Get your patient on Ogsiveo (Nirogacestat)
Ogsiveo prior authorization resources
Most recent state uniform prior authorization forms
Brand Resources
Ogsiveo patient education
Patient toolkit
Dosage & administration
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
The recommended dosage of OGSIVEO is 150 mg administered orally twice daily until disease progression or unacceptable toxicity. OGSIVEO may be taken with or without food.
Instruct patients to swallow OGSIVEO tablets whole and not to break, crush, or chew prior to swallowing.
If a patient vomits or misses a dose, instruct the patient to take the next dose at its scheduled time.
2.2 Dos ag e Modifications for Adverse Reactions
The recommended dose modifications for OGSIVEO for selected severe adverse reactions are summarized in Table 1 [ see Warnings and Precautions (5 ) , Adverse Reactions (6 ) ]. For other severe adverse reactions, life-threatening adverse reactions, or persistent intolerable Grade 2 adverse reactions, withhold drug until resolved to Grade ≤ 1 or baseline. Only restart at a dosage of 100 mg twice daily after considering the potential benefit and likelihood of recurrence of the adverse reaction. Permanently discontinue OGSIVEO for recurrence of severe or life-threatening adverse reaction upon rechallenge at the reduced dose.
| Adverse Reaction | Severity | OGSIVEO Dosage Modifications |
| Diarrhea persisting for ≥ 3 days despite maximal medical therapy [ see Warnings and Precautions (5.1 ) ] | Grades 3 or 4 | Withhold OGSIVEO until resolved to Grade ≤ 1 or baseline, then restart at a dosage of 100 mg twice daily. |
| Increased ALT or AST [see Warnings and Precautions (5.3 ) ] | Grade 2 (≥ 3 to 5 × ULN) | Withhold OGSIVEO until ALT, AST, or both are resolved to < 3 × ULN or baseline, then restart at a dosage of 100 mg twice daily. |
| Grades 3 or 4 (> 5 × ULN) | Permanently discontinue. | |
| Hypophosphatemia persisting for ≥ 3 days despite maximal replacement therapy [see Warnings and Precautions (5.5 ) ] | Grades 3 or 4 | Withhold OGSIVEO until resolved to Grade ≤ 1 or baseline, then restart at a dose of 100 mg twice daily. |
| Hypokalemia despite maximal replacement therapy [see Warnings and Precautions (5.5 ) ] | Grades 3 or 4 | Withhold OGSIVEO until resolved to Grade ≤ 1 or baseline, then restart at a dosage of 100 mg twice daily. |

Ogsiveo prescribing information
1 INDICATIONS AND USAGE
OGSIVEO is indicated for adult patients with progressing desmoid tumors who require systemic treatment.
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
The recommended dosage of OGSIVEO is 150 mg administered orally twice daily until disease progression or unacceptable toxicity. OGSIVEO may be taken with or without food.
Instruct patients to swallow OGSIVEO tablets whole and not to break, crush, or chew prior to swallowing.
If a patient vomits or misses a dose, instruct the patient to take the next dose at its scheduled time.
2.2 Dos ag e Modifications for Adverse Reactions
The recommended dose modifications for OGSIVEO for selected severe adverse reactions are summarized in Table 1 [ see Warnings and Precautions (5 ) , Adverse Reactions (6 ) ]. For other severe adverse reactions, life-threatening adverse reactions, or persistent intolerable Grade 2 adverse reactions, withhold drug until resolved to Grade ≤ 1 or baseline. Only restart at a dosage of 100 mg twice daily after considering the potential benefit and likelihood of recurrence of the adverse reaction. Permanently discontinue OGSIVEO for recurrence of severe or life-threatening adverse reaction upon rechallenge at the reduced dose.
