Pindolol - Pindolol tablet prescribing information
INDICATIONS AND USAGE
Pindolol tablets are indicated in the management of hypertension. It may be used alone or concomitantly with other antihypertensive agents, particularly with a thiazide-type diuretic.
DOSAGE AND ADMINISTRATION
The dosage of pindolol tablets should be individualized. The recommended initial dose of pindolol tablets is 5 mg b.i.d. alone or in combination with other antihypertensive agents. An antihypertensive response usually occurs within the first week of treatment. Maximal response, however, may take as long as or occasionally longer than 2 weeks. If a satisfactory reduction in blood pressure does not occur within 3 to 4 weeks, the dose may be adjusted in increments of 10 mg/day at these intervals up to a maximum of 60 mg/day.
CONTRAINDICATIONS
Pindolol tablets are contraindicated in: 1) bronchial asthma; 2) overt cardiac failure; 3) cardiogenic shock; 4) second and third degree heart block; 5) severe bradycardia. (See WARNINGS .)
ADVERSE REACTIONS
Most adverse reactions have been mild. The incidences listed in the following table are derived from 12-week comparative double-blind, parallel design trials in hypertensive patients given pindolol tablets as monotherapy, given various active control drugs as monotherapy, or given placebo. Data for pindolol tablets and the positive controls were pooled from several trials because no striking differences were seen in the individual studies, with one exception. When considering all adverse reactions reported, the frequency of edema was noticeably higher in positive control trials [16% pindolol tablets vs. 9% positive control] than in placebo controlled trials [6% pindolol tablets vs. 3% placebo]. The table includes adverse reactions either volunteered or elicited, and at least possibly drug related, which were reported in greater than 2% of pindolol tablets patients and other selected important reactions.
| (and at least possibly drug-related) | |||
| Body System/ Adverse Reactions | Pindolol Tablets (N = 322) % | Active Controls Active Controls: Patients received either propranolol, a-methyldopa or a diuretic (hydrochlorothiazide or chlorthalidone). (N = 188) % | Placebo (N = 78) % |
| Central Nervous System | |||
| Bizarre or Many Dreams | 5 | 0 | 6 |
| Dizziness | 9 | 11 | 1 |
| Fatigue | 8 | 4 | 4 |
| Hallucinations | <1 | 0 | 0 |
| Insomnia | 10 | 3 | 10 |
| Nervousness | 7 | 3 | 5 |
| Weakness | 4 | 2 | 1 |
| Autonomic Nervous System | |||
| Paresthesia | 3 | 1 | 6 |
| Cardiovascular | |||
| Dyspnea | 5 | 4 | 6 |
| Edema | 6 | 3 | 1 |
| Heart Failure | <1 | <1 | 0 |
| Palpitations | <1 | 1 | 0 |
| Musculoskeletal | |||
| Chest Pain | 3 | 1 | 3 |
| Joint Pain | 7 | 4 | 4 |
| Muscle Cramps | 3 | 1 | 0 |
| Muscle Pain | 10 | 9 | 8 |
| Gastrointestinal | |||
| Abdominal Discomfort | 4 | 4 | 5 |
| Nausea | 5 | 2 | 1 |
| Skin | |||
| Pruritus | 1 | <1 | 0 |
| Rash | <1 | <1 | 1 |
The following selected (potentially important) adverse reactions were seen in 2% or fewer patients and their relationship to pindolol tablets is uncertain. CENTRAL NERVOUS SYSTEM: anxiety, lethargy; AUTONOMIC NERVOUS SYSTEM: visual disturbances, hyperhidrosis; CARDIOVASCULAR: bradycardia, claudication, cold extremities, heart block, hypotension, syncope, tachycardia, weight gain; GASTROINTESTINAL: diarrhea, vomiting; RESPIRATORY: wheezing; UROGENITAL: impotence, pollakiuria; MISCELLANEOUS: eye discomfort or burning eyes.
