Get your patient on Phenylephrine Hydrochloride - Phenylephrine Hydrochloride solution/ Drops (Phenylephrine Hydrochloride)
Phenylephrine Hydrochloride - Phenylephrine Hydrochloride solution/ Drops prescribing information
INDICATIONS AND USAGE
Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% and 10% is indicated to dilate the pupil.
DOSAGE AND ADMINISTRATION
- For patients 1 year of age and older, apply one drop of Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% or 10% at 3 to 5 minute intervals up to a maximum of 3 drops per eye (2.1 ).
- In pediatric patients less than 1 year of age, one drop of Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% should be instilled at 3 to 5 minute intervals up to a maximum of 3 drops per eye (2.2 ).
General Dosing Recommendations
For patients 1 year of age or greater, apply one drop of either Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% or 10% every 3 to 5 minutes to the conjunctival fornix as required up to a maximum of 3 drops. If necessary, this dose may be repeated.
In order to obtain a greater degree of mydriasis, Phenylephrine Hydrochloride Ophthalmic Solution, USP 10% may be needed.
Pediatric Patients Less Than 1 Year of Age
In pediatric patients less than 1 year of age, one drop of Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% should be instilled at 3 to 5 minute intervals up to a maximum of 3 drops per eye.
DOSAGE FORMS AND STRENGTHS
Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% is a clear, colorless, sterile topical ophthalmic solution containing phenylephrine hydrochloride 2.5%.
Phenylephrine Hydrochloride Ophthalmic Solution, USP 10% is a clear, colorless, sterile topical ophthalmic solution containing phenylephrine hydrochloride 10%.
USE IN SPECIFIC POPULATIONS
Pregnancy
Animal reproduction studies have not been conducted with topical phenylephrine. It is also not known whether phenylephrine can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% and 10% should be given to a pregnant woman only if clearly needed.
Nursing Mothers
It is not known whether this drug is excreted in human breast milk. Because many drugs are excreted in human milk, caution should be exercised when Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% and 10% is administered to a nursing woman.
Pediatric Use
Phenylephrine Hydrochloride Ophthalmic Solution, USP 10% is contraindicated in pediatric patients less than 1 year of age. [See Contraindications (4.2) ] .
Geriatric Use
No overall differences in safety and effectiveness have been observed between elderly and younger adult patients.
CONTRAINDICATIONS
- Phenylephrine Hydrochloride Ophthalmic Solution, USP 10% is contraindicated in patients with hypertension, or thyrotoxicosis. Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% should be used in these patients (4.1 ).
- Phenylephrine Hydrochloride Ophthalmic Solution, USP 10% is contraindicated in pediatric patients less than 1 year of age due to the increased risk of systemic toxicity. Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% should be used in these patients (4.2 ).
Cardiac and Endocrine Disease
Phenylephrine Hydrochloride Ophthalmic Solution, USP 10% is contraindicated in patients with hypertension, or thyrotoxicosis. Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% should be used in these patients.
Pediatric Patients Less Than 1 Year of Age
Phenylephrine Hydrochloride Ophthalmic Solution, USP 10% is contraindicated in pediatric patients less than 1 year of age due to the increased risk of systemic toxicity. Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% should be used in these patients [See Dosage and Administration (2.2 )].
WARNINGS AND PRECAUTIONS
- Not for injection. Phenylephrine Hydrochloride Ophthalmic Solution, USP is indicated for topical ophthalmic use (5.1 ).
- Reports of serious cardiovascular reactions, some fatal, with Phenylephrine Hydrochloride Ophthalmic Solution, USP 10% solution. Monitor blood pressure in patients with cardiovascular disease (5.2 ).
- Significant elevations in blood pressure have been reported. Caution in pediatric patients less than 5 years of age, and in patients with elevated blood pressure (5.3 ).
- Rebound miosis has been reported one day after instillation (5.4 ).
Topical Ophthalmic Use
Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% and 10% is not indicated for injection.
Cardiovascular Reactions
There have been reports of serious cardiovascular reactions, including ventricular arrhythmias and myocardial infarctions, in patients using phenylephrine 10%. These episodes, some fatal, have usually occurred in patients with pre-existing cardiovascular diseases. Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% should be used in these patients.
Elevation in Blood Pressure
A significant elevation in blood pressure is not common but has been reported following conjunctival instillation of recommended doses of phenylephrine 10%. The risk is less with phenylephrine 2.5%. Caution should be exercised with the use of phenylephrine 10% in pediatric patients less than 5 years of age and patients with hyperthyroidism or cardiovascular disease. The post-treatment blood pressure of patients with cardiac and endocrine diseases and any patients who develop symptoms should be carefully monitored.
Rebound Miosis
Rebound miosis has been reported one day after receiving phenylephrine ophthalmic solution, and re instillation of the drug produced a lesser mydriatic effect.
ADVERSE REACTIONS
The following serious adverse reactions are described below and elsewhere in the labeling:
- Cardiac Disease [See Warnings and Precautions (5.2) ].
- Elevation in Blood Pressure [See Warnings and Precautions (5.3) ].
The following adverse reactions have been identified following use of phenylephrine hydrochloride ophthalmic solution. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Ocular Adverse Reactions
Eye pain and stinging on instillation, temporary blurred vision and photophobia, and conjunctival sensitization may occur.
