Methadone Hydrochloride - Methadone Hydrochloride injection, Solution prescribing information
WARNING: SERIOUS AND LIFE-THREATENING RISKS FROM USE OF METHADONE HYDROCHLORIDE INJECTION
Addiction, Abuse, and Misuse
Because the use of Methadone Hydrochloride Injection exposes patients and other users to the risks of opioid addiction, abuse, and misuse, which can lead to overdose and death, assess each patient’s risk prior to prescribing and reassess all patients regularly for the development of these behaviors and conditions (see WARNINGS ).
Life-Threatening Respiratory Depression
Serious, life-threatening, or fatal respiratory depression may occur with use of Methadone Hydrochloride Injection, especially during initiation or following a dosage increase. To reduce the risk of respiratory depression, proper dosing and titration of Methadone Hydrochloride Injection are essential (see WARNINGS ).
Neonatal Opioid Withdrawal Syndrome (NOWS)
Neonatal opioid withdrawal syndrome (NOWS) is an expected and treatable outcome of use of Methadone Hydrochloride Injection during pregnancy. NOWS may be life-threatening if not recognized and treated in the neonate. The balance between the risks of NOWS and the benefits of maternal Methadone Hydrochloride Injection use may differ based on the risks associated with the mother’s underlying condition, pain, or addiction. Advise the patient of the risk of NOWS so that appropriate planning for management of the neonate can occur (see WARNINGS ).
Managing Risks From Concomitant Use With Benzodiazepines Or Other CNS Depressants
Concomitant use of opioids with benzodiazepines or other central nervous system (CNS) depressants, including alcohol, may result in profound sedation, respiratory depression, coma, and death. Reserve concomitant prescribing of Methadone Hydrochloride Injection and benzodiazepines or other CNS depressants for use in patients for whom alternative treatment options are inadequate (see WARNINGS , PRECAUTIONS ).
Life-Threatening QT Prolongation
QT interval prolongation and serious arrhythmia (torsades de pointes) have occurred during treatment with methadone. Most cases involve patients being treated for pain with large, multiple daily doses of methadone, although cases have been reported in patients receiving doses commonly used for maintenance treatment of opioid addiction. Closely monitor patients with risk factors for development of prolonged QT interval, a history of cardiac conduction abnormalities, and those taking medications affecting cardiac conduction for changes in cardiac rhythm during initiation and titration of Methadone Hydrochloride Injection (see WARNINGS ).
Interactions with Drugs Affecting Cytochrome P450 Isoenzymes
The concomitant use of Methadone Hydrochloride Injection with all cytochrome P450 3A4, 2B6, 2C19, 2C9 or 2D6 inhibitors may result in an increase in methadone plasma concentrations, which could cause potentially fatal respiratory depression. In addition, discontinuation of concomitantly used cytochrome P450 3A4 2B6, 2C19, or 2C9 inducers may also result in an increase in methadone plasma concentration. Follow patients closely for respiratory depression and sedation and consider dosage reduction with any changes of concomitant medications that result in an increase in methadone levels (see WARNINGS , PRECAUTIONS: Drug Interactions ).
Conditions for Distribution and Use of Methadone Products for the Treatment of Opioid Addiction
For detoxification and maintenance of opioid dependence, methadone should be administered in accordance with the treatment standards cited in 42 CFR Section 8, including limitations on unsupervised administration (see INDICATIONS AND USAGE ).
INDICATIONS AND USAGE
- Methadone Hydrochloride Injection is indicated for the management of severe and persistent pain that requires an opioid analgesic and that cannot be adequately treated with alternative options, including immediate-release opioids. Limitations of Use
- Because of the risks of addiction, abuse, misuse, overdose, and death, which can occur at any dosage or duration (see WARNINGS ), and persist over the course of therapy, reserve opioid analgesics, including Methadone Hydrochloride Injection, for use in patients for whom alternative treatment options are ineffective, not tolerated, or would be otherwise inadequate to provide sufficient management of pain.
