Get your patient on Ovide - Malathion lotion (Malathion)
Ovide - Malathion lotion prescribing information
INDICATIONS AND USAGE
OVIDE Lotion is indicated for patients infected with Pediculus humanus capitis(head lice and their ova) of the scalp hair.
DOSAGE AND ADMINISTRATION
- Apply OVIDE Lotion on DRY hair in amount just sufficient to thoroughly wet the hair and scalp. Pay particular attention to the back of the head and neck while applying OVIDE Lotion. Wash hands after applying to scalp.
- Allow hair to dry naturally - use no electric heat source, and allow hair to remain uncovered.
- After 8 to 12 hours, the hair should be shampooed.
- Rinse and use a fine - toothed (nit) comb to remove dead lice and eggs.
- If lice are still present after 7 - 9 days, repeat with a second application of OVIDE Lotion.
Further treatment is generally not necessary. Other family members should be evaluated by a physician to determine if infested, and if so, receive treatment.
CONTRAINDICATIONS
OVIDE Lotion is contraindicated for neonates and infants because their scalps are more permeable and may have increased absorption of malathion. OVIDE Lotion should also not be used on individuals known to be sensitive to malathion or any of the ingredients in the vehicle.
ADVERSE REACTIONS
Malathion has been shown to be irritating to the skin and scalp. Other adverse reactions reported are chemical burns including second-degree burns. Accidental contact with the eyes can result in mild conjunctivitis.
It is not known if OVIDE Lotion has the potential to cause contact allergic sensitization.
DESCRIPTION
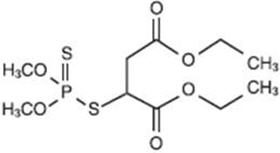
OVIDE Lotion contains 0.005 g of malathion per mL in a vehicle of isopropyl alcohol (78%), terpineol, dipentene, and pine needle oil. The chemical name of malathion is (±) - [(dimethoxyphosphinothioyl) - thio] butanedioic acid diethyl ester. Malathion has a molecular weight of 330.36, represented by C 10 H 19 O 6 PS 2 , and has the following chemical structure:
CLINICAL PHARMACOLOGY
Malathion is an organophosphate agent which acts as a pediculicide by inhibiting cholinesterase activity in vivo. Inadvertent transdermal absorption of malathion has occurred from its agricultural use. In such cases, acute toxicity was manifested by excessive cholinergic activity, i.e., increased sweating, salivary and gastric secretion, gastrointestinal and uterine motility, and bradycardia (see OVERDOSAGE ). Because the potential for transdermal absorption of malathion from OVIDE Lotion is not known at this time, strict adherence to the dosing instructions regarding its use in children, method of application, duration of exposure, and frequency of application is required.
Clinical Studies
Two controlled clinical trials evaluated the pediculicidal activity of OVIDE Lotion. Patients applied the lotion to the hair and scalp in quantities, up to a maximum of 2 fl. oz., sufficient to thoroughly wet the hair and scalp. The lotion was allowed to air dry and was shampooed with Prell shampoo 8 to 12 hours after application. Patients in both the OVIDE Lotion group and in the vehicle group were examined immediately after shampooing, 24 hours after, and 7 days after for the presence of live lice. Results are shown in the following table:
| Treatment | Immediately After | 24 Hrs. After | 7 Days After |
|---|---|---|---|
| OVIDE Lotion | 129/129 | 122/129 | 114/126 |
| OVIDE Vehicle | 105/105 | 63/105 | 31/105 |
The presence or absence of ova at day 7 was not evaluated in these studies. The presence or absence of live lice or ova at 14 days following treatment was not evaluated in these studies. The residual amount of malathion on hair and scalp is unknown.
HOW SUPPLIED
OVIDE ® (malathion) Lotion, 0.5%, is supplied in bottles of 2 fl. oz. (59 mL) NDC 51672-5293-4.
Stored at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]
Flammable. Keep away from heat and open flame.