Nelarabine - Nelarabine injection prescribing information
WARNING: NEUROLOGIC ADVERSE REACTIONS
Severe neurologic adverse reactions have been reported with the use of nelarabine injection. These adverse reactions have included altered mental states including severe somnolence, central nervous system effects including convulsions, and peripheral neuropathy ranging from numbness and paresthesias to motor weakness and paralysis. There have also been reports of adverse reactions associated with demyelination, and ascending peripheral neuropathies similar in appearance to Guillain-Barré syndrome [see Warnings and Precautions (5.1) ] .
Full recovery from these adverse reactions has not always occurred with cessation of therapy with nelarabine injection. Monitor frequently for signs and symptoms of neurologic toxicity during treatment with nelarabine injection. Discontinue nelarabine injection for neurologic adverse reactions of NCI Common Toxicity Criteria for Adverse Events (CTCAE) Grade 2 or greater [see Warnings and Precautions (5.1) ] .
1 INDICATIONS & USAGE
Nelarabine injection is indicated for the treatment of T-cell acute lymphoblastic leukemia (T-ALL) and T-cell lymphoblastic lymphoma (T-LBL) in adult and pediatric patients age 1 year and older whose disease has not responded to or has relapsed following treatment with at least 2 chemotherapy regimens.
2 DOSAGE & ADMINISTRATION
- Adult Dose : 1500 mg/m 2 administered intravenously over 2 hours on Days 1, 3, and 5 repeated every 21 days. (2.1 )
- Pediatric Dose : 650 mg/m 2 administered intravenously over 1 hour daily for 5 consecutive days repeated every 21 days. (2.1 )
- Discontinue treatment for neurologic reactions greater than or equal to Grade 2. (2.2 )
- Dosage may be delayed for hematologic reactions. (2.2 )
- Take measures to prevent hyperuricemia. (2.4 )
2.1 Recommended Dosage
This product is for intravenous use only.
Adult Dosage : The recommended adult dose of nelarabine injection is 1500 mg/m 2 administered intravenously over 2 hours on Days 1, 3, and 5 repeated every 21 days. Administer nelarabine injection undiluted.
Pediatric Dosage : The recommended pediatric dose of nelarabine injection is 650 mg/m 2 administered intravenously over 1 hour daily for 5 consecutive days repeated every 21 days. Administer nelarabine injection undiluted.
The recommended duration of treatment for adult and pediatric patients has not been clearly established. In clinical trials, treatment was generally continued until there was evidence of disease progression, the patient experienced unacceptable toxicity, the patient became a candidate for hematopoietic stem cell transplantation (HSCT), or the patient no longer continued to benefit from treatment.
2.2 Dosage Modification
Discontinue nelarabine injection if the patient develops a neurologic adverse reaction of NCI CTCAE Grade 2 or greater. Dosage may be delayed for other toxicity, including hematologic toxicity [see Boxed Warning , Warnings and Precautions (5.1 , 5.2 )].
2.3 Dosage in Special Populations
Nelarabine injection has not been studied in patients with renal or hepatic dysfunction [see Use in Specific Populations (8.6 , 8.7 )]. No dose adjustment is recommended for patients with a creatinine clearance (CLCr) greater than or equal to 50 mL/min [see Clinical Pharmacology (12.3) ]. There are insufficient data to support a dose recommendation for patients with a CLCr less than 50 mL/min.
2.4 Prevention of Hyperuricemia
Take precautions against hyperuricemia (e.g., hydration, urine alkalinization, and prophylaxis with allopurinol) [see Warnings and Precautions (5.4) ].
2.5 Instructions for Handling, Preparation, and Administration
Handling : Nelarabine injection is a cytotoxic agent. Caution should be used during handling and preparation. Use of gloves and other protective clothing to prevent skin contact is recommended. Proper aseptic technique should be used. Guidelines for proper handling and disposal of anticancer drugs have been published. 1
Preparation and Administration : Administer nelarabine injection undiluted. Transfer the appropriate dose of nelarabine injection into polyvinylchloride (PVC) infusion bags or glass containers and administer as a 2-hour infusion in adult patients and as a 1-hour infusion in pediatric patients.
Prior to administration, inspect the drug product visually for particulate matter and discoloration.
Discard unused portion.
Stability: Nelarabine Injection is stable in PVC infusion bags and glass containers for up to 8 hours at up to 30°C.
3 DOSAGE FORMS & STRENGTHS
Injection: 250 mg/50 mL (5 mg/mL) single-dose vial
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Based on its mechanism of action and findings in animal studies, nelarabine injection can cause fetal harm when administered to a pregnant woman [see Clinical Pharmacology (12.1) ]. Limited available data with nelarabine injection use in pregnant women are insufficient to determine a drug-associated risk for major birth defects, miscarriage or adverse maternal, or fetal outcomes. There are risks to the pregnant woman associated with untreated leukemia or lymphoma (see Clinical Considerations). In animal reproduction studies, intravenous administration of nelarabine to pregnant rabbits during the period of organogenesis resulted in teratogenicity at maternal doses below the recommended human adult dose of 1500 mg/m 2 /day (see Data) . Advise pregnant women of the potential risk to the fetus.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies are 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo-fetal Risk
There are risks to the mother from untreated leukemia or lymphoma, including anemia, thrombocytopenia, and death.