- Methadone Hydrochloride Injection is not indicated as an as-needed (prn) analgesic.
- For use in temporary treatment of opioid dependence in patients unable to take oral medication. Limitations of Use
- Injectable methadone products are not approved for the outpatient treatment of opioid dependence. In this patient population, parenteral methadone is to be used only for patients unable to take oral medication, such as hospitalized patients.
Conditions for Distribution and Use of Methadone Products for the Treatment of Opioid Addiction
Code of Federal Regulations, Title 42, Sec 8.
Methadone products when used for the treatment of opioid addiction in detoxification or maintenance programs, shall be dispensed only by opioid treatment programs (and agencies, practitioners or institutions by formal agreement with the program sponsor) certified by the Substance Abuse and Mental Health Services Administration and approved by the designated state authority. Certified treatment programs shall dispense and use methadone in oral form only and according to the treatment requirements stipulated in the Federal Opioid Treatment Standards (42 CFR 8.12). See below for important regulatory exceptions to the general requirement for certification to provide opioid agonist treatment.
Failure to abide by the requirements in these regulations may result in criminal prosecution, seizure of the drug supply, revocation of the program approval, and injunction precluding operation of the program.
Regulatory Exceptions to the General Requirement for Certification to Provide Opioid Agonist Treatment: During inpatient care, when the patient was admitted for any condition other than concurrent opioid addiction (pursuant to 21CFR 1306.07(c)), to facilitate the treatment of the primary admitting diagnosis.
During an emergency period of no longer than 3 days while definitive care for the addiction is being sought in an appropriately licensed facility (pursuant to 21CFR 1306.07(b)).
DOSAGE AND ADMINISTRATION
Important General Information
Consider the following important factors that differentiate methadone from other opioids:
- The peak respiratory depressant effect of methadone occurs later and persists longer than its peak pharmacologic effect.
- A high degree of opioid tolerance does not eliminate the possibility of methadone overdose, iatrogenic or otherwise. Deaths have been reported during conversion to methadone from chronic, high-dose treatment with other opioid agonists and during initiation of methadone treatment of addiction in subjects previously abusing high doses of other opioid agonists.
- There is high interpatient variability in absorption, metabolism, and relative analgesic potency. Population-based conversion ratios between methadone and other opioids are not accurate when applied to individuals.
- With repeated dosing, methadone is retained in the liver and then slowly released, prolonging the duration of potential toxicity.
- Steady-state plasma concentrations are not attained until 3 to 5 days after initiation of dosing .
- Methadone has a narrow therapeutic index, especially when combined with other drugs.
It is safer to underestimate a patient’s 24-hour methadone dosage and provide rescue medication (e.g., immediate-release opioid) than to overestimate the 24-hour methadone dosage and manage an adverse reaction due to an overdose. While useful tables of opioid equivalents are readily available, there is substantial inter-patient variability in the relative potency of different opioid drugs and products. Frequently reevaluate patients for signs and symptoms of opioid withdrawal and for signs of oversedation/toxicity after converting patients to methadone.
Methadone Hydrochloride Injection for Management of Pain
Methadone Hydrochloride Injection should be prescribed only by healthcare professionals who are knowledgeable about the use of extended-release/long-acting opioids and how to mitigate the associated risks.
Consider the following important factors that differentiate methadone from other opioid analgesics:
- There is high interpatient variability in absorption, metabolism, and relative analgesic potency. Population-based equianalgesic conversion ratios between methadone and other opioids are not accurate when applied to individuals.
- The duration of analgesic action of methadone is 4 to 8 hours (based on single-dose studies) but the plasma elimination half-life is 8 to 59 hours.
- With repeated dosing, the potency of methadone increases due to systemic accumulation.
- Steady-state plasma concentrations, and full analgesic effects, are not attained until at least 3 to 5 days on a dose and may take longer in some patients.