| Adverse Reaction | Severity | OGSIVEO Dosage Modifications |
| Diarrhea persisting for ≥ 3 days despite maximal medical therapy [ see Warnings and Precautions (5.1 ) ] | Grades 3 or 4 | Withhold OGSIVEO until resolved to Grade ≤ 1 or baseline, then restart at a dosage of 100 mg twice daily. |
| Increased ALT or AST [see Warnings and Precautions (5.3 ) ] | Grade 2 (≥ 3 to 5 × ULN) | Withhold OGSIVEO until ALT, AST, or both are resolved to < 3 × ULN or baseline, then restart at a dosage of 100 mg twice daily. |
| Grades 3 or 4 (> 5 × ULN) | Permanently discontinue. | |
| Hypophosphatemia persisting for ≥ 3 days despite maximal replacement therapy [see Warnings and Precautions (5.5 ) ] | Grades 3 or 4 | Withhold OGSIVEO until resolved to Grade ≤ 1 or baseline, then restart at a dose of 100 mg twice daily. |
| Hypokalemia despite maximal replacement therapy [see Warnings and Precautions (5.5 ) ] | Grades 3 or 4 | Withhold OGSIVEO until resolved to Grade ≤ 1 or baseline, then restart at a dosage of 100 mg twice daily. |
3 DOSAGE FORMS AND STRENGTHS
The 100 mg tablets are round, light orange, film-coated, and debossed with “100” on one face. Each 100 mg tablet contains 100 mg nirogacestat.
The 150 mg tablets are oval, yellow orange, film-coated, and debossed with “150” on one face. Each 150 mg tablet contains 150 mg nirogacestat
8 USE IN SPECIFIC POPULATIONS
- Lactation : Advise not to breastfeed. (8.2 )
8.1 Pregnancy
Risk Summary
Based on findings from animal studies and its mechanism of action, OGSIVEO can cause fetal harm or loss of pregnancy when administered to a pregnant woman [ see C linical Pharmacology (12.1 ) ] . Oral administration of nirogacestat to pregnant rats during the period of organogenesis resulted in embryo-fetal toxicity and embryo-fetal death at maternal exposures below the human exposure at the recommended dose of 150 mg twice daily [see Data ] . There are no available data on the use of OGSIVEO in pregnant women. Advise pregnant women of the potential risk to a fetus.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
Daily oral administration of nirogacestat to pregnant rats during the period of organogenesis resulted in decreased fetal body weights, pre- and post-implantation loss, and fetal subcutis edema at doses ≥ 20 mg/kg/day (approximately 0.85 times the recommended dose of 150 mg twice daily based on area under the curve).
8.2 Lactation
Risk Summary
There are no data on the presence of nirogacestat or its metabolites in human milk or the effects of nirogacestat on a breastfed child or milk production. Because of the potential for serious adverse reactions in breastfed children, advise women not to breastfeed during treatment with OGSIVEO and for 1 week after the last dose.
8.3 Females and Males of Reproductive Potential
OGSIVEO can cause fetal harm when administered to a pregnant woman (see Use in Specific Populations (8.1 ) ].
Pregnancy Testing
Verify the pregnancy status of females of reproductive potential prior to initiating OGSIVEO [ see Use in Specific Populations (8.1 ) ].
Contraception
Females
Advise females of reproductive potential to use effective contraception during treatment with OGSIVEO and for 1 week after the last dose. OGSIVEO can affect ovarian function and the development of the ovarian follicle and therefore may reduce the effectiveness of hormonal contraceptives. Addition of a barrier method is recommended for females using hormonal contraceptives.
Males
Advise males with female partners of reproductive potential to use effective contraception during treatment with OGSIVEO and for 1 week after the last dose.
Infertility
Based on findings in animal studies, OGSIVEO can impair female and male fertility. OGSIVEO has been shown to interfere with folliculogenesis and spermatogenesis in nonclinical studies resulting in changes that included ovarian atrophy [ see Nonclinical Toxicology (13.1 ) ].
8.4 Pediatric Use
The safety and effectiveness of OGSIVEO have not been established in pediatric patients. Epiphyseal disorder, manifesting as a widening of the epiphyseal growth plate, has been reported in pediatric patients with open growth plates treated with OGSIVEO.
8.5 Geriatric Use
Of the total number of OGSIVEO-treated patients in the DeFi study, 3 (4%) were 65 years of age and older and none were 75 years of age and older. Clinical studies of OGSIVEO did not include sufficient numbers of patients 65 years of age and older to determine whether they respond differently than younger adult patients.
4 CONTRAINDICATIONS
None.
