Dextromethorphan Hydrobromide Oral Solution is CONTRAINDICATED for children under 2 years of age (see WARNINGS – Boxed Warning and Use in Pediatric Patients ).
The average effective dose for adults is 5 mL (one teaspoon) every 4 to 6 hours, not to exceed 30.0 mL in 24 hours. For children 6 years to under 12 years of age, the dose is 2.5 to 5.0 mL (one-half to one teaspoon) every 4 to 6 hours, not to exceed 20.0 mL in 24 hours. For children 2 years to under 6 years of age, the dose is 1.25 to 2.5 mL (one-quarter to one-half teaspoon) every 4 to 6 hours, not to exceed 10.0 mL in 24 hours.
CONTRAINDICATIONS
Promethazine Hydrochloride and Dextromethorphan Hydrobromide Oral Solution is contraindicated for use in pediatric patients less than two years of age.
Promethazine is contraindicated in comatose states, and in individuals known to be hypersensitive or to have had an idiosyncratic reaction to promethazine or to other phenothiazines.
Antihistamines are contraindicated for use in the treatment of lower respiratory tract symptoms, including asthma.
Dextromethorphan should not be used in patients receiving a monoamine oxidase inhibitor (see PRECAUTIONS – Drug Interactions ).
ADVERSE REACTIONS
PROMETHAZINE
Central Nervous System – Drowsiness is the most prominent CNS effect of this drug. Sedation, somnolence, blurred vision, dizziness; confusion, disorientation and extrapyramidal symptoms such as oculogyric crisis, torticollis, and tongue protrusion; lassitude, tinnitus, incoordination, fatigue, euphoria, nervousness, diplopia, insomnia, tremors, convulsive seizures, excitation, catatonic-like states, hysteria. Hallucinations have also been reported.
Cardiovascular – Increased or decreased blood pressure, tachycardia, bradycardia, faintness.
Dermatologic – Dermatitis, photosensitivity, urticaria.
Hematologic – Leukopenia, thrombocytopenia, thrombocytopenic purpura, agranulocytosis.
Gastrointestinal – Dry mouth, nausea, vomiting, jaundice.
Respiratory – Asthma, nasal stuffiness, respiratory depression (potentially fatal) and apnea (potentially fatal) (see WARNINGS – Respiratory Depression .)
Other – Angioneurotic edema. Neuroleptic malignant syndrome (potentially fatal) has also been reported (see WARNINGS – Neuroleptic Malignant Syndrome .)
Paradoxical Reactions
Hyperexcitability and abnormal movements have been reported in patients following a single administration of Promethazine Hydrochloride and Dextromethorphan Hydrobromide Oral Solution. Consideration should be given to the discontinuation of promethazine Hydrochloride and to the use of other drugs if these reactions occur. Respiratory depression, nightmares, delirium, and agitated behavior have also been reported in some of these patients.
DEXTROMETHORPHAN
Dextromethorphan occasionally causes slight drowsiness, dizziness, and gastrointestinal disturbances.
To report SUSPECTED ADVERSE REACTIONS, contact ANI Pharmaceuticals, Inc. at 1-855-204-1431 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Monoamine Oxidase (MAO) Inhibitors – Hyperpyrexia, hypotension, and death have been reported coincident with the co-administration of monoamine oxidase (MAO) inhibitors and products containing dextromethorphan. Drug interactions, including an increased incidence of extrapyramidal effects, have been reported when some MAO inhibitors and phenothiazines are used concomitantly. Thus, concomitant administration of Promethazine Hydrochloride and Dextromethorphan Hydrobromide Oral Solution and MAO inhibitors should be avoided (see CONTRAINDICATIONS ).
CNS Depressants – Promethazine may increase, prolong, or intensify the sedative action of other central-nervous-system depressants, such as alcohol, sedatives/hypnotics (including barbiturates), narcotics, narcotic analgesics, general anesthetics, tricyclic antidepressants, and tranquilizers; therefore, such agents should be avoided or administered in reduced dosage to patients receiving promethazine. When given concomitantly with Promethazine Hydrochloride and Dextromethorphan Hydrobromide Oral Solution, the dose of barbiturates should be reduced by at least one-half, and the dose of narcotics should be reduced by one-quarter to one-half. Dosage must be individualized. Excessive amounts of promethazine relative to a narcotic may lead to restlessness and motor hyperactivity in the patient with pain; these symptoms usually disappear with adequate control of the pain.
Epinephrine – Because of the potential for promethazine to reverse epinephrine’s vasopressor effect, epinephrine should NOT be used to treat hypotension associated with Promethazine Hydrochloride and Dextromethorphan Hydrobromide Oral Solution overdose.