Use the lowest effective dosage for the shortest duration of time consistent with individual patient treatment goals (see WARNINGS ). Because the risk of overdose increases as opioid doses increase, reserve titration to higher doses of Methadone Hydrochloride Injection for patients in whom lower doses are insufficiently effective and in whom the expected benefits of using a higher dose opioid clearly outweigh the substantial risks.
Initiate the dosing regimen for each patient individually, taking into account the patient’s underlying cause and severity of pain, prior analgesic treatment and response, and risk factors for addiction, abuse, and misuse (see WARNINGS ).
Respiratory depression can occur at any time during opioid therapy, especially when initiating and following dosage increases with Methadone Hydrochloride Injection. Consider this risk when selecting an initial dose and when making dose adjustments (see WARNINGS ).
Methadone Hydrochloride Injection multiple-dose vials may be administered intravenously, subcutaneously or intramuscularly. The absorption of subcutaneous and intramuscular methadone has not been well characterized and appears to be unpredictable. Local tissue reactions may occur.
Parenteral products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Use of Parenteral Methadone in Patients who are not Opioid Tolerant
When parenteral methadone is used in patients who are not tolerant to opioids, the usual intravenous methadone starting dose is 2.5 mg to 10 mg every 8 to 12 hours, slowly titrated to effect. More frequent administration may be required during methadone initiation in order to maintain adequate analgesia, and extreme caution is necessary to avoid overdosage, taking into account methadone's long elimination half-life.
Conversion from Oral Methadone to Parenteral Methadone
Conversion from oral methadone to parenteral methadone should initially use a 2:1 dose ratio (e.g., 10 mg oral methadone to 5 mg parenteral methadone).
Conversion from other Opioid Analgesics to Parenteral Methadone
Switching a patient from another opioid analgesic to methadone requires caution due to the uncertainty of dose conversion ratios and incomplete cross-tolerance. Deaths have occurred in opioid tolerant patients during conversion to methadone.
The potency of methadone relative to other opioid analgesics is nonlinear and increases with increasing dose. Table 1 provides an estimated conversion factor for use when converting patients from another opioid to methadone. Because of the high inter-patient variability in absorption, metabolism, and relative potency, it is critical to avoid overestimating the methadone dose which can lead to fatal respiratory depression. The dose conversion scheme below is derived from various consensus guidelines for converting patients to methadone from morphine. The guidelines used to construct this table, however, were all designed for converting patients from oral morphine to oral methadone. The third column assumes a 2:1 ratio for converting from oral to intravenous methadone. Clinicians should consult published conversion guidelines to determine the equivalent morphine dose for patients converting from other opioids.
Consider the following when using the information in Table 1:
- This is not a table of equianalgesic doses.
- The conversion factors in this table are only for the conversion from another oral opioid analgesic to methadone hydrochloride tablets.
- The table cannot be used to convert from methadone hydrochloride tablets to another opioid. Doing so will result in an overestimation of the dose of the new opioid and may result in fatal overdose.