Data
Animal Data
In an embryo-fetal development study in which pregnant rabbits were administered daily doses of nelarabine during organogenesis, increased incidences of fetal malformations, anomalies, and variations were observed at doses greater than or equal to 360 mg/m 2 /day (8-hour IV infusion; approximately 25% of the recommended human adult dose compared on a mg/m 2 basis), which was the lowest dose tested. Cleft palate was seen in rabbits given 3600 mg/m 2 /day (approximately 2-fold the adult dose), absent pollices (digits) in rabbits given greater than or equal to 1200 mg/m 2 /day (approximately 75% of the recommended adult dose), while absent gall bladder, absent accessory lung lobes, fused or extra sternebrae, and delayed ossification was seen at all doses. Maternal body weight gain and fetal body weights were reduced in rabbits given 3600 mg/m 2 /day (approximately 2-fold the adult dose), but could not account for the increased incidence of malformations seen at this or lower administered doses.
8.2 Lactation
Risk Summary
There are no data on the presence of nelarabine or ara-G in human or animal milk, the effect on the breastfed child, or the effect on milk production. Because of the potential for serious adverse reactions in the breastfed child from nelarabine injection, such as severe neurological reactions, advise women not to breastfeed during treatment with nelarabine injection.
8.3 Females and Males of Reproductive Potential
Pregnancy Testing
Nelarabine injection can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1) ] . Verify the pregnancy status of females of reproductive potential prior to starting treatment with nelarabine injection.
Contraception
Females
Nelarabine injection can cause fetal harm when administered to a pregnant woman [see Warnings and Precautions (5.3) , Use in Specific Populations (8.1) ] . Because of the potential for genotoxicity, advise females of reproductive potential to use effective contraception during treatment with nelarabine injection.
Males
Because of the potential for genotoxicity, advise males (including those who have had vasectomies) with female partners of reproductive potential to use condoms during treatment with nelarabine injection and for 3 months after the last dose [see Nonclinical Toxicology (13.1) ] .
8.4 Pediatric Use
The safety and effectiveness of nelarabine injection for relapsed or refractory T-ALL and T-LBL has been established in pediatric patients age 1 year and older. The effectiveness of nelarabine injection in pediatric patients is supported by one single-arm clinical trial, and safety has been assessed in 165 pediatric patients age 1 year and older across multiple Phase I and Phase II trials. The trial establishing efficacy included 84 patients age 21 years and younger, who had relapsed or refractory T-ALL or T-LBL. The most frequent adverse reactions of any grade occurring on treatment in this study were hematologic laboratory abnormalities. Hematologic toxicity observed in the pediatric population was higher than that seen in the adult population [see Dosage and Administration (2.1) , Adverse Reactions (6.1) , Clinical Studies (14.2) ].
Nervous system adverse reactions have been reported for 42% of pediatric patients across the Phase I and Phase II trials. The incidence of nervous system adverse reactions was less in the pediatric population than that seen in adult patients with relapsed/refractory T-ALL/T-LBL [see Adverse Reactions (6.1) ].
In a phase III study of nelarabine injection in combination with multi-agent chemotherapy as first-line therapy, there were 411 patients with T-ALL or T-LBL treated with nelarabine injection. The safety profile in the 357 patients age 1 to 16 years was consistent with that seen in older patients in the study [see Adverse Reactions (6.1) ].
Due to lack of long-term follow up data, a determination of the impact of nelarabine injection on the growth and pubertal development of pediatric patients cannot be made.
8.5 Geriatric Use
Clinical studies of nelarabine injection did not include sufficient numbers of patients age 65 and over to determine whether they respond differently from younger patients. In an exploratory analysis, increasing age, especially age 65 years and older, appeared to be associated with increased rates of neurologic adverse reactions. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection [see Use in Specific Populations (8.6) ] .
8.6 Renal Impairment
Ara-G clearance decreased as renal function decreased [see Clinical Pharmacology (12.3) ] . Because the risk of adverse reactions to this drug may be greater in patients with moderate (CLCr 30 to 50 mL/min) or severe (CLCr less than 30 mL/min) renal impairment, these patients should be closely monitored for toxicities when treated with nelarabine injection [see Dosage and Administration (2.3) ].
8.7 Hepatic Impairment
The influence of hepatic impairment on the pharmacokinetics of nelarabine has not been evaluated. Because the risk of adverse reactions to this drug may be greater in patients with severe hepatic impairment (total bilirubin greater than 3 times upper limit of normal), these patients should be closely monitored for toxicities when treated with nelarabine injection.
4 CONTRAINDICATIONS
None.
5 WARNINGS AND PRECAUTIONS
5.1 Neurologic Adverse Reactions
Nervous system adverse reactions of any grade were reported for 223 (76%) adult patients across the Phase I and Phase II trials, and Grade 3 or higher (severe, life-threatening, or fatal) adverse reactions were reported for 55 (19%) patients following initiation of nelarabine injection therapy [see Adverse Reactions (6.1) ] . Based on patients with complete data, the median time to onset of first event is 5 days from start of first infusion (range: 1-166), and the median duration is 6 days (range: 1-393 days).
Nervous system adverse reactions of any grade were reported for 69 (42%) pediatric patients across the Phase I and Phase II trials, and Grade 3 or higher (severe, life-threatening, or fatal) adverse reactions were reported for 25 (15%) patients following initiation of nelarabine injection therapy [see Adverse Reactions (6.1) ] . Based on patients with complete data, the median time to onset of first event is 8 days from start of first infusion (range: 1-269), and the median duration is 2 days (range: 1-82 days).
Common signs and symptoms of nelarabine injection-related neurotoxicity include somnolence, headache, paresthesia and dysesthesia, dizziness, neuropathy (sensory and motor), cerebellar disturbances and tremor. Severe neurologic toxicity can manifest as coma, status epilepticus, craniospinal demyelination, or ascending neuropathy similar in presentation to Guillain-Barré syndrome.
