Get your patient on Methylergonovine Maleate - Methylergonovine Maleate injection, Solution (Methylergonovine Maleate)
Methylergonovine Maleate - Methylergonovine Maleate injection, Solution prescribing information
INDICATIONS AND USAGE
Following delivery of the placenta, for routine management of uterine atony, hemorrhage and subinvolution of the uterus. For control of uterine hemorrhage in the second stage of labor following delivery of the anterior shoulder.
DOSAGE AND ADMINISTRATION
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration.
Intramuscularly
1 mL, 0.2 mg, after delivery of the anterior shoulder, after delivery of the placenta, or during the puerperium. May be repeated as required, at intervals of 2-4 hours.
Intravenously
1 mL, 0.2 mg, administered slowly over a period of no less than 60 seconds (See WARNINGS ).
CONTRAINDICATIONS
Hypertension; toxemia; pregnancy; and hypersensitivity.
ADVERSE REACTIONS
Clinical trials experience
Common Adverse Reactions
The most common adverse reaction is hypertension associated in several cases with seizure and/or headache. Hypotension has also been reported. Abdominal pain (caused by uterine contractions), nausea and vomiting have occurred occasionally.
Rare Adverse Reactions
Rarely observed reactions have included: acute myocardial infarction, transient chest pains, vasoconstriction, vasospasm, coronary arterial spasm, bradycardia, tachycardia, dyspnea, hematuria, thrombophlebitis, water intoxication, hallucinations, leg cramps, dizziness, tinnitus, nasal congestion, diarrhea, diaphoresis, palpitation, rash, and foul taste.
There have been rare isolated reports of anaphylaxis, without a proven causal relationship to the drug product.
Post marketing Experience
The following adverse drug reactions have been derived from post-marketing experience with Methylergonovine Maleate Injection via spontaneous case reports. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency which is therefore categorized as not known.
Nervous system disorders
Cerebrovascular accident, paraesthesia
Cardiac disorders
Ventricular fibrillation, ventricular tachycardia, angina pectoris, atrioventricular block
Drug Interactions
CYP 3A4 Inhibitors (e.g., Macrolide Antibiotics and Protease Inhibitors)
There have been rare reports of serious adverse events in connection with the coadministration of certain ergot alkaloid drugs (e.g., dihydroergotamine and ergotamine) and potent CYP 3A4 inhibitors, resulting in vasospasm leading to cerebral ischemia and/or ischemia of the extremities. Although there have been no reports of such interactions with methylergonovine alone, strong and moderate CYP 3A4 inhibitors should not be co-administered with methylergonovine. Examples of some of the strong CYP 3A4 inhibitors include saquinavir, grapefruit juice, nefazodone, macrolide antibiotics (e.g., troleandomycin, clarithromycin), HIV protease or reverse transcriptase inhibitors (e.g., ritonavir, indinavir, nelfinavir) or azole antifungals (e.g., ketoconazole, itraconazole, voriconazole). Moderate inhibitors include fluconazole, fluvoxamine and clotrimazole. Weak CYP 3A4 inhibitors should be administered with caution. Weak inhibitors include chlorzoxazone, cilostazol, and ranitidine. These lists are not exhaustive, and the prescriber should consider the effects on CYP 3A4 of other agents being considered for concomitant use with methylergonovine.
CYP3A4 inducers
Drugs (e.g. nevirapine, rifampin) that are strong inducers of CYP3A4 are likely to decrease the pharmacological action of Methylergonovine Maleate Injection.
Beta-blockers
Caution should be exercised when Methylergonovine Maleate Injection is used concurrently with beta-blockers. Concomitant administration with beta-blockers may enhance the vasoconstrictive action of ergot alkaloids.
Anesthetics
Anesthetics like halothane and methoxyflurane may reduce the oxytocic potency of Methylergonovine Maleate Injection.
Glyceryl trinitrate and other antianginal drugs
Methylergonovine maleate produces vasoconstriction and can be expected to reduce the effect of glyceryl trinitrate and other antianginal drugs.
No pharmacokinetic interactions involving other cytochrome P450 isoenzymes are known.
Caution should be exercised when Methylergonovine Maleate Injection is used concurrently with other vasoconstrictors, ergot alkaloids, or prostaglandins.