Total Daily Baseline Oral Morphine Dose | Estimated Daily Oral Methadone Requirement as Percent of Total Daily Morphine Dose | Estimated Daily Intravenous Methadone as Percent of Total Daily Oral Morphine Dose The total daily methadone dose derived from the table above may then be divided to reflect the intended dosing schedule (i.e., for administration every 8 hours, divide total daily methadone dose by 3). |
Less than 100 mg | 20% to 30% | 10% to 15% |
100 mg to 300 mg | 10% to 20% | 5% to 10% |
300 mg to 600 mg | 8% to 12% | 4% to 6% |
600 mg to 1000 mg | 5% to 10% | 3% to 5% |
Greater than 1000 mg | Less than 5% | Less than 3% |
Total Daily Baseline Parenteral Morphine Dose | Estimated Daily Parenteral Methadone Requirement as Percent of Total Daily Morphine Dose The total daily methadone dose derived from the table above may then be divided to reflect the intended dosing schedule (i.e., for administration every 8 hours, divide total daily methadone dose by 3). |
10 mg to 30 mg | 40% to 66% |
30 mg to 50 mg | 27% to 66% |
50 mg to 100 mg | 22% to 50% |
100 mg to 200 mg | 15% to 34% |
200 mg to 500 mg | 10% to 20% |
Note: Equianalgesic methadone dosing varies not only between patients, but also within the same patient, depending on baseline morphine (or other opioid) dose. Tables 1 and 2 have been included in order to illustrate this concept and to provide a safe starting point for opioid conversion. Methadone dosing should not be based solely on these tables. Methadone conversion and dose titration methods should always be individualized to account for the patient's prior opioid exposure, general medical condition, concomitant medication, and anticipated breakthrough medication use. The endpoint of titration is achievement of adequate pain relief, balanced against tolerability of opioid side effects. If a patient develops intolerable opioid related side effects, the methadone dose, or dosing interval, may need to be decreased.
Methadone Hydrochloride Injection for Treatment of Opioid Dependence
Detoxification and Maintenance Treatment of Opioid Dependence
For detoxification and maintenance of opiate dependence, methadone should be administered in accordance with the treatment standards cited in 42CFR Section 8.12, including limitations on unsupervised administration. Injectable methadone products are not approved for the outpatient treatment of opioid dependence. Parenteral methadone should be used only for patients who are unable to take oral medication, such as during hospitalization. The patient's oral methadone dose should be converted to an equivalent parenteral dose using the considerations above.
Risk of Relapse in Patients on Methadone Maintenance Treatment of Opioid Addiction
Abrupt opioid discontinuation can lead to development of opioid withdrawal symptoms (see PRECAUTIONS ). Presentation of these symptoms has been associated with an increased risk of susceptible patients to relapse to illicit drug use and should be considered when assessing the risks and benefit of methadone use.
Considerations for Management of Acute Pain During Methadone Maintenance Treatment
Maintenance patients on a stable dose of methadone who experience physical trauma, postoperative pain or other causes of acute pain cannot be expected to derive analgesia from their stable dose of methadone regimens. Such patients should be given analgesics, including opioids, that would be indicated in other patients experiencing similar nociceptive stimulation. Due to the opioid tolerance induced by methadone, when opioids are required for management of acute pain in methadone patients, somewhat higher and/or more frequent doses will often be required than would be the case for other, non-tolerant patients.
Dosage Adjustment During Pregnancy
Methadone clearance may be increased during pregnancy. During pregnancy, a woman's methadone dose may need to be increased, or the dosing interval decreased (see PRECAUTIONS: Pregnancy ).
CONTRAINDICATIONS
Methadone Hydrochloride Injection is contraindicated in patients with:
- Significant respiratory depression
- Acute or severe bronchial asthma in an unmonitored setting or in the absence of resuscitative equipment
- Known or suspected gastrointestinal obstruction, including paralytic ileus
- Hypersensitivity to methadone hydrochloride (e.g. anaphylaxis) or any other ingredient in Methadone Hydrochloride Injection.
ADVERSE REACTIONS
The following serious adverse reactions are described, or described in greater detail, in other sections:
- Addiction, Abuse, and Misuse (see WARNINGS )
- Life Threatening Respiratory Depression (see WARNINGS )
- QT Prolongation (see WARNINGS )
- Neonatal Opioid Withdrawal Syndrome (see WARNINGS )
- Interactions with CNS Depressants (see WARNINGS )
- Serotonin Syndrome (see WARNINGS )
- Adrenal Insufficiency (see WARNINGS )
- Severe Hypotension (see WARNINGS )
- Gastrointestinal Adverse Reactions (see WARNINGS )
- Seizures (see WARNINGS )
- Withdrawal (see WARNINGS )
- Hypoglycemia (see WARNINGS )
The following adverse reactions associated with the use of methadone were identified in clinical studies or post-marketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The major hazards of methadone are respiratory depression and, to a lesser degree, systemic hypotension. Respiratory arrest, shock, cardiac arrest, and death have occurred.