Full recovery from these adverse reactions has not always occurred with cessation of therapy with nelarabine injection. Patients treated previously or concurrently with intrathecal chemotherapy or previously with craniospinal irradiation may be at increased risk for neurologic adverse events.
Monitor patients frequently for signs and symptoms of neurologic toxicity during and for at least 24 hours after completion of treatment with nelarabine injection. Discontinue nelarabine injection for neurologic adverse reactions of NCI CTCAE Grade 2 or greater and provide supportive care [see Dosage and Administration (2.2) , Adverse Reactions (6.1) ] .
5.2 Hematologic Adverse Reactions
Leukopenia, thrombocytopenia, anemia, and neutropenia, including febrile neutropenia, have been associated with nelarabine injection therapy. Complete blood counts including platelets should be monitored regularly [see Dosage and Administration (2.2) , Adverse Reactions (6.1) ] .
5.3 Embryo-Fetal Toxicity
Based on its mechanism of action and findings in animal studies, nelarabine injection can cause fetal harm when administered to a pregnant woman [see Clinical Pharmacology (12.1) ] . In animal reproduction studies, intravenous administration of nelarabine to pregnant rabbits during the period of organogenesis resulted in teratogenicity at maternal doses below the recommended human adult dose of 1500 mg/m 2 /day (see Data) .
Advise pregnant women of the potential risk to the fetus. Advise females of reproductive potential to use effective contraception during treatment with nelarabine injection. Advise males with female partners of reproductive potential to use condoms during treatment with nelarabine injection and for 3 months after the last dose [see Use in Specific Populations (8.1 , 8.3 ), Nonclinical Toxicology (13.1) ].
5.4 Tumor Lysis Syndrome
Patients receiving nelarabine injection should receive intravenous hydration according to standard medical practice for the management of hyperuricemia in patients at risk for tumor lysis syndrome. Consideration should be given to the use of allopurinol in patients at risk of hyperuricemia [see Dosage and Administration (2.4) ].
5.5 Vaccinations
Avoid the administration of live vaccines to immunocompromised patients.
5.6 Effects on Ability to Drive and Use Machines
Patients treated with nelarabine injection may experience somnolence during and for several days after treatment [see Adverse Reactions (6.1) ]. Advise patients to refrain from driving or engaging in hazardous occupations or activities until somnolence has resolved.
6 ADVERSE REACTIONS
The following clinically-significant adverse reactions are discussed in greater detail in other sections of the label:
- Neurologic [see Boxed Warning, Warnings and Precautions (5.1) ]
- Hematologic [see Warnings and Precautions (5.2) ]
- Tumor Lysis Syndrome [see Warnings and Precautions (5.4) ]
- Effects on Ability to Drive and Use Machines [see Warnings and Precautions (5.6) ]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Relapsed or Refractory T-ALL and T-LBL
Nelarabine injection was studied in 459 patients in Phase I and Phase II clinical trials.
Adult Patient : The safety profile of nelarabine injection is based on data from 103 adult patients treated with the recommended dose and schedule in 2 studies: an adult T- ALL/T-cell T-LBL trial and an adult chronic lymphocytic leukemia trial.
The most common adverse reactions in adults were fatigue; gastrointestinal disorders (nausea, diarrhea, vomiting, and constipation); hematologic disorders (anemia, neutropenia, and thrombocytopenia); respiratory disorders (cough and dyspnea); nervous system disorders (somnolence and dizziness); and pyrexia.
The most common adverse reactions in adults by Body System, including severe or life-threatening adverse reactions (NCI CTCAE Grade 3 or Grade 4) and fatal adverse reactions (Grade 5) are shown in Table 1.
| Table 1. Most Commonly Reported (≥ 5% Overall) Adverse Reactions in Adult Patients Treated With 1500 mg/m 2 of Nelarabine Injection Administered Intravenously Over 2 Hours on Days 1, 3, and 5 Repeated Every 21 Days | ||||
| Body System Adverse Reaction | Percentage of Patients (N = 103) | |||
| Toxicity Grade | ||||
| Grade 3 % | Grades 4 and 5 a % | All Grades % | ||
| Blood and Lymphatic System Disorders | ||||
| Anemia | 20 | 14 | 99 | |
| Thrombocytopenia | 37 | 22 | 86 | |
| Neutropenia | 14 | 49 | 81 | |
| Febrile neutropenia | 9 | 1 | 12 | |
| Cardiac Disorders | ||||
| Sinus tachycardia | 1 | 0 | 8 | |
| Gastrointestinal Disorders | ||||
| Nausea | 0 | 0 | 41 | |
| Diarrhea | 1 | 0 | 22 | |
| Vomiting | 1 | 0 | 22 | |
| Constipation | 1 | 0 | 21 | |
| Abdominal pain | 1 | 0 | 9 | |