DESCRIPTION
Methylergonovine Maleate Injection, USP is a semi-synthetic ergot alkaloid used for the prevention and control of postpartum hemorrhage.
Methylergonovine Maleate Injection, USP is available in sterile ampules of 1 mL, containing 0.2 mg methylergonovine maleate for intramuscular or intravenous injection.
Ampules: 1 mL, clear, colorless solution. Active Ingredient: methylergonovine maleate, USP, 0.2 mg. Inactive Ingredients: maleic acid, 0.10 mg; sodium chloride, 7.0 mg; water for injection, qs to 1 mL.
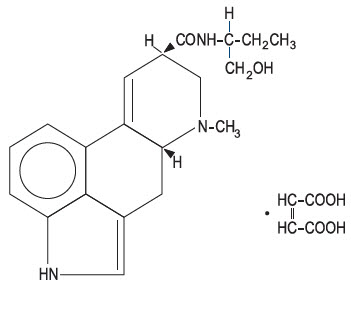
Chemically, methylergonovine maleate is designated as ergoline-8-carboxamide, 9,10-didehydro- N -[l-(hydroxymethyl)propyl]-6-methyl-, [8β( S )]-, ( Z )-2-butenedioate (1:1) (salt). Its structural formula is
 |
| C 20 H 25 N 3 O 2 ∙C 4 H 4 O 4 Mol. wt. - 455.51 |
CLINICAL PHARMACOLOGY
Mechanism of Action
Methylergonovine maleate acts directly on the smooth muscle of the uterus and increases the tone, rate, and amplitude of rhythmic contractions. Thus, it induces a rapid and sustained tetanic uterotonic effect which shortens the third stage of labor and reduces blood loss.
Pharmacodynamics
The onset of action after IV administration is immediate; after I.M. administration, 2-5 minutes, and after oral administration, 5-10 minutes.
Pharmacokinetics
Pharmacokinetic studies following an IV injection have shown that methylergonovine is rapidly distributed from plasma to peripheral tissues within 2-3 minutes or less. The bioavailability after oral administration was reported to be about 60% with no accumulation after repeated doses. During delivery, with intramuscular injection, bioavailability increased to 78%. Ergot alkaloids are mostly eliminated by hepatic metabolism and excretion, and the decrease in bioavailability following oral administration is probably a result of first-pass metabolism in the liver.
Bioavailability studies conducted in fasting healthy female volunteers have shown that oral absorption of a 0.2 mg methylergonovine tablet was fairly rapid with a mean peak plasma concentration of 3243 ± 1308 pg/mL observed at 1.12 ± 0.82 hours. A delayed gastrointestinal absorption (T max about 3 hours) of methylergonovine maleate tablet might be observed in postpartum women during continuous treatment with this oxytocic agent.
For a 0.2 mg intramuscular (IM) injection, a mean peak plasma concentration of 5918 ± 1952 pg/mL was observed at 0.41 ± 0.21 hours. The extent of absorption of the tablet, based upon methylergonovine plasma concentrations, was found to be equivalent to that of the IM solution given orally, and the extent of oral absorption of the IM solution was proportional to the dose following administration of 0.1, 0.2, and 0.4 mg. When given intramuscularly, the extent of absorption of methylergonovine maleate solution was about 25% greater than the tablet. The volume of distribution (Vd ss /F) of intramuscularly administered methylergonovine was calculated to be 56.1 ± 17.0 liters, and the plasma clearance (CLp/F) was calculated to be 14.4 ± 4.5 liters per hour. The decline of plasma level of intramuscularly administered methylergonovine was biphasic with a mean elimination half-life of 3.39 hours (range 1.5 to 12.7 hours).
HOW SUPPLIED
Ampules
1 mL size
| Boxes of 10 | NDC 63704-004-01 |
Store and Dispense
Ampules
Store in refrigerator, 2°C-8°C (36°F-46°F). Protect from light. Administer only if solution is clear and colorless.
Mechanism of Action
Methylergonovine maleate acts directly on the smooth muscle of the uterus and increases the tone, rate, and amplitude of rhythmic contractions. Thus, it induces a rapid and sustained tetanic uterotonic effect which shortens the third stage of labor and reduces blood loss.