Get your patient on Phendimetrazine Tartrate - Phendimetrazine Tartrate capsule, Extended Release (Phendimetrazine Tartrate)
Phendimetrazine Tartrate - Phendimetrazine Tartrate capsule, Extended Release prescribing information
INDICATIONS AND USAGE
Phendimetrazine tartrate extended-release capsules are indicated in the management of exogenous obesity as a short term adjunct (a few weeks) in a regimen of weight reduction based on caloric restriction in patients with an initial body mass index (BMI) of greater than or equal to 30 kg/m 2 or greater than or equal to 27 kg/m 2 in the presence of other risk factors (e.g., controlled hypertension, diabetes, hyperlipidemia) who have not responded to appropriate weight reducing regimen (diet and/or exercise) alone.
Below is a chart of Body Mass Index (BMI) based on various heights and weights. BMI is calculated by taking the patient’s weight, in kilograms (kg), divided by the patient’s height, in meters (m), squared. Metric conversions are as follows: pounds ÷ 2.2 = kg; inches × 0.0254 = meters
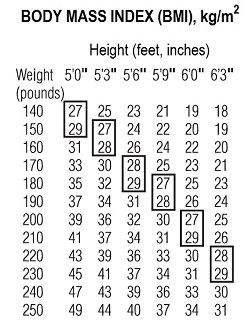
The usefulness of agents of this class (see CLINICAL PHARMACOLOGY ) should be measured against possible risk factors inherent in their use such as those described below. Phendimetrazine tartrate is indicated for use as monotherapy only.
DOSAGE AND ADMINISTRATION
Since the product is an extended-release dosage form, limit to one extended-release capsule (105 mg phendimetrazine tartrate) in the morning (30 to 60 minutes before morning meal).
Each extended-release capsule contains 105 mg phendimetrazine tartrate in a Brown/Clear capsule imprinted E 5254.
CONTRAINDICATIONS
- History of cardiovascular disease (e.g., coronary artery disease, stroke, arrhythmias, congestive heart failure, uncontrolled hypertension, pulmonary hypertension)
- During or within 14 days following the administration of monoamine oxidase inhibitors
- Hyperthyroidism
- Glaucoma
- Agitated states
- History of drug abuse
- Pregnancy (see PRECAUTIONS, Pregnancy )
- Nursing
- Use in combination with other anorectic agents or CNS stimulants
- Known hypersensitivity or idiosyncratic reactions to sympathomimetics
ADVERSE REACTIONS
The following adverse reactions are described, or described in greater detail, in other sections:
- Primary pulmonary hypertension (see WARNINGS )
- Valvular heart disease (see WARNINGS )
- Effect on the ability to engage in potentially hazardous tasks (see WARNINGS )
- Withdrawal effects following prolonged high dosage administration (see DRUG ABUSE AND DEPENDENCE )
The following adverse reactions to phendimetrazine have been identified:
Cardiovascular
Primary pulmonary hypertension and/or regurgitant cardiac valvular disease, palpitation, tachycardia, elevated blood pressure, ischemic events.
Central Nervous System
Overstimulation, restlessness, insomnia, agitation, flushing, tremor, sweating, dizziness, headache, psychotic state, blurring of vision.
Gastrointestinal
Dryness of the mouth, nausea, stomach pain, diarrhea, constipation.
Genitourinary
Urinary frequency, dysuria, changes in libido.
Drug Interactions
Monoamine Oxidase Inhibitors
Use of phendimetrazine tartrate is contraindicated during or within 14 days following the administration of monoamine oxidase inhibitors because of the risk of hypertensive crisis.
Alcohol
Concomitant use of alcohol with phendimetrazine tartrate may result in an adverse drug reaction.
Insulin and Oral Hypoglycemic Medications
Requirements may be altered.
Adrenergic Neuron Blocking Drugs
Phendimetrazine tartrate may decrease the hypotensive effect of adrenergic neuron blocking drugs.
DESCRIPTION
Phendimetrazine tartrate, as the dextro isomer, has the chemical name of (2S,3S)-3,4-dimethyl-2-phenylmorpholine L-(+)-tartrate (1:1).
The structural formula is as follows:
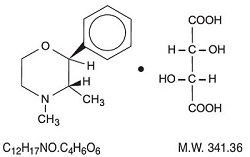
Phendimetrazine tartrate is a white, odorless crystalline powder.
It is freely soluble in water; sparingly soluble in warm alcohol, insoluble in chloroform, acetone, ether and benzene. Each capsule, for oral administration, contains 105 mg phendimetrazine tartrate manufactured in a special base designed for prolonged release.
Inactive ingredients: FD&C Blue No. 1, FD&C Red No. 40, gelatin, pharmaceutical glaze, povidone, silica gel, sodium lauryl sulfate, corn starch, sucrose, talc and trace amounts of red imprinting ink.
CLINICAL PHARMACOLOGY
Phendimetrazine tartrate is a phenylalkylamine sympathomimetic amine with pharmacological activity similar to the prototype drugs of this class used in obesity, the amphetamines. Actions include central nervous system stimulation and elevation of blood pressure. Tachyphylaxis and tolerance have been demonstrated with all drugs of this class in which these phenomena have been looked for.
Drugs of this class used in obesity are commonly known as “anorectics” or “anorexigenics”. It has not been established, however, that the action of such drugs in treating obesity is primarily one of appetite suppression. Other central nervous system actions or metabolic effects, may be involved, for example. Adult obese subjects instructed in dietary management and treated with anorectic drugs, lose more weight on the average than those treated with placebo and diet, as determined in relatively short term clinical trials.
The magnitude of increased weight loss of drug-treated patients over placebo-treated patients is only a fraction of a pound a week. The rate of weight loss is greatest in the first weeks of therapy for both drug and placebo subjects and tends to decrease in succeeding weeks. The possible origins of the increased weight loss due to the various drug effects are not established. The amount of weight loss associated with the use of an anorectic drug varies from trial to trial, and the increased weight loss appears to be related in part to variables other than the drug prescribed, such as the physician investigator, the population treated, and the diet prescribed. Studies do not permit conclusions as to the relative importance of the drug and non-drug factors on weight loss.
The natural history of obesity is measured in years, whereas the studies cited are restricted to a few weeks duration, thus, the total impact of drug-induced weight loss over that of diet alone must be considered clinically limited.
The active drug, 105 mg of phendimetrazine tartrate in each capsule of this special extended-release dosage form approximates the action of three 35 mg immediate release doses taken at four hour intervals.
The major route of elimination is via the kidneys where most of the drug and metabolites are excreted. Some of the drug is metabolized to phenmetrazine and also phendimetrazine-N-oxide.
The average half-life of elimination when studied under controlled conditions is about 3.7 hours for both the extended-release and immediate release forms. The absorption half-life of the drug from the immediate release 35 mg phendimetrazine tablets is appreciably more rapid than the absorption rate of the drug from the extended-release formulation.
HOW SUPPLIED
NDC 72989-409-30 - bottle of 30 capsules
NDC 72989-409-11 - bottle of 1,000 capsules
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Protect from moisture.
DISPENSE IN A TIGHT CONTAINER AS DEFINED IN THE USP.
To report SUSPECTED ADVERSE REACTIONS, contact Acertis Pharmaceuticals, LLC at 1-888-848-3593 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Distributed by
Acertis Pharmaceuticals, LLC
Raleigh, NC 27601
Rev. 05/2025
MF5254REV05/25
OE2820