| Stomatitis | 1 | 0 | 8 | |
| Abdominal distension | 0 | 0 | 6 | |
| General Disorders and Administration Site Conditions | ||||
| Fatigue | 10 | 2 | 50 | |
| Pyrexia | 5 | 0 | 23 | |
| Asthenia | 0 | 1 | 17 | |
| Edema, peripheral | 0 | 0 | 15 | |
| Edema | 0 | 0 | 11 | |
| Pain | 3 | 0 | 11 | |
| Rigors | 0 | 0 | 8 | |
| Gait, abnormal | 0 | 0 | 6 | |
| Chest pain | 0 | 0 | 5 | |
| Noncardiac chest pain | 0 | 1 | 5 | |
| Infections | ||||
| Infection | 2 | 1 | 9 | |
| Pneumonia | 4 | 1 | 8 | |
| Sinusitis | 1 | 0 | 7 | |
| Hepatobiliary Disorders | ||||
| AST increased | 1 | 1 | 6 | |
| Metabolism and Nutrition Disorders | ||||
| Anorexia | 0 | 0 | 9 | |
| Dehydration | 3 | 1 | 7 | |
| Hyperglycemia | 1 | 0 | 6 | |
| Musculoskeletal and Connective Tissue Disorders | ||||
| Myalgia | 1 | 0 | 13 | |
| Arthralgia | 1 | 0 | 9 | |
| Back pain | 0 | 0 | 8 | |
| Muscular weakness | 5 | 0 | 8 | |
| Pain in extremity | 1 | 0 | 7 | |
| Nervous System Disorders (see Table 2) | ||||
| Psychiatric Disorders | ||||
| Confusional state | 2 | 0 | 8 | |
| Insomnia | 0 | 0 | 7 | |
| Depression | 1 | 0 | 6 | |
| Respiratory, Thoracic, and Mediastinal Disorders | ||||
| Cough | 0 | 0 | 25 | |
| Dyspnea | 4 | 2 | 20 | |
| Pleural effusion | 5 | 1 | 10 | |
| Epistaxis | 0 | 0 | 8 | |
| Dyspnea, exertional | 0 | 0 | 7 | |
| Wheezing | 0 | 0 | 5 | |
| Vascular Disorders | ||||
| Petechiae | 2 | 0 | 12 | |
| Hypotension | 1 | 1 | 8 | |
| Abbreviation: AST, aspartate transaminase. a Five (5) patients had a fatal adverse reaction. Fatal adverse reactions included hypotension (n = 1), respiratory arrest (n = 1), pleural effusion/pneumothorax (n = 1), pneumonia (n = 1), and cerebral hemorrhage/coma/leukoencephalopathy (n = 1). | ||||
Other Adverse Reactions: Blurred vision was also reported in 4% of adult patients.
There was a single report of biopsy-confirmed progressive multifocal leukoencephalopathy in the adult patient population.
Neurologic Adverse Reactions: Nervous system adverse reactions, were reported for 76% of adult patients across the Phase I and Phase II trials. The most common neurologic adverse reactions (≥ 2%) in adult patients including all grades (NCI CTCAE) are shown in Table 2.
| Table 2. Neurologic Adverse Reactions (≥ 2%) in Adult Patients Treated With 1500 mg/m 2 of Nelarabine Injection Administered Intravenously Over 2 Hours on Days 1, 3, and 5 Repeated Every 21 Days | |||||
| Nervous System Disorders Adverse Reaction | Percentage of Patients (N =103) | ||||
| Grade 1 % | Grade 2 % | Grade 3 % | Grade 4 % | All Grades % | |
| Somnolence | 20 | 3 | 0 | 0 | 23 |
| Dizziness | 14 | 8 | 0 | 0 | 21 |
| Peripheral neurologic disorders, any adverse reaction | 8 | 12 | 2 | 0 | 21 |
| - Neuropathy | 0 | 4 | 0 | 0 | 4 |
| - Peripheral neuropathy | 2 | 2 | 1 | 0 | 5 |
| - Peripheral motor neuropathy | 3 | 3 | 1 | 0 | 7 |
| - Peripheral sensory neuropathy | 7 | 6 | 0 | 0 | 13 |
| Hypoesthesia | 5 | 10 | 2 | 0 | 17 |
| Headache | 11 | 3 | 1 | 0 | 15 |
| Paresthesia | 11 | 4 | 0 | 0 | 15 |
| Ataxia | 1 | 6 | 2 | 0 | 9 |
| Depressed level of consciousness | 4 | 1 | 0 | 1 | 6 |
| Tremor | 2 | 3 | 0 | 0 | 5 |
| Amnesia | 2 | 1 | 0 | 0 | 3 |
| Dysgeusia | 2 | 1 | 0 | 0 | 3 |
| Balance disorder | 1 | 1 | 0 | 0 | 2 |
| Sensory loss | 0 | 2 | 0 | 0 | 2 |
One patient had a fatal neurologic adverse reaction, cerebral hemorrhage/coma/leukoencephalopathy.
Most nervous system adverse reactions in the adult patients were evaluated as Grade 1 or 2. The additional Grade 3 adverse reactions in adult patients, were aphasia, convulsion, hemiparesis, and loss of consciousness, each reported in 1 patient (1%). The additional Grade 4 adverse reactions were cerebral hemorrhage, coma, intracranial hemorrhage, leukoencephalopathy, and metabolic encephalopathy, each reported in one patient (1%).
The other neurologic adverse reactions reported as Grade 1, 2, or unknown in adult patients were abnormal coordination, burning sensation, disturbance in attention, dysarthria, hyporeflexia, neuropathic pain, nystagmus, peroneal nerve palsy, sciatica, sensory disturbance, sinus headache, and speech disorder, each reported in one patient (1%).
Pediatric Patient : The safety profile for children is based on data from 84 pediatric patients treated with the recommended dose and schedule in a T-ALL/T-LBL treatment trial.
The most common adverse reactions in pediatric patients were hematologic disorders (anemia, leukopenia, neutropenia, and thrombocytopenia). Of the non-hematologic adverse reactions in pediatric patients, the most frequent adverse reactions reported were headache, increased transaminase levels, decreased blood potassium, decreased blood albumin, increased blood bilirubin, and vomiting.
