Nortriptyline Hydrochloride - Nortriptyline Hydrochloride solution prescribing information
Suicidality and Antidepressant Drugs
Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of nortriptyline hydrochloride oral solution or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Nortriptyline hydrochloride oral solution is not approved for use in pediatric patients. (See WARNINGS: Clinical Worsening and Suicide Risk , PRECAUTIONS: Information for Patients, and PRECAUTIONS: Pediatric Use ).
INDICATIONS AND USAGE
Nortriptyline hydrochloride oral solution is indicated for the relief of symptoms of depression. Endogenous depressions are more likely to be alleviated than are other depressive states.
DOSAGE AND ADMINISTRATION
Nortriptyline hydrochloride oral solution is not recommended for children.
Nortriptyline hydrochloride oral solution is administered orally in the form of an oral solution. Lower than usual dosages are recommended for elderly patients and adolescents. Lower dosages are also recommended for outpatients than for hospitalized patients who will be under close supervision. The physician should initiate dosage at a low level and increase it gradually, noting carefully the clinical response and any evidence of intolerance. Following remission, maintenance medication may be required for a longer period of time at the lowest dose that will maintain remission.
If a patient develops minor side effects, the dosage should be reduced. The drug should be discontinued promptly if adverse effects of a serious nature or allergic manifestations occur.
Usual Adult Dose
25 mg 3 or 4 times daily; dosage should begin at a low level and be increased as required. As an alternate regimen, the total daily dosage may be given once a day. When doses above 100 mg daily are administered, plasma levels of nortriptyline should be monitored and maintained in the optimum range of 50 to 150 ng/mL. Doses above 150 mg per day are not recommended.
Elderly and Adolescent Patients
30 to 50 mg/day, in divided doses, or the total daily dosage may be given once a day.
Switching a Patient To or From a Monoamine Oxidase Inhibitor (MAOI) Intended to Treat Psychiatric Disorders
At least 14 days should elapse between discontinuation of an MAOI intended to treat psychiatric disorders and initiation of therapy with nortriptyline hydrochloride oral solution. Conversely, at least 14 days should be a lowed after stopping nortriptyline hydrochloride oral solution before starting an MAOI intended to treat psychiatric disorders (See CONTRAINDICATIONS ).
Use of Nortriptyline Hydrochloride With Other MAOIs, Such as Linezolid or Methylene Blue
Do not start nortriptyline hydrochloride oral solution in a patient who is being treated with linezolid or intravenous methylene blue because there is increased risk of serotonin syndrome. In a patient who requires more urgent treatment of a psychiatric condition, other interventions, including hospitalization, should be considered (See CONTRAINDICATIONS ).
In some cases, a patient already receiving nortriptyline therapy may require urgent treatment with linezolid or intravenous methylene blue. If acceptable alternatives to linezolid or intravenous methylene blue treatment are not available and the potential benefits of linezolid or intravenous methylene blue treatment are judged to outweigh the risks of serotonin syndrome in a particular patient, nortriptyline hydrochloride oral solution should be stopped promptly, and linezolid or intravenous methylene blue can be administered. The patient should be monitored for symptoms of serotonin syndrome for two weeks or until 24 hours after the last dose of linezolid or intravenous methylene blue, whichever comes first. Therapy with nortriptyline hydrochloride oral solution may be resumed 24 hours after the last dose of linezolid or intravenous methylene blue (See WARNINGS ).
The risk of administering methylene blue by non-intravenous routes (such as oral tablets or by local injection) or in intravenous doses much lower than 1 mg/kg with nortriptyline hydrochloride is unclear. The clinician should, nevertheless, be aware of the possibility of emergent symptoms of serotonin syndrome with such use (See WARNINGS ).
CONTRAINDICATIONS
Monoamine Oxidase Inhibitors (MAOIs)
The use of MAOIs intended to treat psychiatric disorders with nortriptyline hydrochloride oral solution or within 14 days of stopping treatment with nortriptyline hydrochloride oral solution is contraindicated because of an increased risk of serotonin syndrome. The use of nortriptyline hydrochloride oral solution within 14 days of stopping an MAOI intended to treat psychiatric disorders is also contraindicated (See WARNINGS and DOSAGE AND ADMINISTRATION ).
Starting nortriptyline hydrochloride oral solution in a patient who is being treated with MAOIs such as linezolid or intravenous methylene blue is also contraindicated because of an increased risk of serotonin syndrome (See WARNINGS and DOSAGE AND ADMINISTRATION ).
Hypersensitivity to Tricyclic Antidepressants
Cross-sensitivity between nortriptyline hydrochloride oral solution and other dibenzazepines is a possibility.
Myocardial Infarction
Nortriptyline hydrochloride oral solution USP is contraindicated during the acute recovery period after myocardial infarction.