5 WARNINGS AND PRECAUTIONS
- Diarrhea : Severe diarrhea can occur. Monitor and dose modify for Grade 3-4 diarrhea. (5.1 )
- Ovarian Toxicity : Female reproductive function and fertility may be impaired. Advise females of reproductive potential of the potential risk prior to treatment and monitor routinely. (5.2 )
- Hepatotoxicity : Elevated AST and ALT can occur. Monitor AST and ALT regularly and modify dose as recommended. (5.3 )
- Non-Melanoma Skin Cancers : Perform dermatologic examination prior to initiation of OGSIVEO and routinely during treatment. (5.4 )
- Electrolyte Abnormalities : Monitor phosphate and potassium regularly and modify dose as recommended. (5.5 )
- Embryo-Fetal Toxicity : Can cause fetal harm. Advise patients of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.6 , 8.1 , 8.3 )
5.1 Diarrhea
Diarrhea, sometimes severe, can occur in patients treated with OGSIVEO [see Adverse Reactions (6.1 ) ] .
In DeFi, diarrhea occurred in 84% of patients treated with OGSIVEO, and included Grade 3 events in 16% of patients. Median time to first diarrhea event for patients treated with OGSIVEO was 9 days (range: 2 to 434 days). Monitor patients and manage using antidiarrheal medications. Modify dose as recommended [ see Dosage and Administration (2.2 ) ].
5.000000000000000e+00 2 Ovarian Toxicity
Female reproductive function and fertility may be impaired in patients being treated with OGSIVEO. Impact on fertility may depend on factors including the duration of therapy and the state of gonadal function at the time of treatment. The long-term effects of OGSIVEO on fertility have not been established. Advise patients on the potential risks for ovarian toxicity before initiating treatment with OGSIVEO [ see Use in Specific Populations (8.3 ) ]. Monitor patients for changes in menstrual cycle regularity or the development of symptoms of estrogen deficiency, including hot flashes, night sweats, and vaginal dryness.
5.000000000000000e+00 3 Hepatotoxicity
ALT or AST elevations occurred in 30% and 33% of patients who received OGSIVEO in DeFi, respectively. Grade 3 ALT or AST elevations (> 5 × ULN) occurred in 6% and 2.9% of patients, respectively [see Adverse Reactions (6.1 ) ] . Monitor liver function tests regularly and modify dose as recommended [see Dosage and Administration (2.2 ) ] .
5.000000000000000e+00 4 Non-Melanoma Skin Cancers
New non-melanoma skin cancers can occur in patients treated with OGSIVEO. In DeFi, cutaneous squamous cell carcinoma and basal cell carcinoma occurred in 2.9% and 1.4% of patients, respectively [see Adverse Reactions (6.1 ) ]. Perform dermatologic evaluations prior to initiation of OGSIVEO and routinely during treatment.
5.000000000000000e+00 5 Electrolyte Abnormalities
Electrolyte abnormalities can occur in patients treated with OGSIVEO. In DeFi, these included decreased phosphate (65%) and decreased potassium (22%). Phosphate <2 mg/dL occurred in 20% of patients who received OGSIVEO. Grade 3 decreased potassium occurred in 1.4% of patients [ see A dverse Reactions (6.1 ) ] . Monitor phosphate and potassium levels regularly and supplement as necessary. Modify dose as recommended [see Dosage and Administration (2.2 ) ].
5.000000000000000e+00 6 Embryo-Fetal Toxicity
Based on findings from animal studies and its mechanism of action, OGSIVEO can cause fetal harm when administered to pregnant women. Oral administration of nirogacestat to pregnant rats during the period of organogenesis resulted in embryo-fetal toxicity and death at maternal exposures below the human exposure at the recommended dose of 150 mg twice daily. Advise pregnant women of the potential risk to a fetus. Advise females and males of reproductive potential to use effective contraception during treatment with OGSIVEO and for 1 week after the last dose [ see Use in Specific Populations (8.1 , 8.3 ) ].
6 ADVERSE REACTIONS
The following serious adverse reactions are described elsewhere in the labeling:
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of OGSIVEO was evaluated in 69 patients enrolled in DeFi with progressing desmoid tumor [see Clinical Studies (14 ) ]. Patients received OGSIVEO 150 mg orally twice daily or placebo orally twice daily until disease progression or unacceptable toxicity. The median duration of exposure to OGSIVEO was 20.6 months (range: 0.3 to 33.6).
Serious adverse reactions occurred in 20% of patients who received OGSIVEO. Serious adverse reactions occurring in ≥ 2% of patients were ovarian toxicity (4%).
Permanent discontinuation of OGSIVEO due to an adverse reaction occurred in 20% of patients. Adverse reactions which resulted in permanent discontinuation of OGSIVEO in ≥ 2% of patients were diarrhea, ovarian toxicity, increased ALT, and increased AST.