The most common adverse reactions in pediatric patients by System Organ Class including severe or life threatening adverse reactions (NCI CTCAE Grade 3 or Grade 4) and fatal adverse reactions (Grade 5) are shown in Table 3.
| Table 3. Most Commonly Reported (≥ 5% Overall) Adverse Reactions in Pediatric Patients Treated With 650 mg/m 2 of Nelarabine Injection Administered Intravenously Over 1 Hour Daily for 5 Consecutive Days Repeated Every 21 Days | |||
| Body System Adverse Reaction | Percentage of Patients (N = 84) | ||
| Toxicity Grade | |||
| Grade 3 % | Grade 4 and 5 a % | All Grades % | |
| Blood and Lymphatic System Disorders | |||
| Anemia | 45 | 10 | 95 |
| Neutropenia | 17 | 62 | 94 |
| Thrombocytopenia | 27 | 32 | 88 |
| Leukopenia | 14 | 7 | 38 |
| Hepatobiliary Disorders | |||
| Transaminases increased | 4 | 0 | 12 |
| Blood albumin decreased | 5 | 1 | 10 |
| Blood bilirubin increased | 7 | 2 | 10 |
| Metabolic/Laboratory | |||
| Blood potassium decreased | 4 | 2 | 11 |
| Blood calcium decreased | 1 | 1 | 8 |
| Blood creatinine increased | 0 | 0 | 6 |
| Blood glucose decreased | 4 | 0 | 6 |
| Blood magnesium decreased | 2 | 0 | 6 |
| Nervous System Disorders (see Table 4) | |||
| Gastrointestinal Disorders | |||
| Vomiting | 0 | 0 | 10 |
| General Disorders & Administration Site Conditions | |||
| Asthenia | 1 | 0 | 6 |
| Infections & Infestations | |||
| Infection | 2 | 1 | 5 |
| a Three (3) patients had a fatal adverse reaction. Fatal adverse reactions included neutropenia and pyrexia (n = 1), status epilepticus/seizure (n = 1), and fungal pneumonia (n = 1). | |||
Neurologic Adverse Reactions: Nervous system adverse reactions were reported for 42% of pediatric patients across the Phase I and Phase II trials. The most common neurologic adverse reactions (≥ 2%) in pediatric patients including all grades (NCI CTCAE) are shown in Table 4.
| Table 4. Neurologic Adverse Reactions (≥ 2%) in Pediatric Patients Treated With 650 mg/m 2 of Nelarabine Injection Administered Intravenously Over 1 Hour Daily for 5 Consecutive Days Repeated Every 21 Days | |||||
| Nervous System Disorders Adverse Reaction | Percentage of Patients (N = 84) | ||||
| Grade 1 % | Grade 2 % | Grade 3 % | Grade 4 and 5 a % | All Grades % | |
| Headache | 8 | 2 | 4 | 2 | 17 |
| Peripheral neurologic disorders, any adverse reaction | 1 | 4 | 7 | 0 | 12 |
| - Peripheral neuropathy | 0 | 4 | 2 | 0 | 6 |
| - Peripheral motor neuropathy | 1 | 0 | 2 | 0 | 4 |
| - Peripheral sensory neuropathy | 0 | 0 | 6 | 0 | 6 |
| Somnolence | 1 | 4 | 1 | 1 | 7 |
| Hypoesthesia | 1 | 1 | 4 | 0 | 6 |
| Seizures | 0 | 0 | 0 | 6 | 6 |
| - Convulsions | 0 | 0 | 0 | 3 | 4 |
| - Grand mal convulsions | 0 | 0 | 0 | 1 | 1 |
| - Status epilepticus | 0 | 0 | 0 | 1 | 1 |
| Motor dysfunction | 1 | 1 | 1 | 0 | 4 |
| Nervous system disorder | 1 | 2 | 0 | 0 | 4 |
| Paresthesia | 0 | 2 | 1 | 0 | 4 |
| Tremor | 1 | 2 | 0 | 0 | 4 |
| Ataxia | 1 | 0 | 1 | 0 | 2 |
| a One (1) patient had a fatal neurologic adverse reaction, status epilepticus. | |||||
The other Grade 3 neurologic adverse reaction in pediatric patients was hypertonia reported in 1 patient (1%). The additional Grade 4 neurologic adverse reactions, were third nerve paralysis, and sixth nerve paralysis, each reported in 1 patient (1%).
The other neurologic adverse reactions reported as Grade 1, 2, or unknown in pediatric patients were dysarthria, encephalopathy, hydrocephalus, hyporeflexia, lethargy, mental impairment, paralysis, and sensory loss, each reported in 1 patient (1%).
Nelarabine Injection in Combination with Multi-Agent Chemotherapy in T-ALL and T-LBL
Nelarabine injection was studied in combination with multi-agent chemotherapy in a randomized clinical trial [NCT00408005]. The safety population in this trial included 804 patients with newly-diagnosed T-ALL (85%) or T-LBL (15%) treated with (n = 411) or without (n =393) nelarabine injection in combination with the augmented Berlin-Frankfurt-Münster chemotherapy regimen (aBFM) after initial induction therapy. Patients assigned to nelarabine injection received 650 mg/m 2 intravenously over 1 hour daily for 5 consecutive days, during consolidation Days 1 to 5 and 43 to 47, delayed intensification Days 29 to 33, and during the initial 3 courses of maintenance Days 29 to 33. The median age on enrollment was 9.5 years (range: 1-29), the majority of patients were male (73%) and white (69%). Sixty-five percent of patients assigned to the nelarabine injection arms received at least 85% of the planned dose through the third course of maintenance therapy compared to 79% of patients on the control arms who received 3 courses of maintenance therapy.
