Ofloxacin - Ofloxacin solution/ Drops prescribing information
INDICATIONS AND USAGE
Ofloxacin otic solution 0.3% is indicated for the treatment of infections caused by susceptible isolates of the designated microorganisms in the specific conditions listed below:
Otitis Externa in adults and pediatric patients, 6 months and older, due to Escherichia coli, Pseudomonas aeruginosa, Staphylococcus aureus
Chronic Suppurative Otitis Media in patients 12 years and older with perforated tympanic membranes due to, Proteus mirabilis , Pseudomonas aeruginosa, and Staphylococcus aureus .
Acute Otitis Media in pediatric patients one year and older with tympanostomy tubes due to Haemophilus influenzae, Moraxella catarrhalis , Pseudomonas aeruginosa, Staphylococcus aureus, and Streptococcus pneumoniae .
DOSAGE AND ADMINISTRATION
Otitis Externa: The recommended dosage regimen for the treatment of otitis externa is:
- For pediatric patients (from 6 months to 13 years old): Five drops (0.25 mL, 0.75 mg ofloxacin) instilled into the affected ear once daily for seven days.
- For patients 13 years and older: Ten drops (0.5 mL, 1.5 mg ofloxacin) instilled into the affected ear once daily for seven days.
- The solution should be warmed by holding the bottle in the hand for one or two minutes to avoid dizziness which may result from the instillation of a cold solution. The patient should lie with the affected ear upward, and then the drops should be instilled. This position should be maintained for five minutes to facilitate penetration of the drops into the ear canal. Repeat, if necessary, for the opposite ear.
Acute Otitis Media in pediatric patients with tympanostomy tubes: The recommended dosage regimen for the treatment of acute otitis media in pediatric patients (from 1 to 12 years old) with tympanostomy tubes is:
Five drops (0.25 mL, 0.75 mg ofloxacin) instilled into the affected ear twice daily for ten days. The solution should be warmed by holding the bottle in the hand for one or two minutes to avoid dizziness that may result from the instillation of a cold solution. The patient should lie with the affected ear upward, and then the drops should be instilled. The tragus should then be pumped 4 times by pushing inward to facilitate penetration of the drops into the middle ear. This position should be maintained for five minutes. Repeat, if necessary, for the opposite ear.
Chronic Suppurative Otitis Media with perforated tympanic membranes: The recommended dosage regimen for the treatment of chronic suppurative otitis media with perforated tympanic membranes in patients 12 years and older is:
Ten drops (0.5 mL, 1.5 mg ofloxacin) instilled into the affected ear twice daily for fourteen days. The solution should be warmed by holding the bottle in the hand for one or two minutes to avoid dizziness that may result from the instillation of a cold solution. The patient should lie with the affected ear upward, before instilling the drops. The tragus should then be pumped 4 times by pushing inward to facilitate penetration into the middle ear. This position should be maintained for five minutes. Repeat, if necessary, for the opposite ear.
CONTRAINDICATIONS
Ofloxacin otic solution 0.3% is contraindicated in patients with a history of hypersensitivity to ofloxacin, to other quinolones, or to any of the components in this medication.
ADVERSE REACTIONS
Subjects with Otitis Externa
In the Phase III clinical trials performed in support of once-daily dosing, 799 subjects with otitis externa and intact tympanic membranes were treated with ofloxacin otic solution. The studies, which served as the basis for approval, were 020 (pediatric, adolescents and adults), 016 (adolescents and adults) and 017 (pediatric). The following treatment-related adverse events occurred in 2 or more of the subjects:
† Studies 002/003 (BID) and 016/017 (QD) were active-controlled and comparative. | |||
| Adverse Event | Incidence Rate | ||
| Studies 002/003 † BID (N=229) | Studies 016/017 † QD (N=310) | Studies 020 † QD (N=489) | |
| Application Site | 3% | 16.8% | 0.6% |
| Reaction | |||
| Pruritus | 4% | 1.2% | 1.0% |
| Earache | 1% | 0.6% | 0.8% |
| Dizziness | 1% | 0.0% | 0.6% |
| Headache | 0% | 0.3% | 0.2% |
| Vertigo | 1% | 0.0% | 0.0% |
Study 020 (QD) was open and non-comparative.
An unexpected increased incidence of application site reaction was seen in studies 016/017 and was similar for both ofloxacin and the active control drug (neomycin-polymyxin B sulfate- hydrocortisone). This finding is believed to be the result of specific questioning of the subjects regarding the incidence of application site reactions.
In once daily dosing studies, there were also single reports of nausea, seborrhea, transient loss of hearing, tinnitus, otitis externa, otitis media, tremor, hypertension and fungal infection.
In twice daily dosing studies, the following treatment-related adverse events were each reported in a single subject: dermatitis, eczema, erythematous rash, follicular rash, hypoaesthesia, tinnitus, dyspepsia, hot flushes, flushing and otorrhagia.