The most frequently observed adverse reactions include lightheadedness, dizziness, sedation, nausea, vomiting, and sweating. These effects seem to be more prominent in ambulatory patients and in those who are not suffering severe pain. In such individuals, lower doses of methadone are advisable. Other adverse reactions that have been reported in patients (including opioid addicts taking methadone for detoxification or maintenance) receiving methadone include the following:
Body as a Whole : asthenia (weakness), edema, headache
Cardiovascular: Arrhythmias, bigeminal rhythms, bradycardia, extrasystoles, tachycardia, Torsade de Pointes, ventricular fibrillation, ventricular tachycardia. ECG abnormalities, prolonged QT interval, T-wave inversion, cardiomyopathy, flushing, heart failure, hypotension, palpitations, phlebitis, syncope
Digestive: Abdominal pain, anorexia, biliary tract spasm, constipation, dry mouth, glossitis
Hematologic and Lymphatic: Reversible thrombocytopenia has been described in opioid addicts with chronic hepatitis.
Metabolic and Nutritional: Hypokalemia, hypomagnesemia, weight gain
Central Nervous System: Agitation, confusion, seizures, disorientation, dysphoria, euphoria, insomnia, hallucinations, seizures, visual disturbances, congenital oculomotor disorders (nystagmus, strabismus)
Respiratory: Pulmonary edema
Skin and Appendages: Intramuscular and Subcutaneous: Local tissue reactions (pain, erythema, swelling), particularly with continuous subcutaneous infusion
Intravenous: Pruritis, urticaria, other skin rashes, and rarely, hemorrhagic urticaria
Special Senses: Visual disturbances
Urogenital: Antidiuretic effect, amenorrhea, urinary retention or hesitancy, reduced libido and/or potency
Serotonin Syndrome : Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of opioids with serotonergic drugs.
Adrenal Insufficiency : Cases of adrenal insufficiency have been reported with opioid use, more often following greater than one month of use.
Anaphylaxis: Anaphylaxis has been reported with ingredients contained in Methadone Hydrochloride Injection.
Androgen Deficiency: Cases of androgen deficiency have occurred with use of opioids for an extended period of time.
Hyperalgesia and Allodynia: Cases of hyperalgesia and allodynia have been reported with opioid therapy of any duration (see WARNINGS ).
Hypoglycemia: Cases of hypoglycemia have been reported in patients taking methadone (see WARNINGS ).
Opioid-Induced Esophageal Dysfunction (OIED): Cases of OIED have been reported in patients taking opioids and may occur more frequently in patients taking higher doses of opioids, and/or in patients taking opioids longer term (see WARNINGS).
Adverse Reactions from Observational Studies
A prospective, observational cohort study estimated the risks of addiction, abuse, and misuse in patients initiating long-term use of Schedule II opioid analgesics between 2017 and 2021. Study participants included in one or more analyses had been enrolled in selected insurance plans or health systems for at least one year, were free of at least one outcome at baseline, completed a minimum number of follow-up assessments, and either: 1) filled multiple extended-release/long-acting opioid analgesic prescriptions during a 90-day period (n = 978); or 2) filled any Schedule II opioid analgesic prescriptions covering at least 70 of 90 days (n = 1,244). Those included also had no dispensing of the qualifying opioids in the previous 6 months.
Over 12 months:
- approximately 1% to 6% of participants across the two cohorts newly met criteria for addiction, as assessed with two validated interview-based measures of moderate-to-severe opioid use disorder based on Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) criteria, and
- approximately 9% and 22% of participants across the two cohorts newly met criteria for prescription opioid abuse and misuse (defined in DRUG ABUSE AND DEPENDENCE ), respectively, as measured with a validated self-reported instrument.