There was one fatal neurological adverse reaction in the nelarabine injection arm. The incidence of the following Grades 3 and 4 adverse reactions were higher in the nelarabine injection treated arms compared to the control arms: abnormal transaminases, motor and sensory neuropathy, nausea and vomiting, and dehydration. The incidence of seizures of any grade was 3% (14 of 411). Rhabdomyolysis was diagnosed in 2% (7 of 411) of nelarabine injection treated patients and occurred after the first course of nelarabine injection during the consolidation phase of therapy.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of nelarabine injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Infections and Infestations : Fatal opportunistic infections
Metabolism and Nutrition Disorders : Tumor lysis syndrome
Nervous System Disorders : Demyelination and ascending peripheral neuropathies similar in appearance to Guillain-Barré syndrome
Musculoskeletal and Connective Disorders : Rhabdomyolysis, blood creatine phosphokinase increased
7 DRUG INTERACTIONS
Administration of nelarabine injection in combination with adenosine deaminase (ADA) inhibitors, such as pentostatin, is not recommended [see Clinical Pharmacology (12.3) ].
11 DESCRIPTION
Nelarabine is a prodrug of the cytotoxic deoxyguanosine analogue, 9-β- D -arabinofuranosylguanine (ara-G).
The chemical name for nelarabine is 2-amino-9-β- D -arabinofuranosyl-6-methoxy-9 H -purine. It has the molecular formula C 11 H 15 N 5 O 5 and a molecular weight of 297.27. Nelarabine has the following structural formula:
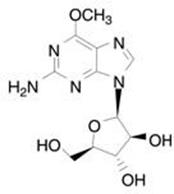
Nelarabine is slightly soluble to soluble in water and melts with decomposition between 209°C and 217°C.
Nelarabine injection is supplied as a clear, colorless, sterile solution in glass single-dose vials. Each vial contains 250 mg of nelarabine (5 mg nelarabine per mL) and the inactive ingredient sodium chloride (4.5 mg per mL) in 50 mL Water for Injection, USP. Nelarabine injection is intended for intravenous infusion.
Hydrochloric acid and sodium hydroxide may have been used to adjust the pH. The solution pH ranges from 5.0 to 7.0.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Nelarabine is a prodrug of the deoxyguanosine analogue 9-β- D -arabinofuranosylguanine (ara-G), a nucleoside metabolic inhibitor. Nelarabine is demethylated by ADA to ara-G, mono-phosphorylated by deoxyguanosine kinase and deoxycytidine kinase, and subsequently converted to the active 5’-triphosphate, ara-GTP. Accumulation of ara-GTP in leukemic blasts allows for incorporation into deoxyribonucleic acid (DNA), leading to inhibition of DNA synthesis and cell death. Other mechanisms may contribute to the cytotoxic and systemic toxicity of nelarabine.
12.3 Pharmacokinetics
Absorption : Following intravenous administration of nelarabine to adult patients with refractory leukemia or lymphoma, plasma ara-G C max values generally occurred at the end of the nelarabine infusion and were generally higher than nelarabine C max values, suggesting rapid and extensive conversion of nelarabine to ara-G. Mean plasma nelarabine and ara-G C max values were 5.0 ± 3.0 mcg/mL and 31.4 ± 5.6 mcg/mL, respectively, after a 1500 mg/m 2 nelarabine dose infused over 2 hours in adult patients. The area under the concentration-time curve (AUC) of ara-G is 37 times higher than that for nelarabine on Day 1 after nelarabine IV infusion of 1500 mg/m 2 dose (162 ± 49 mcg•h/mL versus 4.4 ± 2.2 mcg•h/mL, respectively). Comparable C max and AUC values were obtained for nelarabine between Days 1 and 5 at the nelarabine adult dosage of 1500 mg/m 2 , indicating that nelarabine does not accumulate after multiple-dosing. There are not enough ara-G data to make a comparison between Day 1 and Day 5. After a nelarabine adult dose of 1500 mg/m 2 , intracellular C max for ara-GTP appeared within 3 to 25 hours on Day 1. Exposure (AUC) to intracellular ara-GTP was 532 times higher than that for nelarabine and 14 times higher than that for ara-G (2,339 ± 2,628 mcg•h/mL versus 4.4 ± 2.2 mcg•h/mL and 162 ± 49 mcg•h/mL, respectively). Because the intracellular levels of ara-GTP were so prolonged, its elimination half-life could not be accurately estimated.
Distribution : Nelarabine and ara-G are extensively distributed throughout the body. For nelarabine, V SS values were 197 ± 216 L/m 2 in adult patients. For ara-G, V SS /F values were 50 ± 24 L/m 2 in adult patients.
Nelarabine and ara-G are not substantially bound to human plasma proteins (< 25%) in vitro , and binding is independent of nelarabine or ara-G concentrations up to 600 μM.
Metabolism : The principal route of metabolism for nelarabine is O-demethylation by ADA to form ara-G, which undergoes hydrolysis to form guanine. In addition, some nelarabine is hydrolyzed to form methylguanine, which is O-demethylated to form guanine. Guanine is N-deaminated to form xanthine, which is further oxidized to yield uric acid.
