Milrinone Lactate - Milrinone Lactate injection, Solution prescribing information
INDICATIONS AND USAGE
Milrinone Lactate Injection is indicated for the short-term intravenous treatment of patients with acute decompensated heart failure. Patients receiving milrinone lactate should be observed closely with appropriate electrocardiographic equipment. The facility for immediate treatment of potential cardiac events, which may include life threatening ventricular arrhythmias, must be available. The majority of experience with intravenous milrinone lactate has been in patients receiving digoxin and diuretics.
DOSAGE AND ADMINISTRATION
Milrinone Lactate Injection, USP should be administered with a loading dose followed by a continuous infusion (maintenance dose) according to the following guidelines:
LOADING DOSE
50 mcg/kg: Administer slowly over 10 minutes
| Loading Dose (mL) Using 1 mg/mL Concentration | ||||||||||
| Patient Body Weight (kg) | ||||||||||
| kg | 30 | 40 | 50 | 60 | 70 | 80 | 90 | 100 | 110 | 120 |
| mL | 1.5 | 2 | 2.5 | 3 | 3.5 | 4 | 4.5 | 5 | 5.5 | 6 |
The loading dose may be given undiluted, but diluting to a rounded total volume of 10 or 20 mL (see Maintenance Dose for diluents) may simplify the visualization of the injection rate.
| Infusion Rate | Total Daily Dose (24 Hours) | ||
| Minimum | 0.375 mcg/kg/min | 0.59 mg/kg | Administer as a continuous intravenous infusion |
| Standard | 0.5 mcg/kg/min | 0.77 mg/kg | |
| Maximum | 0.75 mcg/kg/min | 1.13 mg/kg |
Milrinone lactate drawn from vials should be diluted prior to maintenance dose administration. The diluents that may be used are 0.45% Sodium Chloride Injection USP, 0.9% Sodium Chloride Injection USP, or 5% Dextrose Injection USP. The table below shows the volume of diluent in milliliters (mL) that must be used to achieve 200 mcg/mL concentration for infusion, and the resultant total volumes.
| Desired Infusion Concentration mcg/mL | Milrinone Lactate Injection 1 mg/mL (mL) | Diluent (mL) | Total Volume (mL) |
| 200 | 10 | 40 | 50 |
| 200 | 20 | 80 | 100 |
The infusion rate should be adjusted according to hemodynamic and clinical response. Patients should be closely monitored. In controlled clinical studies, most patients showed an improvement in hemodynamic status as evidenced by increases in cardiac output and reductions in pulmonary capillary wedge pressure.
Note: See Dosage Adjustment in Renally Impaired Patients . Dosage may be titrated to the maximum hemodynamic effect and should not exceed 1.13 mg/kg/day. Duration of therapy should depend upon patient responsiveness.
The maintenance dose in mL/hr by patient body weight (kg) may be determined by reference to the following table.
| Maintenance Dose (mcg/kg/min) | Patient Body Weight (kg) | |||||||||
| 30 | 40 | 50 | 60 | 70 | 80 | 90 | 100 | 110 | 120 | |
| 0.375 | 3.4 | 4.5 | 5.6 | 6.8 | 7.9 | 9 | 10.1 | 11.3 | 12.4 | 13.5 |
| 0.4 | 3.6 | 4.8 | 6 | 7.2 | 8.4 | 9.6 | 10.8 | 12 | 13.2 | 14.4 |
| 0.5 | 4.5 | 6 | 7.5 | 9 | 10.5 | 12 | 13.5 | 15 | 16.5 | 18 |
| 0.6 | 5.4 | 7.2 | 9 | 10.8 | 12.6 | 14.4 | 16.2 | 18 | 19.8 | 21.6 |
| 0.7 | 6.3 | 8.4 | 10.5 | 12.6 | 14.7 | 16.8 | 18.9 | 21 | 23.1 | 25.2 |
| 0.75 | 6.8 | 9 | 11.3 | 13.5 | 15.8 | 18 | 20.3 | 22.5 | 24.8 | 27 |
When administering milrinone lactate by continuous infusion, it is advisable to use a calibrated electronic infusion device.
Intravenous drug products should be inspected visually and should not be used if particulate matter or discoloration is present.