A retrospective, observational cohort study estimated the risk of opioid-involved overdose or opioid overdose-related death in patients with new long-term use of Schedule II opioid analgesics from 2006 through 2016 (n = 220,249). Included patients had been enrolled in either one of two commercial insurance programs, one managed care program, or one Medicaid program for at least 9 months. New long-term use was defined as having Schedule II opioid analgesic prescriptions covering at least 70 days’ supply over the 3 months prior to study entry and none during the preceding 6 months. Patients were excluded if they had an opioid-involved overdose in the 9 months prior to study entry. Overdose was measured using a validated medical code-based algorithm with linkage to the National Death Index database. The 5-year cumulative incidence estimates for opioid-involved overdose or opioid overdose-related death ranged from approximately 1.5% to 4% across study sites, counting only the first event during follow-up. Approximately 17% of first opioid overdoses observed over the entire study period (5-11 years, depending on the study site) were fatal. Higher baseline opioid dose was the strongest and most consistent predictor of opioid-involved overdose or opioid overdose-related death. Study exclusion criteria may have selected patients at lower risk of overdose, and substantial loss to follow-up (approximately 80%) also may have biased estimates.
The risk estimates from the studies described above may not be generalizable to all patients receiving opioid analgesics, such as those with exposures shorter or longer than the duration evaluated in the studies.
Drug Interactions
Inhibitors of CYP3A4, CYP2B6, CYP2C19, CYP2C9, or CYP2D6 | |
Clinical Impact: | Methadone undergoes hepatic N-demethylation by several cytochrome P450 (CYP) isoforms, including CYP3A4, CYP2B6, CYP2C19, CYP2C9, and CYP2D6. The concomitant use of Methadone Hydrochloride Injection and CYP3A4, CYP2B6, CYP2C19, CYP2C9, or CYP2D6 inhibitors can increase the plasma concentration of methadone, resulting in increased or prolonged opioid effects, and may result in a fatal overdose, particularly when an inhibitor is added after a stable dose of Methadone Hydrochloride Injection is achieved. These effects may be more pronounced with concomitant use of drugs that inhibit more than one of the CYP enzymes listed above. After stopping a CYP3A4, CYP2B6, CYP2C19, CYP2C9, or CYP2D6 inhibitor, as the effects of the inhibitor decline, the methadone plasma concentration can decrease (see CLINICAL PHARMACOLOGY ), resulting in decreased opioid efficacy or withdrawal symptoms in patients physically dependent on methadone. |
Intervention: | If concomitant use is necessary, consider dosage reduction of Methadone Hydrochloride Injection until stable drug effects are achieved. Monitor patients for respiratory depression and sedation at frequent intervals. If a CYP3A4, CYP2B6, CYP2C19, CYP2C9, or CYP2D6 inhibitor is discontinued, follow patients for signs of opioid withdrawal and consider increasing the Methadone Hydrochloride Injection dosage until stable drug effects are achieved. |
Examples: | Macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g., ketoconazole), protease inhibitors (e.g., ritonavir), fluconazole, fluvoxamine, Some selective serotonin reuptake inhibitors (SSRIs) (e.g., sertraline, fluvoxamine) |
Inducers of CYP3A4, CYP2B6, CYP2C19, or CYP2C9 | |
Clinical Impact: | The concomitant use of Methadone Hydrochloride Injection and CYP3A4, CYP2B6, CYP2C19, or CYP2C9 inducers can decrease the plasma concentration of methadone (see CLINICAL PHARMACOLOGY ), resulting in decreased efficacy or onset of withdrawal symptoms in patients physically dependent on methadone. These effects could be more pronounced with concomitant use of drugs that can induce multiple CYP enzymes. After stopping a CYP3A4, CYP2B6, CYP2C19, or CYP2C9 inducer, as the effects of the inducer decline, the methadone plasma concentration can increase (see CLINICAL PHARMACOLOGY ), which could increase or prolong both the therapeutic effects and adverse reactions, and may cause serious respiratory depression, sedation, or death. |