Dosage interruptions of OGSIVEO due to an adverse reaction occurred in 51% of patients. Adverse reactions which required dosage interruption in ≥ 2% of patients included diarrhea, rash, stomatitis, hypophosphatemia, fatigue, folliculitis, nausea, and ovarian toxicity.
Dose reductions of OGSIVEO due to an adverse reaction occurred in 42% of patients. Adverse reactions which required dose reductions in ≥ 2% of patients included diarrhea, rash, stomatitis, hypophosphatemia, folliculitis, hidradenitis, and ovarian toxicity.
The most common (≥ 15% with a difference between arms of ≥ 5% compared to placebo) adverse reactions that occurred in patients receiving OGSIVEO were diarrhea, ovarian toxicity, rash, nausea, fatigue, stomatitis, headache, abdominal pain, cough, alopecia, upper respiratory tract infection and dyspnea.
Table 2 summarizes the adverse reactions that occurred in DeFi.
| Adverse Reaction | OGSIVEO (N = 69) | Placebo (N = 72) | ||
| All Grades (%) | Grade 3 (%) | All Grades (%) | Grade 3 (%) | |
| Gastrointestinal | ||||
| Diarrhea | 84 | 16 | 35 | 1.4 |
| Nausea | 54 | 1.4 | 39 | 0 |
| Stomatitis a | 39 | 4 | 4 | 0 |
| Abdominal Pain a | 22 | 1.4 | 14 | 1.4 |
| Reproductive S ystem | ||||
| Ovarian toxicity a , b | 75 c | 0 | 0 | 0 |
| Skin and S ubcutaneous T issue | ||||
| Rash a | 68 | 6 | 14 | 0 |
| Alopecia | 19 | 0 | 1.4 | 0 |
| General | ||||
| Fatigue a | 54 | 2.9 | 38 | 0 |
| Nervous S ystem | ||||
| Headache a | 30 | 0 | 15 | 0 |
| Respiratory | ||||
| Cough a | 20 | 0 | 6 | 0 |
| Dyspnea | 16 | 0 | 6 | 0 |
| Infections | ||||
| Upper respiratory tract | 17 | 0 | 2.8 | 0 |
| infection a | ||||
a Includes multiple related composite terms.
b Investigator assessment of ovarian toxicity included ovarian failure, premature menopause, amenorrhea, and menopause
c The number of females of reproductive potential in each arm is used as the denominator (OGSIVEO N = 36, Placebo N = 37).
Clinically relevant adverse reactions occurring in < 15% of patients receiving OGSIVEO in DeFi included non-melanoma skin cancers, epistaxis, hidradenitis suppurativa, folliculitis, influenza-like illness, and renal tubular disorder.
Table 3 summarizes laboratory abnormalities in DeFi.
| Laboratory Abnormality | OGSIVEO | Placebo | ||
| All Grades (%) | Grade 3 or 4 (%) | All Grades (%) | Grade 3 or 4 (%) | |
| Chemistry | ||||
| Decreased phosphate a ,b | 65 | Not Applicable | 11 | Not Applicable |
| Increased urine glucose c ,d | 51 | Not Applicable | 0 | Not Applicable |
| Increased urine protein c | 40 | 0 | 25 | 0 |
| Increased aspartate aminotransferase a | 33 | 2.9 | 18 | 1.4 |
| Increased alanine aminotransferase a | 30 | 6 | 21 | 1.4 |
| Decreased potassium a | 22 | 1.4 | 4.2 | 0 |
a The denominator used to calculate the rate was 69 for nirogacestat and 72 for placebo based on the number of patients with a baseline value and at least one post-treatment value.
b CTCAE Version 5.0 does not include numeric thresholds for grading of hypophosphatemia; all grades represent patients with lab value < Lower Limit of Normal (LLN).
c The denominator used to calculate the rate was 68 for nirogacestat and 69 for placebo based on the number of patients with a baseline value and at least one post-treatment value.
d CTCAE Version 5.0 does not include numeric thresholds for grading of increased urine glucose.