Dosage Adjustment in Renally Impaired Patients
Data obtained from patients with severe renal impairment (creatinine clearance = 0 to 30 mL/min) but without congestive heart failure have demonstrated that the presence of renal impairment significantly increases the terminal elimination half-life of milrinone lactate. Reductions in infusion rate may be necessary in patients with renal impairment. For patients with clinical evidence of renal impairment, the recommended infusion rate can be obtained from the following table:
| Creatinine Clearance (mL/min/1.73m 2 ) | Infusion Rate (mcg/kg/min) |
| 5 | 0.2 |
| 10 | 0.23 |
| 20 | 0.28 |
| 30 | 0.33 |
| 40 | 0.38 |
| 50 | 0.43 |
CONTRAINDICATIONS
Milrinone Lactate Injection is contraindicated in patients who are hypersensitive to it.
ADVERSE REACTIONS
Cardiovascular Effects
In patients receiving milrinone lactate in Phase II and III clinical trials, ventricular arrhythmias were reported in 12.1%: Ventricular ectopic activity, 8.5%; nonsustained ventricular tachycardia, 2.8%; sustained ventricular tachycardia, 1% and ventricular fibrillation, 0.2% (2 patients experienced more than one type of arrhythmia). Holter recordings demonstrated that in some patients injection of milrinone lactate increased ventricular ectopy, including nonsustained ventricular tachycardia. Life-threatening arrhythmias were infrequent and when present have been associated with certain underlying factors such as preexisting arrhythmias, metabolic abnormalities (e.g. hypokalemia), abnormal digoxin levels and catheter insertion. Milrinone lactate was not shown to be arrhythmogenic in an electrophysiology study. Supraventricular arrhythmias were reported in 3.8% of the patients receiving milrinone lactate. The incidence of both supraventricular and ventricular arrhythmias has not been related to the dose or plasma milrinone concentration.
Other cardiovascular adverse reactions include hypotension, 2.9% and angina/chest pain, 1.2%.
In the post marketing experience, there have been rare cases of “torsades de pointes” reported.
CNS Effects
Headaches, usually mild to moderate in severity, have been reported in 2.9% of patients receiving milrinone lactate.
Other Effects
Other adverse reactions reported, but not definitely related to the administration of milrinone lactate include hypokalemia, 0.6%; tremor, 0.4%; and thrombocytopenia, 0.4%.
Isolated spontaneous reports of bronchospasm and anaphylactic shock have been received; and in the post-marketing experience, liver function test abnormalities and skin reactions such as rash have been reported.
Post-Marketing Adverse Event Reports
In addition to adverse events reported from clinical trials, the following events have been reported from worldwide post-marketing experience with milrinone: Isolated spontaneous reports of bronchospasm and anaphylactic shock. Liver function test abnormalities and skin reactions such as rash. Administration site conditions: Infusion site reaction. To report SUSPECTED ADVERSE REACTIONS, contact Mullan Pharmaceutical Inc., at 1-800-673-9839 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
Milrinone lactate injection is a member of a class of bipyridine inotropic/vasodilator agents with phosphodiesterase inhibitor activity, distinct from digitalis glycosides or catecholamines. Milrinone lactate is designated chemically as 1,6-dihydro-2-methyl-6-oxo-[3,4'-bipyridine]-5-carbonitrile lactate and has the following structure:
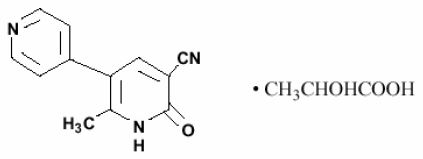
Milrinone is an off-white to tan crystalline compound with a molecular weight of 211.2 and an empirical formula of C 12 H 9 N 3 O. It is slightly soluble in methanol, and very slightly soluble in chloroform and in water. As the lactate salt, it is stable and colorless to pale yellow in solution. Milrinone lactate is available as sterile aqueous solutions of the lactate salt of milrinone for injection or infusion intravenously.
Sterile, single-dose vials: Single-dose vials of 10 and 20 mL contain in each mL milrinone lactate equivalent to 1 mg milrinone and 47 mg Dextrose, Anhydrous, USP, in Water for Injection, USP. The pH is adjusted to between 3.2 and 4.0 with lactic acid or sodium hydroxide. The total concentration of lactic acid can vary between 0.95 mg/mL and 1.29 mg/mL. These vials require preparation of dilutions prior to administration to patients intravenously.