POTENTIAL ADVERSE EFFECTS
In addition, other adverse effects not aforementioned have been reported with other beta-adrenergic blocking agents and should be considered potential adverse effects of pindolol tablets.
Central Nervous System: Reversible mental depression progressing to catatonia; an acute reversible syndrome characterized by disorientation for time and place, short-term memory loss, emotional lability, slightly clouded sensorium, and decreased performance on neuropsychometrics.
Cardiovascular: Intensification of AV block. (See CONTRAINDICATIONS .)
Allergic: Erythematous rash; fever combined with aching and sore throat; laryngospasm; respiratory distress.
Hematologic: Agranulocytosis; thrombocytopenic and nonthrombocytopenic purpura.
Gastrointestinal: Mesenteric arterial thrombosis; ischemic colitis.
Miscellaneous: Reversible alopecia; Peyronie's disease.
The oculomucocutaneous syndrome associated with the beta-blocker practolol has not been reported with pindolol tablets during investigational use and extensive foreign experience amounting to over 4 million patient-years.
Drug Interactions
Catecholamine-depleting drugs (e.g., reserpine) may have an additive effect when given with beta-blocking agents. Patients receiving pindolol tablets plus a catecholamine-depleting agent should, therefore, be closely observed for evidence of hypotension and/or marked bradycardia which may produce vertigo, syncope, or postural hypotension.
Pindolol tablets have been used with a variety of antihypertensive agents, including hydrochlorothiazide, hydralazine, and guanethidine without unexpected adverse interactions.
Pindolol tablets have been shown to increase serum thioridazine levels when both drugs are coadministered. Pindolol levels may also be increased with this combination.
Risk of Anaphylactic Reaction
While taking beta blockers, patients with a history of severe anaphylactic reaction to a variety of allergens may be more reactive to repeated challenge, either accidental, diagnostic, or therapeutic. Such patients may be unresponsive to the usual doses of epinephrine used to treat allergic reaction.
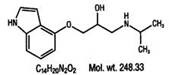
DESCRIPTION
Pindolol, a synthetic beta-adrenergic receptor blocking agent with intrinsic sympathomimetic activity is 1-(Indol-4-yloxy)-3-(isopropylamino)-2-propanol.
Its structural formula is:
Pindolol, USP is a white to off-white, odorless, crystalline powder which is practically insoluble in water; slightly soluble in methanol; and very slightly soluble in chloroform.
Each tablet for oral administration contains pindolol, USP and the following inactive ingredients: microcrystalline cellulose, pregelatinized starch, croscarmellose sodium, sodium lauryl sulfate, colloidal silicon dioxide, magnesium stearate.
CLINICAL PHARMACOLOGY
Pindolol tablets are a nonselective beta-adrenergic antagonist (beta-blocker) which possesses intrinsic sympathomimetic activity (ISA) in therapeutic dosage ranges but does not possess quinidine-like membrane stabilizing activity.
HOW SUPPLIED
Pindolol Tablets, USP are available containing 5 mg or 10 mg of pindolol, USP
The 5 mg tablets are white to off-white colored, round shaped, biconvex scored tablets debossed with 'P' above the score and '5' below the score on one side of the tablet and plain on the other side, free from physical defects.
They are available as follows:
NDC 76385-131-01
Bottles of 100 tablets
The 10 mg tablets are white to off-white colored, round shaped, biconvex scored tablets debossed with 'P' above the score and '10' below the score on one side of the tablet and plain on the other side, free from physical defects.
They are available as follows:
NDC 76385-132-01
Bottles of 100 tablets
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Protect from light.
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
To report SUSPECTED ADVERSE REACTIONS, contact Unichem Pharmaceuticals (USA) Inc. at 1-866-562-4616 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
MADE IN INDIA
Distributed by:
UNICHEM PHARMACEUTICALS (USA), INC.
East Brunswick, NJ 08816 USA.
Rev. 10/2025
213940