Systemic Adverse Reactions
A marked increase in blood pressure has been reported particularly, but not limited to, low weight premature neonates, infants and hypertensive patients.
Cardiovascular reactions which have occurred primarily in hypertensive patients following topical ocular use of phenylephrine hydrochloride ophthalmic solution 10% include marked increase in blood pressure, syncope, myocardial infarction, tachycardia, arrhythmia and subarachnoid hemorrhage [See Warnings and Precautions (5.2 and 5.3) ].
DRUG INTERACTIONS
Agents That May Exaggerate Pressor Responses
Concomitant use of phenylephrine and atropine may enhance the pressor effects and induce tachycardia in some patients. Phenylephrine may potentiate the cardiovascular depressant effects of some inhalation anesthetic agents.
DESCRIPTION
Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% and 10% is a sterile, clear, colorless, topical mydriatic agent for ophthalmic use. The chemical name is (R)-3-hydroxy-α-[(methylamino)methyl]benzenemethanol hydrochloride. Phenylephrine hydrochloride is represented by the following structural formula:
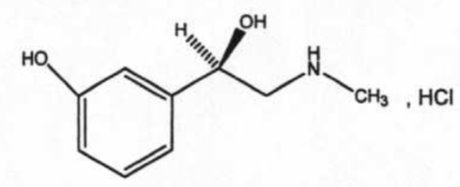
Phenylephrine hydrochloride has a molecular weight of 203.67 and an empirical formula of C 9 H 13 NO 2 -HCl.
Each mL of Phenylephrine Hydrochloride Ophthalmic Solution, 2.5% contains: ACTIVE: phenylephrine hydrochloride 25 mg (2.5%); INACTIVES: sodium phosphate monobasic, sodium phosphate dibasic; boric acid, water for injection. Hydrochloric acid and/or sodium hydroxide may be added to adjust pH (4.0-7.5). The solution has a tonicity of 500 mOsm/kg; PRESERVATIVE: benzalkonium chloride 0.01%.
Each mL of Phenylephrine Hydrochloride Ophthalmic Solution, USP 10% contains: ACTIVE: phenylephrine hydrochloride 100 mg (10%); INACTIVES: sodium phosphate monobasic, sodium phosphate dibasic; water for injection. Hydrochloric acid and/or sodium hydroxide may be added to adjust pH (4.0-7.5). The solution has a tonicity of 1000 mOsm/kg; PRESERVATIVE: benzalkonium chloride 0.01%.
CLINICAL PHARMACOLOGY
Mechanism of Action
Phenylephrine is an alpha-1 adrenergic receptor agonist used for dilation of the pupil due to its vasoconstrictor and mydriatic action. Phenylephrine possesses predominantly α-adrenergic effects. In the eye, phenylephrine acts locally as a potent vasoconstrictor and mydriatic, by constricting ophthalmic blood vessels and the radial muscle of the iris.
Pharmacodynamics
Maximal mydriasis occurs in 20-90 minutes with recovery after 3 to 8 hours.
Systemic absorption of sufficient quantities of phenylephrine may lead to systemic α-adrenergic effects, such as rise in blood pressure which may be accompanied by a reflex atropine-sensitive bradycardia.
Pharmacokinetics
The systemic exposure following topical administration of phenylephrine hydrochloride ophthalmic solution has not been studied. A higher systemic absorption is expected for the 10% solution and when the corneal barrier function has been compromised.
CLINICAL STUDIES
Pupillary dilation following topical administration of phenylephrine hydrochloride ophthalmic solution has been demonstrated in controlled clinical studies in adults and pediatric patients with different levels of iris pigmentation. Pupil movement is generally seen within 15 minutes, maximal mydriasis between 20 to 90 minutes and recovery after 3 to 8 hours. Darker irides tend to dilate slower than lighter irides.
HOW SUPPLIED/STORAGE AND HANDLING
Phenylephrine Hydrochloride Ophthalmic Solution, USP 2.5% is supplied as a sterile, aqueous, topical ophthalmic solution with a fill volume of 10 mL in a 10 mL opaque, white low density polyethylene (LDPE) bottle with a linear low density polyethylene (LLDPE) dropper tip and red cap (NDC 42702-102-10), and a fill volume of 15mL in a 15mL opaque, white LDPE bottle with an LLDPE dropper tip and red cap (NDC 42702-102-15).
Phenylephrine HCl Ophthalmic Solution, USP 10% is supplied as a sterile, aqueous, topical ophthalmic solution supplied with a fill volume of 5 mL in a 10 mL opaque, white LDPE bottle with an LLDPE dropper tip and red cap (NDC 42702-103-05).
Storage and Handling:
Store at 2°C to 25°C (36°F to 77°F).
Keep container tightly closed. Do not use if solution is brown or contains a precipitate.
After opening, Phenylephrine Hydrochloride Ophthalmic Solution, USP can be used until the expiration date on the bottle.
Mechanism of Action
Phenylephrine is an alpha-1 adrenergic receptor agonist used for dilation of the pupil due to its vasoconstrictor and mydriatic action. Phenylephrine possesses predominantly α-adrenergic effects. In the eye, phenylephrine acts locally as a potent vasoconstrictor and mydriatic, by constricting ophthalmic blood vessels and the radial muscle of the iris.