Naloxone Hydrochloride - Naloxone Hydrochloride injection prescribing information
INDICATIONS AND USAGE
Naloxone hydrochloride injection is indicated for the complete or partial reversal of opioid depression, including respiratory depression, induced by natural and synthetic opioids including, propoxyphene, methadone and certain mixed agonist-antagonist analgesics: nalbuphine, pentazocine and butorphanol and cyclazocine. Naloxone hydrochloride is also indicated for the diagnosis of suspected or known acute opioid overdosage.
Naloxone hydrochloride injection may be useful as an adjunctive agent to increase blood pressure in the management of septic shock. (see CLINICAL PHARMACOLOGY ; Adjunctive Use in Septic Shock ).
DOSAGE AND ADMINISTRATION
Naloxone hydrochloride injection may be administered intravenously, intramuscularly, or subcutaneously. The most rapid onset of action is achieved by intravenous administration, which is recommended in emergency situations.
Since the duration of action of some opioids may exceed that of naloxone, the patient should be kept under continued surveillance. Repeated doses of naloxone should be administered, as necessary.
Intravenous Infusion
Naloxone hydrochloride injection may be diluted for intravenous infusion in normal saline or 5% dextrose solutions. The addition of 2 mg of naloxone in 500 mL of either solution provides a concentration of 0.004 mg/mL. Mixtures should be used within 24 hours. After 24 hours, the remaining unused mixture must be discarded. The rate of administration should be titrated in accordance with the patient’s response. Naloxone hydrochloride injection should not be mixed with preparations containing bisulfite, metabisulfite, long-chain or high molecular weight anions, or any solution having an alkaline pH. No drug or chemical agent should be added to naloxone hydrochloride injection unless its effect on the chemical and physical stability of the solution has first been established.
General
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
Usage in Adults
Opioid Overdose—Known or Suspected: An initial dose of 0.4 mg to 2 mg of naloxone hydrochloride may be administered intravenously. If the desired degree of counteraction and improvement in respiratory functions are not obtained, it may be repeated at two-to-three-minute intervals. If no response is observed after 10 mg of naloxone hydrochloride have been administered, the diagnosis of opioid-induced or partial opioid-induced toxicity should be questioned. Intramuscular or subcutaneous administration may be necessary if the intravenous route is not available.
Postoperative Opioid Depression: For the partial reversal of opioid depression following the use of opioids during surgery, smaller doses of naloxone hydrochloride are usually sufficient. The dose of naloxone hydrochloride should be titrated according to the patient’s response. For the initial reversal of respiratory depression, naloxone hydrochloride should be injected in increments of 0.1 to 0.2 mg intravenously at two-to three-minute intervals to the desired degree of reversal—i.e., adequate ventilationand alertness without significant pain or discomfort. Larger than necessary dosage of naloxone may result in significant reversal of analgesia and increase in blood pressure. Similarly, too rapid reversal may induce nausea, vomiting, sweating, or circulatory stress. Repeat doses of naloxone may be required within one- to two-hour intervals depending upon the amount, type (i.e., short or long acting) and time interval since last administration of opioid. Supplemental intramuscular doses have been shown to produce a longer lasting effect.
Septic Shock: The optimal dosage of naloxone hydrochloride or duration of therapy for the treatment of hypotension in septic shock patients has not been established (see CLINICAL PHARMACOLOGY ).
Usage in Children
Opioid Overdose—Known or Suspected: The usual initial dose in children is 0.01 mg/kg body weight given I.V. If this dose does not result in the desired degree of clinical improvement, a subsequent dose of 0.1 mg/kg body weight may be administered. If an l.V. route of administration is not available, naloxone may be administered I.M. or S.C. in divided doses. If necessary, naloxone hydrochloride injection can be diluted with sterile water for injection.
Postoperative Opioid Depression : Follow the recommendations and cautions under Adult Postoperative Depressio n. For the initial reversal of respiratory depression naloxone hydrochloride should be injected in increments of 0.005 mg to 0.01 mg intravenously at two- to three-minute intervals to the desired degree of reversal.
Usage in Neonates Opioid-lnduced Depression: The usual initial dose is 0.01 mg/kg body weight administered I.V., I.M., or S.C. This dose may be repeated in accordance with adult administration guidelines for postoperative opioid depression.