7 DRUG INTERACTIONS
- Strong or moderate CYP3A inhibitors : Avoid concomitant use. (7.1 )
- Strong or moderate CYP3A inducers : Avoid concomitant use. (7.1 )
- Gastric acid reducing agents : Avoid concomitant use with proton pump inhibitors and H2-receptor antagonists. If concomitant use cannot be avoided, OGSIVEO administration can be staggered with antacids. (7.1 )
7.1 Effect s of Other Drugs on OGSIVEO
| Strong or Moderate CYP3A Inhibitors | |
| Prevention or Management | Avoid concomitant use of OGSIVEO with strong or moderate CYP3A inhibitors including grapefruit products, Seville oranges, and starfruit. |
| Clinical Effect | Nirogacestat is a CYP3A substrate. Strong or moderate CYP3A inhibitors increase nirogacestat exposure [see Clinical Pharmacology (12.3 ) ], which may increase the risk of OGSIVEO adverse reactions . |
| Strong or Moderate CYP3A Inducers | |
| Prevention or Management | Avoid concomitant use of OGSIVEO with strong or moderate CYP3A inducers. |
| Clinical Effect | Nirogacestat is a CYP3A substrate. Strong or moderate CYP3A inducers decrease serum nirogacestat exposure [ see Clinical Pharmacology (12.3 ) ], which may reduce the effectiveness of OGSIVEO. |
| Gastric Acid Reducing Agents | |
| Prevention or Management | Avoid concomitant use with proton pump inhibitors and H2 blockers . If concomitant use cannot be avoided, OGSIVEO can be staggered with antacids (e.g., administer OGSIVEO 2 hours before or 2 hours after antacid use). |
| Clinical Effect | Nirogacestat is poorly soluble at pH ≥ 6. Gastric acid reducing agents may decrease serum nirogacestat exposure [ see Clinical Pharmacology (12.3 ) ], which may reduce the effectiveness of OGSIVEO. |
7.2 Effects of OGSIVEO on Other Drugs
| Certain CYP3ASubstrates | |
| Prevention or Management | Avoid concomitant use with CYP3A substrates where minimal concentration changes may lead to serious adverse reactions . |
| Clinical Effect | Nirogacestat increases exposure of CYP3A substrates [see Clinical Pharmacology (12.3 ) ], which may increase the risk of adverse reactions related to these substrates. |
| Certain CYP2C19 Substrates | |
| Prevention or Management | Avoid concomitant use with OGSIVEO where decreased concentrations of CYP2C19 substrates may lead to significant decreases in efficacy of the CYP2C19 substrate unless otherwise recommended in the Prescribing Information for the CYP2C19 substrate. |
| Clinical Effect | Nirogacestat decreases exposure of CYP2C19 substrates [see Clinical Pharmacology (12.3 ) ], which may decrease efficacy of these substrates. |
11 DESCRIPTION
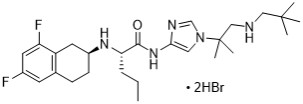
OGSIVEO oral tablets contain nirogacestat (as nirogacestat hydrobromide), a gamma (ɣ) secretase inhibitor. Nirogacestat hydrobromide is chemically known as (S)-2-(((S)-6,8-Difluoro-1,2,3,4-tetrahydronaphthalen-2-yl)amino)-N-(1-(2-methyl-1-(neopentylamino)propan-2-yl)-1H-imidazol-4-yl) pentanamide dihydrobromide. The empirical formula is C 27 H 43 Br 2 F 2 N 5 O and the molecular weight is 651.48 g/mol. Nirogacestat hydrobromide is a white to off white powder with an aqueous solubility of 11.4 mg/mL and a pH of 4.4 in water at 25°C. Nirogacestat dihydrobromide is highly soluble at low pH, however the solubility significantly decreases at pH > 6.0. The molecule has pKa’s of 5.77 and 7.13. The structural formula for nirogacestat hydrobromide is:
OGSIVEO (nirogacestat) tablets are immediate release (IR), film-coated tablets intended for oral administration. Each 100 mg tablet contains 100 mg nirogacestat as 133.050 mg nirogacestat hydrobromide. OGSIVEO 100 mg tablets are round, biconvex with an approximate diameter of 10 mm. They are film coated, light orange in color, and debossed with “100” on one face and plain on the other face. Each 150 mg tablet contains 150 mg nirogacestat as 199.574 mg nirogacestat hydrobromide. OGSIVEO 150 mg tablets are oval, biconvex with approximate dimensions of 8.5 × 17.5 mm. They are film coated, yellow orange in color, and debossed with “150” on one face and plain on the other face.