Excretion : Nelarabine and ara-G are partially eliminated by the kidneys. Mean urinary excretion of nelarabine and ara-G was 6.6 ± 4.7% and 27 ± 15% of the administered dose, respectively, in 28 adult patients over the 24 hours after nelarabine infusion on Day 1. Renal clearance averaged 24 ± 23 L/h for nelarabine and 6.2 ± 5.0 L/h for ara-G in 21 adult patients. Combined Phase I pharmacokinetic data at nelarabine doses of 199 to 2900 mg/m 2 (n = 66 adult patients) indicate that the mean clearance (CL) of nelarabine is 197 ± 189 L/h/m 2 on Day 1. The apparent clearance of ara-G (CL/F) is 10.5 ± 4.5 L/h/m 2 on Day 1. Nelarabine and ara-G are rapidly eliminated from plasma with a mean half-life of 18 minutes and 3.2 hours, respectively, in adult patients.
Pediatrics : No pharmacokinetic data are available in pediatric patients at the once-daily 650 mg/m 2 nelarabine dosage. Combined Phase I pharmacokinetic data at nelarabine doses of 104 to 2900 mg/m 2 indicate that the mean clearance (CL) of nelarabine is about 30% higher in pediatric patients than in adult patients (259 ± 409 L/h/m 2 versus 197 ± 189 L/h/m 2 , respectively) (n = 66 adults, n = 22 pediatric patients) on Day 1. The apparent clearance of ara-G (CL/F) is comparable between the 2 groups (10.5 ± 4.5 L/h/m 2 in adult patients and 11.3 ± 4.2 L/h/m 2 in pediatric patients) on Day 1. Nelarabine and ara-G are extensively distributed throughout the body. For nelarabine, V SS values were 213 ± 358 L/m 2 in pediatric patients. For ara-G, V SS /F values were 33 ± 9.3 L/m 2 in pediatric patients. Nelarabine and ara-G are rapidly eliminated from plasma in pediatric patients, with a half-life of 13 minutes and 2 hours, respectively.
Effect of Age : Age has no effect on the pharmacokinetics of nelarabine or ara-G in adults. Decreased renal function, which is more common in the elderly, may reduce ara-G clearance [see Use in Specific Populations (8.5) ] .
Effect of Gender : Gender has no effect on nelarabine or ara-G pharmacokinetics.
Effect of Race : In general, nelarabine mean clearance and volume of distribution values tend to be higher in whites (n = 63) than in blacks (by about 10%) (n = 15). The opposite is true for ara-G; mean apparent clearance and volume of distribution values tend to be lower in whites than in blacks (by about 15% to 20%). No differences in safety or effectiveness were observed between these groups.
Effect of Renal Impairment : The pharmacokinetics of nelarabine and ara-G have not been specifically studied in renally impaired or hemodialyzed patients. Nelarabine is excreted by the kidney to a small extent (5% to 10% of the administered dose). Ara-G is excreted by the kidney to a greater extent (20% to 30% of the administered nelarabine dose). In the combined Phase I trials, patients were categorized into 3 groups: normal with CLCr greater than 80 mL/min (n = 67), mild with CLCr = 50 to 80 mL/min (n = 15), and moderate with CLCr less than 50 mL/min (n = 3). The mean apparent clearance (CL/F) of ara-G was about 15% and 40% lower in patients with mild and moderate renal impairment, respectively, than in patients with normal renal function [see Use in Specific Populations (8.6) , Dosage and Administration (2.3) ] . No differences in safety or effectiveness were observed.
Effect of Hepatic Impairment : The influence of hepatic impairment on the pharmacokinetics of nelarabine has not been evaluated [see Use in Specific Populations (8.7) ] .
Drug Interactions : Cytochrome P450: Nelarabine and ara-G did not significantly inhibit the activities of the human hepatic cytochrome P450 isoenzymes 1A2, 2A6, 2B6, 2C8, 2C9, 2C19, 2D6, or 3A4 in vitro at concentrations of nelarabine and ara-G up to 100 μM.
Fludarabine: Administration of fludarabine 30 mg/m 2 as a 30-minute infusion 4 hours before a 1200-mg/m 2 infusion of nelarabine did not affect the pharmacokinetics of nelarabine, ara-G, or ara-GTP in 12 patients with refractory leukemia.
Pentostatin: There is in vitro evidence that pentostatin is a strong inhibitor of ADA. Inhibition of ADA may result in a reduction in the conversion of the prodrug nelarabine to its active moiety and consequently in a reduction in efficacy of nelarabine and/or change in adverse reaction profile of either drug [see Drug Interactions (7) ] .
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity testing of nelarabine has not been done. However, nelarabine was mutagenic when tested in vitro in L5178Y/TK mouse lymphoma cells with and without metabolic activation. No studies have been conducted in animals to assess genotoxic potential or effects on fertility. The effect on human fertility is unknown.
14 CLINICAL STUDIES
14.1 Adult Clinical Trial in Relapsed or Refractory T-ALL and T-LBL
The safety and efficacy of nelarabine injection in adult patients were studied in a clinical trial which included 39 treated patients, 28 who had T-ALL or T-LBL that had relapsed following or was refractory to at least two prior induction regimens. A 1500-mg/m 2 dose of nelarabine injection was administered intravenously over 2 hours on Days 1, 3, and 5 repeated every 21 days. Patients who experienced signs or symptoms of Grade 2 or greater neurologic toxicity on therapy were to be discontinued from further therapy with nelarabine injection. Seventeen patients had a diagnosis of T-ALL and 11 had a diagnosis of T-LBL. For patients with ≥ 2 prior inductions, the age range was 16 to 65 years (mean: 34 years) and most patients were male (82%) and Caucasian (61%). Patients with central nervous system (CNS) disease were not eligible.
