Get your patient on Methyltestosterone - Methyltestosterone capsule (Methyltestosterone)
Methyltestosterone - Methyltestosterone capsule prescribing information
INDICATIONS & USAGE
1. Males
Androgens are indicated for replacement therapy in conditions associated with a deficiency or absence of endogenous testosterone:
- Primary hypogonadism (congenital or acquired) — testicular failure due to cryptorchidism, bilateral torsions, orchitis, vanishing testis syndrome; or orchidectomy.
- Hypogonadotropic hypogonadism (congenital or acquired) — gonadotropin or luteinizing hormone-releasing hormone (LHRH) deficiency, or pituitary hypothalamic injury from tumors, trauma, or radiation. (Appropriate adrenal cortical and thyroid hormone replacement therapy are still necessary, however, and are actually of primary importance.) If the above conditions occur prior to puberty, androgen replacement therapy will be needed during the adolescent years for development of secondary sexual characteristics. Prolonged androgen treatment will be required to maintain sexual characteristics in these and other males who develop testosterone deficiency after puberty. Safety and efficacy of methyltestosterone in men with “age-related hypogonadism” (also referred to as “late-onset hypogonadism”) have not been established.
- Androgens may be used to stimulate puberty in carefully selected males with clearly delayed puberty. These patients usually have a familial pattern of delayed puberty that is not secondary to a pathological disorder; puberty is expected to occur spontaneously at a relatively late date. Brief treatment with conservative doses may occasionally be justified in these patients if they do not respond to psychological support. The potential adverse effect on bone maturation should be discussed with the patient and parents prior to androgen administration. An X-ray of the hand and wrist to determine bone age should be obtained every 6 months to assess the effect of treatment on the epiphyseal centers (see WARNINGS ).
2. Females
Androgens may be used secondarily in women with advancing inoperable metastatic (skeletal) mammary cancer who are 1 to 5 years postmenopausal. Primary goals of therapy in these women include ablation of the ovaries. Other methods of counteracting estrogen activity are adrenalectomy, hypophysectomy, and/or antiestrogen therapy. This treatment has also been used in premenopausal women with breast cancer who have benefitted from oophorectomy and are considered to have a hormone-responsive tumor. Judgment concerning androgen therapy should be made by an oncologist with expertise in this field.
DOSAGE & ADMINISTRATION
Prior to initiating methyltestosterone, confirm the diagnosis of hypogonadism by ensuring that serum testosterone concentrations have been measured in the morning on at least two separate days and that these serum testosterone concentrations are below the normal range.
Methyltestosterone capsules are administered orally. The suggested dosage for androgens varies depending on the age, sex, and diagnosis of the individual patient. Dosage is adjusted according to the patient’s response and the appearance of adverse reactions.
Replacement therapy in androgen-deficient males is 10 mg to 50 mg of methyltestosterone daily. Various dosage regimens have been used to induce pubertal changes in hypogonadal males, some experts have advocated lower dosages initially, gradually increasing the dose as puberty progresses with or without a decrease to maintenance levels. Other experts emphasize that higher dosages are needed to induce pubertal changes and lower dosages can be used for maintenance after puberty. The chronological and skeletal ages must be taken into consideration both in determining the initial dose and in adjusting the dose.
Doses used in delayed puberty generally are in the lower range of that given above, and for a limited duration, for example 4 to 6 months.
Women with metastatic breast carcinoma must be followed closely because androgen therapy occasionally appears to accelerate the disease. Thus, many experts prefer to use the shorter acting androgen preparations rather than those with prolonged activity for treating breast carcinoma, particularly during the early stages of androgen therapy. The dosage of methyltestosterone for androgen therapy in breast carcinoma in females is from 50 mg to 200 mg daily.
CONTRAINDICATIONS
Androgens are contraindicated in men with carcinomas of the breast or with known or suspected carcinomas of the prostate, and in women who are or may become pregnant. When administered to pregnant women, androgens cause virilization of the external genitalia of the female fetus. This virilization includes clitoromegaly, abnormal vaginal development, and fusion of genital folds to form a scrotal-like structure. The degree of masculinization is related to the amount of drug given and the age of the fetus, and is most likely to occur in the female fetus when the drugs are given in the first trimester. If the patient becomes pregnant while taking these drugs, she should be apprised of the potential hazard to the fetus.
ADVERSE REACTIONS
Endocrine and Urogenital
Female: The most common side effects of androgen therapy are amenorrhea and other menstrual irregularities, inhibition of gonadotropin secretion and virilization, including deepening of the voice and clitoral enlargement. The latter usually is not reversible after androgens are discontinued. When administered to a pregnant woman, androgens cause virilization of external genitalia of the female fetus.
Male: Gynecomastia, and excessive frequency and duration of penile erections. Oligospermia may occur at high dosages (see CLINICAL PHARMACOLOGY ).
Skin and appendages: Hirsutism, male pattern baldness, and acne.
Cardiovascular Disorders: myocardial infarction, stroke.
Fluid and Electrolyte Disturbances: Retention of sodium, chloride, water, potassium, calcium and inorganic phosphates.
Gastrointestinal: Nausea, cholestatic jaundice, alterations in liver function tests, rarely hepatocellular neoplasms and peliosis hepatis (see WARNINGS ).
Hematologic: Suppression of clotting factors II, V, VII, and X, bleeding in patients on concomitant anticoagulant therapy and polycythemia.
Nervous System: Increased or decreased libido, headache, anxiety, depression, and generalized paresthesia.
Metabolic: Increased serum cholesterol.