OGSIVEO tablets contain the following inactive ingredients: lactose monohydrate, magnesium stearate, microcrystalline cellulose, and sodium starch glycolate type A. The tablets are finished with Opadry ® QX orange film coating consisting of the following ingredients: FD&C yellow #6/sunset yellow FCF aluminum lake, glycerol monocaprylocaprate type 1/mono/diglycerides, iron oxide yellow, macrogol (PEG) polyvinyl alcohol graft copolymer, polyvinyl alcohol – partially hydrolyzed, talc, and titanium dioxide.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Nirogacestat is a gamma secretase inhibitor that blocks proteolytic activation of the Notch receptor. When dysregulated, Notch can activate pathways that contribute to tumor growth.
12.2 Pharmacodynamics
Exposure-Response Relationships
Higher nirogacestat exposure is associated with increased risk of Grade 3 decreased phosphate.
Cardiac Electrophysiology
At the recommended dosage, a mean increase in the QTc interval > 20 ms was not observed.
12.3 Pharmacokinetics
Nirogacestat pharmacokinetic parameters in patients with desmoid tumors are summarized in Table 6 .
| General Information | ||
| Steady state exposure [Mean (%CV)] | C max | 508 (62) ng/mL |
| AUC 0- tau | 3370 (58) ng·h/mL | |
| Time to steady-state | Approximately 6 days | |
| Accumulation ratio [Median (Min, Max)] | 1.6 (1.3, 4.6) | |
| Absorption | ||
| T max [Median (Min, Max)] | 1.5 (0.5, 6.5) hours | |
| Absolute bioavailability | 19% | |
| Food effect [dose-normalized GMR% (90% CI)] | C max | 93 % (55%, 166%) |
| AUC | 114% (76%, 171%) | |
| Distribution | ||
| Serum protein binding | 99.6% | |
| Protein Binding• | Human serum albumin | 94.6% |
| α-1 acid glycoprotein | 97.9% | |
| Apparent volume of distribution (Vz/F) [Mean (%CV)] | 1430 (65) L | |
| Elimination | ||
| Apparent Systemic Clearance (CL/F) [Mean (%CV)] | 45 (58) L/hr | |
| Terminal elimination half-life (t1/2) [Mean (%CV)] | 23 (37) hr | |
| Metabolism | ||
| Primary pathway | N-dealkylation via CYP3A4 (85%) | |
| Secondary pathways | Metabolism by CYP2C9, 2C19 and 2D6 | |
| Excretion | ||
| Feces | 38% | |
| Urine | 17% (<1% unchanged) | |
| Expired air | 9.7% | |
• Protein binding values reflect results from separate assays.
Abbreviations: AUC 0-tau = area under the time concentration curve to the dosing interval;
C max = maximum plasma concentration; T max = time to reach C max ; GMR = geometric mean ratio
Specific Populations
No clinically significant differences in the pharmacokinetics of nirogacestat were observed based on age (18 to 80 years), sex, race (Asian, Black or African American, and White), or mild or moderate renal impairment (eGFR ≥41 mL/min/1.73m 2 ).
No clinically significant differences in the pharmacokinetics of nirogacestat were observed based on age (18 to 80 years), sex, race (Asian 2.1%, Black or African American 26%, and White 66%), or mild or moderate renal impairment (eGFR ≥ 41 mL/min/1.73m 2 ).
Patients with Hepatic Impairment
The mean AUC increased by up to 16% and the mean C max decreased by up to 39% in subjects with moderate hepatic impairment (Child-Pugh Class B or NCI-ODWG Group C). The effect of severe hepatic impairment on nirogacestat pharmacokinetics is unknown.
Drug Interaction Studies
Clinical Studies and Model-Informed Approaches
Strongand moderateCYP3A inhibitors:Nirogacestat C max increased 2.5-fold and AUC 8.2-fold following coadministration of a single dose of OGSIVEO (100 mg) with itraconazole (a strong CYP3A inhibitor). Nirogacestat AUC is predicted to increase 6.3-, 5.2-, and 3.5-fold following coadministration of OGSIVEO 150 mg BID with itraconazole, ketoconazole and clarithromycin (strong CYP3A inhibitors), respectively.
Nirogacestat AUC is predicted to increase 2.7- and 3.2-fold following coadministration of OGSIVEO 150 mg BID with erythromycin (moderate CYP3A inhibitor) and fluconazole (moderate CYP3A inhibitor), respectively.
Strongand moderateCYP3A inducers:Nirogacestat AUC is predicted to decrease to 85% following coadministration of OGSIVEO 150 mg BID with rifampin (strong CYP3A inducer).