CLINICAL PHARMACOLOGY
Milrinone lactate is a positive inotrope and vasodilator, with little chronotropic activity different in structure and mode of action from either the digitalis glycosides or catecholamines.
Milrinone lactate, at relevant inotropic and vasorelaxant concentrations, is a selective inhibitor of peak III cAMP phosphodiesterase isozyme in cardiac and vascular muscle. This inhibitory action is consistent with cAMP mediated increases in intracellular ionized calcium and contractile force in cardiac muscle, as well as with cAMP dependent contractile protein phosphorylation and relaxation in vascular muscle. Additional experimental evidence also indicates that milrinone lactate is not a beta-adrenergic agonist nor does it inhibit sodium-potassium adenosine triphosphatase activity as do the digitalis glycosides.
Clinical studies in patients with congestive heart failure have shown that milrinone lactate produces dose-related and plasma drug concentration-related increases in the maximum rate of increase of left ventricular pressure. Studies in normal subjects have shown that milrinone lactate produces increases in the slope of the left ventricular pressure-dimension relationship, indicating a direct inotropic effect of the drug. Milrinone lactate also produces dose-related and plasma concentration-related increases in forearm blood flow in patients with congestive heart failure, indicating a direct arterial vasodilator activity of the drug.
Both the inotropic and vasodilatory effects have been observed over the therapeutic range of plasma milrinone concentrations of 100 ng/mL to 300 ng/mL.
In addition to increasing myocardial contractility, milrinone lactate improves diastolic function as evidenced by improvements in left ventricular diastolic relaxation.
The acute administration of intravenous milrinone has also been evaluated in clinical trials in excess of 1600 patients, with chronic heart failure, heart failure associated with cardiac surgery, and heart failure associated with myocardial infarction. The total number of deaths, either on therapy or shortly thereafter (24 hours) was 15, less than 0.9%, few of which were thought to be drug-related.
CLINICAL PHARMACOLOGY
Milrinone lactate is a positive inotrope and vasodilator, with little chronotropic activity different in structure and mode of action from either the digitalis glycosides or catecholamines.
Milrinone lactate, at relevant inotropic and vasorelaxant concentrations, is a selective inhibitor of peak III cAMP phosphodiesterase isozyme in cardiac and vascular muscle. This inhibitory action is consistent with cAMP mediated increases in intracellular ionized calcium and contractile force in cardiac muscle, as well as with cAMP dependent contractile protein phosphorylation and relaxation in vascular muscle. Additional experimental evidence also indicates that milrinone lactate is not a beta-adrenergic agonist nor does it inhibit sodium-potassium adenosine triphosphatase activity as do the digitalis glycosides.
Clinical studies in patients with congestive heart failure have shown that milrinone lactate produces dose-related and plasma drug concentration-related increases in the maximum rate of increase of left ventricular pressure. Studies in normal subjects have shown that milrinone lactate produces increases in the slope of the left ventricular pressure-dimension relationship, indicating a direct inotropic effect of the drug. Milrinone lactate also produces dose-related and plasma concentration-related increases in forearm blood flow in patients with congestive heart failure, indicating a direct arterial vasodilator activity of the drug.
Both the inotropic and vasodilatory effects have been observed over the therapeutic range of plasma milrinone concentrations of 100 ng/mL to 300 ng/mL.
In addition to increasing myocardial contractility, milrinone lactate improves diastolic function as evidenced by improvements in left ventricular diastolic relaxation.
The acute administration of intravenous milrinone has also been evaluated in clinical trials in excess of 1600 patients, with chronic heart failure, heart failure associated with cardiac surgery, and heart failure associated with myocardial infarction. The total number of deaths, either on therapy or shortly thereafter (24 hours) was 15, less than 0.9%, few of which were thought to be drug-related.
HOW SUPPLIED
Milrinone Lactate Injection, USP is supplied as 10 mL single-dose vials in a box of 10, NDC 83301-0016-2; as 20 mL single-dose vials box of 10, NDC 83301-0017-2, containing a sterile, clear, colorless to pale yellow solution. Each mL contains milrinone lactate equivalent to 1 mg milrinone.
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]. Avoid freezing.
Discard unused portion after initial use.
Manufactured for:
Mullan Pharmaceutical Inc.
Pasadena, CA 91101
Manufactured by: Shandong New Time Pharmaceutical Co., Ltd., No. 1, North Outer Ring Road, Feixian, Shandong 273400, China. Rev. 10/2023