ADVERSE REACTIONS
Note – Included in the following list are a few adverse reactions that have not been reported with this specific drug. However, the pharmacologic similarities among the tricyclic antidepressant drugs require that each of these reactions be considered when nortriptyline is administered.
Cardiovascular - Hypotension, hypertension, tachycardia, palpitation, myocardial infarction, arrhythmias, heart block, stroke.
Psychiatric - Confusional states (especially in the elderly), with hallucinations, disorientation, delusions; anxiety, restlessness, agitation; insomnia, panic, nightmares; hypomania; exacerbation of psychosis.
Neurologic - Numbness, tingling, paresthesias of extremities; incoordination, ataxia, tremors; peripheral neuropathy; extrapyramidal symptoms; seizures, alteration of EEG patterns; tinnitus.
Anticholinergic - Dry mouth and, rarely, associated sublingual adenitis; blurred vision, disturbance of accommodation, mydriasis; constipation, paralytic ileus; urinary retention, delayed micturition, dilation of the urinary tract.
Allergic - Skin rash, petechiae, urticaria, itching, photosensitization (avoid excessive exposure to sunlight); edema (general or of face and tongue), drug fever, cross- sensitivity with other tricyclic drugs.
Hematologic - Bone-marrow depression, including agranulocytosis; eosinophilia; purpura; thrombocytopenia.
Gastrointestinal - Nausea and vomiting, anorexia, epigastric distress, diarrhea; peculiar taste, stomatitis, abdominal cramps, black tongue.
Endocrine - Gynecomastia in the male; breast enlargement and galactorrhea in the female; increased or decreased libido, impotence; testicular swelling; elevation or depression of blood sugar levels; syndrome of inappropriate ADH (antidiuretic hormone) secretion.
Other - Jaundice (simulating obstructive); altered liver function, weight gain or loss; perspiration; flushing; urinary frequency, nocturia; drowsiness, dizziness, weakness, fatigue; headache; parotid swelling; alopecia.
Withdrawal Symptoms - Though these are not indicative of addiction, abrupt cessation of treatment after prolonged therapy may produce nausea, headache, and malaise.
Postmarketing Experience
The following adverse drug reaction has been reported during post-approval use of nortriptyline hydrochloride. Because this reaction is reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate frequency.
Cardiac Disorders – Brugada syndrome
Eye Disorders – angle-closure glaucoma
To report SUSPECTED ADVERSE REACTIONS, contact Advagen Pharma Ltd, at 866-488-0312 or FDA at 1-800- FDA-1088 or www.fda.gov/medwatch .
DESCRIPTION
Nortriptyline hydrochloride is 1-propanamine,3-(10,11-dihydro- 5H -dibenzo [ a,d ]cyclohepten-5-ylidene)- N-methyl-hydrochloride. The structural formula is as follows:
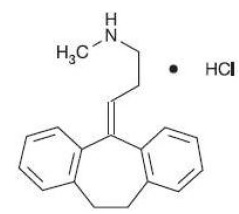
Molecular weight – 299.84 Molecular formula – C 19 H 21 N. HCl
The oral solution contains nortriptyline hydrochloride equivalent to 10 mg/5 mL (38.0 μmol) of the base and alcohol 4% v/v. It also contains artificial cherry flavor, citric acid, purified water, sodium benzoate, sodium hydroxide and sorbitol.
CLINICAL PHARMACOLOGY
The mechanism of mood elevation by tricyclic antidepressants is at present unknown. Nortriptyline hydrochloride is not a monoamine oxidase inhibitor. It inhibits the activity of such diverse agents as histamine, 5-hydroxytryptamine, and acetylcholine. It increases the pressor effect of norepinephrine but blocks the pressor response of phenethylamine. Studies suggest that nortriptyline hydrochloride interferes with the transport, release, and storage of catecholamines. Operant conditioning techniques in rats and pigeons suggest that nortriptyline hydrochloride has a combination of stimulant and depressant properties.
HOW SUPPLIED
Nortriptyline Hydrochloride Oral Solution USP, 10 mg/5 mL base is a clear, colorless solution with cherry flavor, free from any visible foreign and particulate matter, free of precipitation and hazy mass. It is available in 473 mL (16 fl oz) amber PET bottle, NDC 72888-157-39.
Store and Dispense
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]. Preserve in tight, light-resistant containers as defined in the USP.
All trademarks are the property of their respective owners. Dispense with Medication Guide available at: www.advagenpharma.com/medguide/nortriptylinehydrochloridesolution
Manufactured by: Rubicon Research Limited Satara 415004, India.
Distributed by: Advagen Pharma Ltd East Windsor, NJ 08520, USA
Rev: 01/2025
Dispense with Medication Guide available at: www.advagenpharma.com/medguide/nortriptylinehydrochloridesolution