Nirogacestat AUC is predicted to decrease to 67% following coadministration of OGSIVEO 150 mg BID with efavirenz (moderate CYP3A inducer).
CYP3A substrates:Midazolam (CYP3A substrate) C max is predicted to increase 1.8-fold and AUC by 2.1-fold following coadministration of OGSIVEO 150 mg BID.
CYP2C19 substrates:Coadministration of OGSIVEO 150 mg BID with a drug that is a sensitive substrate of CYP2C19 decreases the plasma concentrations of these substrates.
Gastric acid reducing agents:Coadministration of proton pump inhibitors (e.g., omeprazole), histamine type 2 (H2)-receptor antagonists (e.g., famotidine), or antacids (e.g., calcium) is expected to reduce concentrations of nirogacestat.
Otherdrugs:No clinically significant differences in nirogacestat pharmacokinetics were predicted when used concomitantly with cimetidine (weak CYP3A inhibitor).
No clinically significant differences in dabigatran (P-gp substrate) pharmacokinetics were observed when coadministered with nirogacestat.
No clinically significant differences were predicted in the pharmacokinetics of the following drugs when used concomitantly with nirogacestat: rosiglitazone (CYP2C8 substrate) or S-warfarin (CYP2C9 substrate).
In Vitro Studies
CYP450 enzymes:Nirogacestat does not inhibit CYP1A2, 2B6, 2C8, 2C9, 2C19, and 2D6.
Nirogacestat does induce CYP2B6, CYP2C8, CYP2C9, and CYP2C19, but not CYP1A2.
Transporter systems:Nirogacestat is a P-gp substrate, but not of BCRP, OATP1B1, or OATP1B3.
Nirogacestat inhibits P-gp, but not BCRP, MATE1, MATE2-K, OATP1B1, OATP1B3, OAT1, OAT2 and OAT3.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
In a 6-month carcinogenicity study, transgenic rasH2 mice received up to 100 mg/kg/day of oral nirogacestat, resulting in mean exposure levels (AUC) less than those in humans at the recommended dose of 150 mg twice daily. No statistically significant neoplastic findings occurred. The carcinogenic potential of nirogacestat in rats has not been assessed.
Mutagenesis
Nirogacestat was not mutagenic in a bacterial reverse mutation (Ames) assay and was not clastogenic in an in vitro chromosome aberration assay in human lymphocytes or in vivo rat bone marrow micronucleus study.
Impairment of Fertility
Nirogacestat resulted in reduced fertility when administered to male and female rats at doses ≥ 5 mg/kg/day (approximately 0.16 times the recommended dose of 150 mg twice daily based on body surface area (BSA), and a lack of fertility when administered to male and female rats at doses ≥ 40 mg/kg/day (approximately 1.3 times the recommended dose of 150 mg twice daily based on BSA). Adverse findings in rats included ovarian atrophy, reduced testes weights, and decreased sperm concentration and motility.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of OGSIVEO was evaluated in 69 patients enrolled in DeFi with progressing desmoid tumor [see Clinical Studies (14 ) ]. Patients received OGSIVEO 150 mg orally twice daily or placebo orally twice daily until disease progression or unacceptable toxicity. The median duration of exposure to OGSIVEO was 20.6 months (range: 0.3 to 33.6).
Serious adverse reactions occurred in 20% of patients who received OGSIVEO. Serious adverse reactions occurring in ≥ 2% of patients were ovarian toxicity (4%).
Permanent discontinuation of OGSIVEO due to an adverse reaction occurred in 20% of patients. Adverse reactions which resulted in permanent discontinuation of OGSIVEO in ≥ 2% of patients were diarrhea, ovarian toxicity, increased ALT, and increased AST.
Dosage interruptions of OGSIVEO due to an adverse reaction occurred in 51% of patients. Adverse reactions which required dosage interruption in ≥ 2% of patients included diarrhea, rash, stomatitis, hypophosphatemia, fatigue, folliculitis, nausea, and ovarian toxicity.
Dose reductions of OGSIVEO due to an adverse reaction occurred in 42% of patients. Adverse reactions which required dose reductions in ≥ 2% of patients included diarrhea, rash, stomatitis, hypophosphatemia, folliculitis, hidradenitis, and ovarian toxicity.