CONTRAINDICATIONS
Naloxone hydrochloride injection is contraindicated in patients known to be hypersensitive to it or to any of the other ingredients in naloxone hydrochloride.
ADVERSE REACTIONS
Postoperative
The following adverse events have been associated with the use of naloxone hydrochloride in postoperative patients: hypotension, hypertension, ventricular tachycardia and fibrillation, dyspnea, pulmonary edema, and cardiac arrest. Death, coma, and encephalopathy have been reported as sequelae of these events. Excessive doses of naloxone hydrochloride in postoperative patients may result in significant reversal of analgesia and may cause agitation (see PRECAUTIONS and DOSAGE AND ADMINISTRATION ; Usage in Adults-Postoperative Opioid Depression ).
Opioid Depression
Abrupt reversal of opioid depression may result in nausea, vomiting, sweating, tachycardia, increased blood pressure, tremulousness, seizures, ventricular tachycardia and fibrillation, pulmonary edema, and cardiac arrest which may result in death (see PRECAUTIONS ).
Opioid Dependence
Abrupt reversal of opioid effects in persons who are physically dependent on opioids may precipitate an acute withdrawal syndrome which may include, but is not limited to, the following signs and symptoms: body aches, fever, sweating, runny nose, sneezing, piloerection, yawning, weakness, shivering or trembling, nervousness, restlessness or irritability, diarrhea, nausea or vomiting, abdominal cramps, increased blood pressure, tachycardia. In the neonate, opioid withdrawal may also include: convulsions; excessive crying; hyperactive reflexes (see WARNINGS ). Adverse events associated with the postoperative use of naloxone hydrochloride are listed by organ system and in decreasing order of frequency as follows:
Cardiac Disorders: pulmonary edema, cardiac arrest or failure, tachycardia, ventricular fibrillation, and ventricular tachycardia. Death, coma, and encephalopathy have been reported as sequelae of these events. Gastrointestinal Disorders: vomiting, nausea Nervous System Disorders: convulsions, paraesthesia, grand mal convulsion Psychiatric Disorders: agitation, hallucination, tremulousness Respiratory, Thoracic and Mediastinal Disorders: dyspnea, respiratory depression, hypoxia Skin and Subcutaneous Tissue Disorders: nonspecific injection site reactions, sweating Vascular Disorders: hypertension, hypotension, hot flushes or flushing.
See also PRECAUTIONS and DOSAGE AND ADMINISTRATION ; Usage in Adults; Postoperative Opioid Depression .
Drug Interactions
Large doses of naloxone are required to antagonize buprenorphine since the latter has a long duration of action due to its slow rate of binding and subsequent slow dissociation from the opioid receptor. Buprenorphine antagonism is characterized by a gradual onset of the reversal effects and a decreased duration of action of the normally prolonged respiratory depression. The barbiturate methohexital appears to block the acute onset of withdrawal symptoms induced by naloxone in opiate addicts.
DESCRIPTION
Naloxone hydrochloride, an opioid antagonist, is a synthetic congener of oxymorphone. In structure it differs from oxymorphone in that the methyl group on the nitrogen atom is replaced by an allyl group.
NALOXONE HYDROCHLORIDE (-)-17-Allyl-4,5α-epoxy-3,14-dihydroxymorphinan-6-one hydrochloride
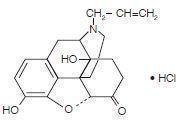
Naloxone hydrochloride occurs as a white to slightly off-white powder, and is soluble in water, in dilute acids, and in strong alkali; slightly soluble in alcohol; practically insoluble in ether and in chloroform.
Naloxone Hydrochloride Injection is available as a sterile solution for intravenous, intramuscular and subcutaneous administration in 1 mg/mL concentration. pH is adjusted to 3.5 ± 0.5 with hydrochloric acid. Each mL also contains 8.35 mg of sodium chloride. Naloxone Hydrochloride Injection is preservative-free.
CLINICAL PHARMACOLOGY
Complete or Partial Reversal of Opioid Depression
Naloxone hydrochloride prevents or reverses the effects of opioids including respiratory depression, sedation, and hypotension. Also, it can reverse the psychotomimetic and dysphoric effects of agonist-antagonist such as pentazocine.