Subjects with Acute Otitis Media with Tympanostomy Tubes (AOM TT) and Subjects with Chronic Suppurative Otitis Media (CSOM) with Perforated Tympanic Membranes
In Phase III clinical trials which formed the basis for approval, the following treatment-related adverse events occurred in 1% or more of the 656 subjects with non-intact tympanic membranes in AOM TT or CSOM treated twice-daily with ofloxacin otic solution:
| Adverse Event | Incidence (N=656) |
| Taste Perversion | 7% |
| Earache | 1% |
| Pruritus | 1% |
| Paraesthesia | 1% |
| Rash | 1% |
| Dizziness | 1% |
Other treatment-related adverse reactions reported in subjects with non-intact tympanic membranes included: diarrhea (0.6%), nausea (0.3%), vomiting (0.3%), dry mouth (0.5%), headache (0.3%), vertigo (0.5%), otorrhagia (0.6%), tinnitus (0.3%), fever (0.3%). The following treatment-related adverse events were each reported in a single subject: application site reaction, otitis externa, urticaria, abdominal pain, dysaesthesia, hyperkinesia, halitosis, inflammation, pain, insomnia, coughing, pharyngitis, rhinitis, sinusitis, and tachycardia.
Post-Marketing Adverse Events
Cases of uncommon transient neuropsychiatric disturbances have been included in spontaneous post- marketing reports. A causal relationship with ofloxacin otic solution 0.3% is unknown.
DESCRIPTION
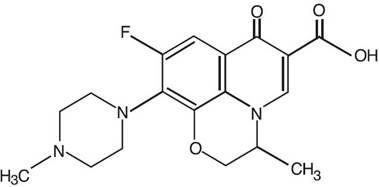
Ofloxacin otic solution 0.3% is a sterile aqueous anti-infective (anti-bacterial) solution for otic use. Chemically, ofloxacin has three condensed 6-membered rings made up of a fluorinated carboxyquinolone with a benzoxazine ring. The chemical name of ofloxacin is: (±)-9- fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido[1,2,3-de]-1,4-benzoxazine-6-carboxylic acid. The empirical formula of ofloxacin is C 18 H 20 FN 3 O 4 and its molecular weight is 361.38. The structural formula is:
Ofloxacin otic contains 0.3% (3mg/mL) ofloxacin with benzalkonium chloride (0.0025%), sodium chloride (0.9%), and water for injection. Hydrochloric acid and sodium hydroxide are added to adjust the pH between 6.0 and 6.6.
CLINICAL PHARMACOLOGY
Pharmacokinetics : Drug concentrations in serum (in subjects with tympanostomy tubes and perforated tympanic membranes), in otorrhea, and in mucosa of the middle ear (in subjects with perforated tympanic membranes) were determined following otic administration of ofloxacin solution. In two single-dose studies, mean ofloxacin serum concentrations were low in adult patients with tympanostomy tubes, with and without otorrhea, after otic administration of a 0.3% solution (4.1 ng/mL (n=3) and 5.4 ng/mL (n=5), respectively). In adults with perforated tympanic membranes, the maximum serum drug level of ofloxacin detected was 10 ng/mL after administration of a 0.3% solution. Ofloxacin was detectable in the middle ear mucosa of some adult subjects with perforated tympanic membranes (11 of 16 subjects). The variability of ofloxacin concentration in middle ear mucosa was high. The concentrations ranged from 1.2 to 602 μg/g after otic administration of a 0.3% solution. Ofloxacin was present in high concentrations in otorrhea (389 - 2850 μg/g, n=13) 30 minutes after otic administration of a 0.3% solution in subjects with chronic suppurative otitis media and perforated tympanic membranes. However, the measurement of ofloxacin in the otorrhea does not necessarily reflect the exposure of the middle ear to ofloxacin.
Microbiology : Ofloxacin has in vitro activity against a wide range of gram-negative and gram-positive microorganisms. Ofloxacin exerts its antibacterial activity by inhibiting DNA gyrase, a bacterial topoisomerase. DNA gyrase is an essential enzyme which controls DNA topology and assists in DNA replication, repair, deactivation, and transcription. Cross-resistance has been observed between ofloxacin and other fluoroquinolones. There is generally no cross-resistance between ofloxacin and other classes of antibacterial agents such as beta-lactams or aminoglycosides.
Ofloxacin has been shown to be active against most isolates of the following microorganisms, both in vitro and clinically in otic infections as described in the INDICATIONS AND USAGE section.
AEROBES, GRAM-POSITIVE :
Staphylococcus aureus
Streptococcus pneumoniae
AEROBES, GRAM-NEGATIVE:
Escherichia coli
Haemophilus influenzae
Moraxella catarrhalis
Proteus mirabilis
Pseudomonas aeruginosa
HOW SUPPLIED
Ofloxacin otic solution 0.3% is supplied in plastic dropper bottles containing 5 mL and 10 mL.
NDC 70069- 686 -01 Ofloxacin Otic solution (5mL)
NDC 70069- 687 -01 Ofloxacin Otic solution (10 mL)
Store at 25 °C (77 °F), excursions permitted to 15°-30°C (59-86 °F). Protect from light.
Rx Only
Manufactured for:
Somerset Therapeutics, LLC
Somerset, NJ 08873
Made in India
Code No.: KR/DRUGS/KTK/28/289/97
1201075
ST-OFO/P/00
Revised: October, 2024