The most common (≥ 15% with a difference between arms of ≥ 5% compared to placebo) adverse reactions that occurred in patients receiving OGSIVEO were diarrhea, ovarian toxicity, rash, nausea, fatigue, stomatitis, headache, abdominal pain, cough, alopecia, upper respiratory tract infection and dyspnea.
Table 2 summarizes the adverse reactions that occurred in DeFi.
| Adverse Reaction | OGSIVEO (N = 69) | Placebo (N = 72) | ||
| All Grades (%) | Grade 3 (%) | All Grades (%) | Grade 3 (%) | |
| Gastrointestinal | ||||
| Diarrhea | 84 | 16 | 35 | 1.4 |
| Nausea | 54 | 1.4 | 39 | 0 |
| Stomatitis a | 39 | 4 | 4 | 0 |
| Abdominal Pain a | 22 | 1.4 | 14 | 1.4 |
| Reproductive S ystem | ||||
| Ovarian toxicity a , b | 75 c | 0 | 0 | 0 |
| Skin and S ubcutaneous T issue | ||||
| Rash a | 68 | 6 | 14 | 0 |
| Alopecia | 19 | 0 | 1.4 | 0 |
| General | ||||
| Fatigue a | 54 | 2.9 | 38 | 0 |
| Nervous S ystem | ||||
| Headache a | 30 | 0 | 15 | 0 |
| Respiratory | ||||
| Cough a | 20 | 0 | 6 | 0 |
| Dyspnea | 16 | 0 | 6 | 0 |
| Infections | ||||
| Upper respiratory tract | 17 | 0 | 2.8 | 0 |
| infection a | ||||
a Includes multiple related composite terms.
b Investigator assessment of ovarian toxicity included ovarian failure, premature menopause, amenorrhea, and menopause
c The number of females of reproductive potential in each arm is used as the denominator (OGSIVEO N = 36, Placebo N = 37).
Clinically relevant adverse reactions occurring in < 15% of patients receiving OGSIVEO in DeFi included non-melanoma skin cancers, epistaxis, hidradenitis suppurativa, folliculitis, influenza-like illness, and renal tubular disorder.
Table 3 summarizes laboratory abnormalities in DeFi.
| Laboratory Abnormality | OGSIVEO | Placebo | ||
| All Grades (%) | Grade 3 or 4 (%) | All Grades (%) | Grade 3 or 4 (%) | |
| Chemistry | ||||
| Decreased phosphate a ,b | 65 | Not Applicable | 11 | Not Applicable |
| Increased urine glucose c ,d | 51 | Not Applicable | 0 | Not Applicable |
| Increased urine protein c | 40 | 0 | 25 | 0 |
| Increased aspartate aminotransferase a | 33 | 2.9 | 18 | 1.4 |
| Increased alanine aminotransferase a | 30 | 6 | 21 | 1.4 |
| Decreased potassium a | 22 | 1.4 | 4.2 | 0 |
a The denominator used to calculate the rate was 69 for nirogacestat and 72 for placebo based on the number of patients with a baseline value and at least one post-treatment value.
b CTCAE Version 5.0 does not include numeric thresholds for grading of hypophosphatemia; all grades represent patients with lab value < Lower Limit of Normal (LLN).
c The denominator used to calculate the rate was 68 for nirogacestat and 69 for placebo based on the number of patients with a baseline value and at least one post-treatment value.
d CTCAE Version 5.0 does not include numeric thresholds for grading of increased urine glucose.
16 HOW SUPPLIED/STORAGE AND HANDLING
OGSIVEO (nirogacestat) is supplied as 100 mg and 150 mg tablets. Each 100 mg light orange, film-coated tablet is debossed with a “100” on one face. Each 150 mg yellow orange, film-coated tablet is debossed with a “150” on one face.
| Strength | Description | Each carton contains | NDC |
| 100 mg | Round, light orange, film-coated tablet debossed with “100” on one side. | One blister card with 14 tablets | 82448-100-14 |
| 150 mg | Oval, yellow orange, film-coated tablet debossed with “150” on one side. | One blister card with 14 tablets | 82448-150-14 |
Store at 20°C to 25°C (68°F to 77°F). Excursions permitted between 15°C to 30°C (59°F to 86°F). See USP Controlled Room Temperature.
12.1 Mechanism of Action
Nirogacestat is a gamma secretase inhibitor that blocks proteolytic activation of the Notch receptor. When dysregulated, Notch can activate pathways that contribute to tumor growth.