Vascular Disorders: venous thromboembolism.
Miscellaneous: Rarely anaphylactoid reactions.
To report SUSPECTED ADVERSE REACTIONS, contact Novitium Pharma LLC at 1-855-204-1431 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Anticoagulants: C-17 substituted derivatives of testosterone, such as methandrostenolone, have been reported to decrease the anticoagulant requirements of patients receiving oral anticoagulants. Patients receiving oral anticoagulant therapy require close monitoring, especially when androgens are started or stopped.
- Oxyphenbutazone: Concurrent administration of oxyphenbutazone and androgens may result in elevated serum levels of oxyphenbutazone.
- Insulin: In diabetic patients the metabolic effects of androgens may decrease blood glucose and insulin requirements.
DESCRIPTION
The androgens are steroids that develop and maintain primary and secondary male sex characteristics.
Androgens are derivatives of cyclopentanoper-hydrophenanthrene. Endogenous androgens are C-19 steroids with a side chain at C-17, and with two angular methyl groups. Testosterone is the primary endogenous androgen. In their active form, all drugs in the class have a 17-beta hydroxy group. 17- alpha alkylation (methyltestosterone) increases the pharmacologic activity per unit weight compared to testosterone when given orally.
Methyltestosterone, a synthetic derivative of testosterone, is an androgenic preparation given by the oral route in a capsule form. It has the following structural formula:
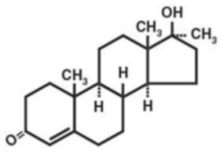
C 20 H 30 O 2 Molecular Weight: 302.46
17-β-hydroxy-17-methylandrost-4-en-3-one
Methyltestosterone, USP occurs as white or creamy white crystals or powder, which is soluble in various organic solvents but is practically insoluble in water.
Each capsule, for oral administration, contains 10 mg of methyltestosterone, USP. In addition, each capsule contains the following inactive ingredients: pregelatinized starch, lactose anhydrous, D&C Yellow #10, gelatin, FD&C Blue #1, FD&C Red #40. Additionally, the capsule imprinting ink contains strong ammonia solution, potassium hydroxide, iron oxide black, propylene glycol and shellac.
FDA approved dissolution test specifications differ from USP.
CLINICAL PHARMACOLOGY
Endogenous androgens are responsible for the normal growth and development of the male sex organs and for maintenance of secondary sex characteristics. These effects include the growth and maturation of prostate, seminal vesicles, penis, and scrotum. The development of male hair distribution, such as beard, pubic, chest, and axillary hair; laryngeal enlargement, vocal cord thickening, alterations in body musculature, and fat distribution. Drugs in this class also cause retention of nitrogen, sodium, potassium, phosphorus, and decreased urinary excretion of calcium. Androgens have been reported to increase protein anabolism and decrease protein catabolism. Nitrogen balance is improved only when there is sufficient intake of calories and protein.
Androgens are responsible for the growth spurt of adolescence and for the eventual termination of linear growth which is brought about by fusion of the epiphyseal growth centers. In children, exogenous androgens accelerate linear growth rates, but may cause a disproportionate advancement in bone maturation. Use over long periods may result in fusion of the epiphyseal growth centers and termination of growth process. Androgens have been reported to stimulate the production of red blood cells by enhancing the production of erythropoietic stimulating factor.
During exogenous administration of androgens, endogenous testosterone release is inhibited through feedback inhibition of pituitary luteinizing hormone (LH). At large doses of exogenous androgens, spermatogenesis may also be suppressed through feedback inhibition of pituitary follicle stimulating hormone (FSH).
There is a lack of substantial evidence that androgens are effective in fractures, surgery, convalescence and functional uterine bleeding.
Pharmacokinetics
Testosterone given orally is metabolized by the gut and 44 percent is cleared by the liver of the first pass. Oral doses as high as 400 mg per day are needed to achieve clinically effective blood levels for full replacement therapy. The synthetic androgen, methyltestosterone, is less extensively metabolized by the liver and has a longer half-life. It is more suitable than testosterone for oral administration.
Testosterone in plasma is 98 percent bound to a specific testosterone-estradiol binding globulin, and about 2 percent is free. Generally, the amount of this sex-hormone binding globulin in the plasma will determine the distribution of testosterone between free and bound forms, and the free testosterone concentration will determine its half-life.
About 90 percent of a dose of testosterone is excreted in the urine as glucuronic and sulfuric acid conjugates of testosterone and its metabolites; and 6 percent of a dose is excreted in the feces, mostly in the unconjugated form. Inactivation of testosterone occurs primarily in the liver. Testosterone is metabolized to various 17-keto steroids through two different pathways. There are considerable variations of the half-life of testosterone as reported in the literature, ranging from 10 to 100 minutes.
In many tissues the activity of testosterone appears to depend on reduction to dihydrotestosterone, which binds to cytosol receptor proteins. The steroid-receptor complex is transported to the nucleus where it initiates transcription events and cellular changes related to androgen action.
HOW SUPPLIED
Methyltestosterone Capsules USP, 10 mg are hard gelatin capsules with red translucent body and cap imprinted “novitium 10 mg” on the body and “255” on the cap in black ink; containing white to off-white powder.
They are available as follows:
Bottles of 100: NDC 70954-255-10
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Dispense in a tightly closed, light-resistant container as defined in the USP, with a child-resistant closure, as required.
Manufactured By:
Novitium Pharma LLC
70 Lake Drive, East Windsor
New Jersey 08520
Issued: 05/2025
LB4274-01