Complete response (CR) in this trial was defined as bone marrow blast counts ≤ 5%, no other evidence of disease, and full recovery of peripheral blood counts. Complete response without complete hematologic recovery (CR•) was also assessed. The results of the trial for patients who had received ≥ 2 prior inductions are shown in Table 5.
| Table 5. Efficacy Results in Adult Patients With ≥ 2 Prior Inductions Treated With 1500 mg/m 2 of Nelarabine Injection Administered Intravenously Over 2 Hours on Days 1, 3, and 5 Repeated Every 21 Days | |
| N = 28 | |
| CR plus CR• % (n) [95% CI] | 21% (6) [8%, 41%] |
| - CR % (n) [95% CI] | 18% (5) [6%, 37%] |
| - CR• % (n) [95% CI] | 4% (1) [0%, 18%] |
| Duration of CR plus CR• (range in weeks) a | 4 to 195+ |
| Median overall survival (weeks) [95% CI] | 20.6 weeks [10.4, 36.4] |
| Abbreviations: CR, complete response; CI, confidence interval; CR•, complete response without hematologic recovery. a Does not include 1 patient who was transplanted (duration of response was 156+ weeks). | |
The mean number of days on therapy was 56 days (range of 10 to 136 days). Time to CR plus CR• ranged from 2.9 to 11.7 weeks.
14.2 Pediatric Clinical Trial in Relapsed or Refractory T-ALL and T-LBL
The safety and efficacy of nelarabine injection in pediatric patients were studied in a clinical trial which included patients age 21 years and younger, who had relapsed or refractory T-ALL or T-LBL. Eighty-four (84) patients, 39 of whom had received two or more prior induction regimens, were treated with 650 mg/m 2 /day of nelarabine injection administered intravenously over 1 hour daily for 5 consecutive days repeated every 21 days (see Table 6). Patients who experienced signs or symptoms of Grade 2 or greater neurologic toxicity on therapy were to be discontinued from further therapy with nelarabine injection.
| Table 6. Pediatric Clinical Trial - Patient Allocation | |
| Patient Population | N |
| Patients treated at 650 mg/m 2 /day x 5 days, every 21 days. | 84 |
| Patients with T-ALL or T-LBL with two or more prior induction treated at 650 mg/m 2 /day x 5 days, every 21 days. | 39 |
| Patients with T-ALL or T-LBL with one prior induction treated at 650 mg/m 2 /day x 5 days, every 21 days. | 31 |
| Abbreviations: T-ALL, T-cell acute lymphoblastic leukemia; T-LBL, T-cell lymphoblastic lymphoma. | |
The 84 patients ranged in age from 2.5 to 21.7 years (overall mean: 11.9 years), 52% were 3 to 12 years of age and most were male (74%) and Caucasian (62%). The majority (77%) of patients had a diagnosis of T-ALL.
Complete response (CR) in this trial was defined as bone marrow blast counts ≤ 5%, no other evidence of disease, and full recovery of peripheral blood counts. Complete response without full hematologic recovery (CR•) was also assessed as a meaningful outcome in this trial. Duration of response is reported from date of response to date of relapse, and may include subsequent stem cell transplant. Efficacy results are presented in Table 7.
| Table 7. Efficacy Results in Patients Age 21 Years and Younger at Diagnosis With ≥ 2 Prior Inductions Treated With 650 mg/m 2 of Nelarabine Injection Administered Intravenously Over 1 Hour Daily for 5 Consecutive Days Repeated Every 21 Days | |
| N = 39 | |
| CR plus CR• % (n) [95% CI] | 23% (9) [11%, 39%] |
| - CR % (n) [95% CI] | 13% (5) [4%, 27%] |
| - CR• % (n) [95% CI] | 10% (4) [3%, 24%] |
| Duration of CR plus CR• (range in weeks) a | 3.3 to 9.3 |
| Median overall survival (weeks) [95% CI] | 13.1 [8.7, 17.4] |
| Abbreviations: CR, complete response; CI, confidence interval; CR•, complete response without hematologic recovery. a Does not include 5 patients who were transplanted or had subsequent systemic chemotherapy (duration of response in these 5 patients was 4.7 to 42.1 weeks). | |
The mean number of days on therapy was 46 days (range: 7-129 days). Median time to CR plus CR• was 3.4 weeks (95% CI: 3.0, 3.7).
16 HOW SUPPLIED/STORAGE AND HANDLING
Nelarabine injection is supplied as a clear, colorless, sterile solution in Type I, clear glass single-dose vials with a gray bromobutyl rubber stopper (not made with natural rubber latex) and an aluminum seal with a white snap-off cap. Each vial contains 250 mg of nelarabine (5 mg nelarabine per mL) and the inactive ingredient sodium chloride (4.5 mg per mL) in 50 mL Water for Injection, USP.
Single-dose Vials are available in the following carton sizes: NDC 39822-0650-1 (package of 1). NDC 39822-0650-2 (package of 6).
Store nelarabine injection between 20°C and 25°C (68°F and 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). See USP Controlled Room Temperature.
12.1 Mechanism of Action
Nelarabine is a prodrug of the deoxyguanosine analogue 9-β- D -arabinofuranosylguanine (ara-G), a nucleoside metabolic inhibitor. Nelarabine is demethylated by ADA to ara-G, mono-phosphorylated by deoxyguanosine kinase and deoxycytidine kinase, and subsequently converted to the active 5’-triphosphate, ara-GTP. Accumulation of ara-GTP in leukemic blasts allows for incorporation into deoxyribonucleic acid (DNA), leading to inhibition of DNA synthesis and cell death. Other mechanisms may contribute to the cytotoxic and systemic toxicity of nelarabine.