Naloxone hydrochloride is an essentially pure opioid antagonist, i.e., it does not possess the “agonistic” or morphine-like properties characteristic of other opioid antagonists. When administered in usual doses and in the absence of opioids or agonistic effects of other opioid antagonists, it exhibits essentially no pharmacologic activity.
Naloxone hydrochloride has not been shown to produce tolerance or cause physical or psychological dependence. In the presence of physical dependence on opioids, naloxone hydrochloride will produce withdrawal symptoms. However, in the presence of opioid dependence, opiate withdrawal symptoms may appear within minutes of naloxone hydrochloride administration and subside in about 2 hours. The severity and duration of the withdrawal syndrome are related to the dose of naloxone hydrochloride and to the degree and type of opioid dependence. While the mechanism of action of naloxone hydrochloride is not fully understood, in vitro evidence suggests that naloxone hydrochloride antagonizes opioid effects by competing for the μ , κ and σ opiate receptor sites in the CNS, with the greatest affinity for the μ receptor.
When naloxone hydrochloride is administered intravenously (I.V.), the onset of action is generally apparent within two minutes. The onset of action is slightly less rapid when it is administered subcutaneously (S.C.) or intramuscularly (I.M.). The duration of action is dependent upon the dose and route of administration of naloxone hydrochloride. Intramuscular administration produces a more prolonged effect than intravenous administration. Since the duration of action of naloxone hydrochloride may be shorter than that of some opiates, the effect of the opiate may return as the effects of naloxone hydrochloride dissipates. The requirement for repeat doses of naloxone hydrochloride will also be dependent upon the amount, type and route of administration of the opioid being antagonized.
Adjunctive Use in Septic Shock
Naloxone hydrochloride has been shown in some cases of septic shock to produce a rise in blood pressure that may last up to several hours; however, this pressor response has not been demonstrated to improve patient survival. In some studies, treatment with naloxone hydrochloride in the setting of septic shock has been associated with adverse effects, including agitation, nausea and vomiting, pulmonary edema, hypotension, cardiac arrhythmias, and seizures. The decision to use naloxone hydrochloride in septic shock should be exercised with caution, particularly in patients who may have underlying pain or have previously received opioid therapy and may have developed opioid tolerance.
Because of the limited number of patients who have been treated, optimal dosage and treatment regimens have not been established.
HOW SUPPLIED
1 mg/mL naloxone hydrochloride injection USP, for intravenous, intramuscular and subcutaneous administration.
Available as follows:
1 mg/mL
2 mL single dose disposable prefilled syringes, in the MIN-I-JET ® system with 21 G. x 11/2” needle. Shrink Wrapped Packages of 10.
NDC 76329-1469-1 Stock No. 1469 (contains no preservative)
2 mL single dose disposable Luer-Jet TM Luer-Lock Prefilled Syringe. Shrink Wrapped Packages of 10. Needle not included
NDC 76329-3369-1 Stock No. 3369 (contains no preservative)
Syringe Assembly Directions:
The MIN-I-JET ® syringe with needle, illustrated below, is the basic unit upon which all the other syringe systems are built; slight adaptations and/ or additional auxiliary parts create the other syringe systems. Assembly directions remain essentially the same.
USE ASEPTIC TECHNIQUE
Do not assemble until ready to use.
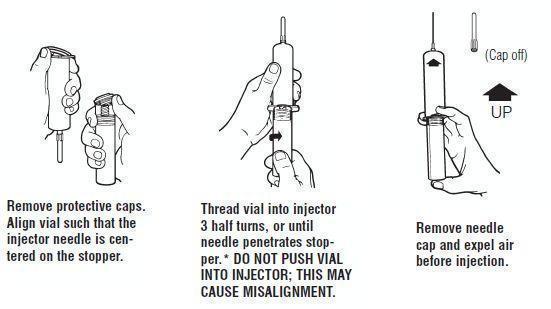
•CAUTION: IMPROPER ENGAGING MAY CAUSE GLASS BREAKAGE AND SUBSEQUENT INJURY.