Anticholinergics – Concomitant use of other agents with anticholinergic properties should be undertaken with caution.
DESCRIPTION
Each 5 mL (teaspoon) of Promethazine Hydrochloride and Dextromethorphan Hydrobromide Oral Solution contains 6.25 mg promethazine hydrochloride USP and 15 mg dextromethorphan hydrobromide USP in a flavored syrup base with a pH between 4.5 and 5.5. The inactive ingredients present are alcohol 7% (v/v), ascorbic acid, citric acid, black currant flavor, D&C Yellow 10, edetate disodium, FD&C Yellow 6, glycerin, L-Menthol, sodium benzoate, sodium citrate, sodium propionate, saccharin sodium, sucrose, and water.
Promethazine hydrochloride USP is a racemic compound; the empirical formula is C 17 H 20 N 2 S•HCl and its molecular weight is 320.88.
Promethazine hydrochloride USP, a phenothiazine derivative, is chemically designated as 10 H -Phenothiazine-10-ethanamine, N , N , α-trimethyl-, monohydrochloride, (+)- with the following structural formula:
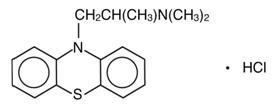
Promethazine hydrochloride USP occurs as a white to faint yellow, practically odorless, crystalline powder which slowly oxidizes and turns blue on prolonged exposure to air. It is freely soluble in water and soluble in alcohol.
Dextromethorphan hydrobromide USP is a salt of the methyl ether of the dextrorotatory isomer of levorphanol, a narcotic analgesic. It is chemically designated as 3-methoxy-17-methyl-9α, 13α, 14α–morphinan hydrobromide monohydrate with the following structural formula:
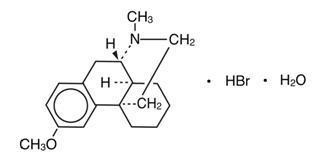
Dextromethorphan hydrobromide monohydrate occurs as white crystals, is sparingly soluble in water, and is freely soluble in alcohol. The empirical formula is C 18 H 25 NO•HBr•H 2 O, and the molecular weight of the monohydrate is 370.33. Dextromethorphan hydrobromide monohydrate is dextrorotatory with a specific rotation of +27.6 degrees in water (20 degrees C, sodium D-line).
CLINICAL PHARMACOLOGY
PROMETHAZINE
Promethazine is a phenothiazine derivative which differs structurally from the antipsychotic phenothiazines by the presence of a branched side chain and no ring substitution. It is thought that this configuration is responsible for its relative lack (1/10 that of chlorpromazine) of dopamine antagonist properties.
Promethazine is an H 1 receptor blocking agent. In addition to its antihistaminic action, it provides clinically useful sedative and antiemetic effects.
Promethazine is well absorbed from the gastrointestinal tract. Clinical effects are apparent within 20 minutes after oral administration and generally last four to six hours, although they may persist as long as 12 hours.
Promethazine is metabolized by the liver to a variety of compounds; the sulfoxides of promethazine and N-demethylpromethazine are the predominant metabolites appearing in the urine.
DEXTROMETHORPHAN
Dextromethorphan is an antitussive agent and, unlike the isomeric levorphanol, it has no analgesic or addictive properties.
Dextromethorphan acts centrally and elevates the threshold for coughing. It is about equal to codeine in depressing the cough reflex. In therapeutic dosage dextromethorphan does not inhibit ciliary activity.
Dextromethorphan is rapidly absorbed from the gastrointestinal tract and exerts its effect in 15 to 30 minutes. The duration of action after oral administration is approximately three to six hours.
Dextromethorphan is metabolized primarily by liver enzymes undergoing O-demethylation, N-demethylation, and partial conjugation with glucuronic acid and sulfate. In humans, (+)-3-hydroxy-N-methylmorphinan, (+)-3-hydroxymorphinan, and traces of unmetabolized drug were found in urine after oral administration.
HOW SUPPLIED
Promethazine Hydrochloride and Dextromethorphan Hydrobromide Oral Solution is a clear, yellow solution supplied as follows:
NDC 62559-758-04 – bottle of 4 fl. oz. (118 mL)
NDC 62559-758-16 – bottle of 16 fl. oz. (473 mL)
Keep bottles tightly closed.
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature].
Protect from light.
Dispense in tight, light-resistant container (USP/NF) with a child-resistant closure.
Distributed by ANI Pharmaceuticals, Inc. Baudette, MN 56623 
Issued: 04/2025 LB4688-02