Intervention: | If concomitant use is necessary, consider increasing the Methadone Hydrochloride Injection dosage until stable drug effects are achieved. Monitor for signs of opioid withdrawal. If a CYP3A4, CYP2B6, CYP2C19, or CYP2C9 inducer is discontinued, consider Methadone Hydrochloride Injection dosage reduction and monitor for signs of respiratory depression and sedation. |
Examples: | Rifampin, carbamazepine, phenytoin, St. John’s Wort, Phenobarbital |
Benzodiazepines and other Central Nervous System (CNS) Depressants | |
Clinical Impact: | Due to additive pharmacologic effect, the concomitant use of benzodiazepines or other CNS depressants, including alcohol, can increase the risk of hypotension, respiratory depression, profound sedation, coma, and death (see WARNINGS ). |
Intervention: | Reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Monitor patients closely for signs of respiratory depression and sedation (see WARNINGS , PRECAUTIONS ). |
Examples: | Benzodiazepines and other sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, gabapentinoids (gabapentin or pregabalin), other opioids, alcohol. |
Potentially Arrhythmogenic Agents | |
Clinical Impact: | Pharmacodynamic interactions may occur with concomitant use of methadone and potentially arrhythmogenic agents or drugs capable of inducing electrolyte disturbances (hypomagnesemia, hypokalemia). |
Intervention: | Monitor patients closely for cardiac conduction changes. |
Examples: | Drugs known to have potential to prolong QT interval : Class I and III antiarrhythmics, some neuroleptics and tricyclic antidepressants, and calcium channel blockers. Drugs capable of inducing electrolyte disturbances : Diuretics, laxatives, and, in rare cases, mineralocortocoid hormones. |
Serotonergic Drugs | |
Clinical Impact: | The concomitant use of opioids with other drugs that affect the serotonergic neurotransmitter system has resulted in serotonin syndrome (see WARNINGS , PRECAUTIONS ). |
Intervention: | If concomitant use is warranted, carefully observe the patient, particularly during treatment initiation and dose adjustment. Discontinue Methadone Hydrochloride Injection if serotonin syndrome is suspected. |
Examples: | Selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, drugs that effect the serotonin neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants (i.e., cyclobenzaprine, metaxalone), monoamine oxidase (MAO) inhibitors (those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue). |
Monoamine Oxidase Inhibitors (MAOIs) | |
Clinical Impact: | MAOI interactions with opioids may manifest as serotonin syndrome (see PRECAUTIONS ) or opioid toxicity (e.g., respiratory depression, coma) (see PRECAUTIONS ) . |
Intervention: | The use of Methadone Hydrochloride Injection is not recommended for patients taking MAOIs or within 14 days of stopping such treatment. |
Mixed Agonist/Antagonist and Partial Agonist Opioid Analgesics | |
Clinical Impact: | May reduce the analgesic effect of Methadone Hydrochloride Injection and/or precipitate withdrawal symptoms. |
Intervention: | Avoid concomitant use. |
Examples: | butorphanol, nalbuphine, pentazocine, buprenorphine |
Muscle Relaxants | |
Clinical Impact: | Methadone may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression. |
Intervention: | Monitor patients for signs of respiratory depression that may be greater than otherwise expected and decrease the dosage of Methadone Hydrochloride Injection and/or the muscle relaxant as necessary. |
Examples: | cyclobenzaprine, metaxalone |
Diuretics | |
Clinical Impact: | Opioids can reduce the efficacy of diuretics by inducing the release of antidiuretic hormone. |
Intervention: | Monitor patients for signs of diminished diuresis and/or effects on blood pressure and increase the dosage of the diuretic as needed. |
Anticholinergic Drugs | |
Clinical Impact: | The concomitant use of anticholinergic drugs may increase risk of urinary retention and/or severe constipation, which may lead to paralytic ileus. |
Intervention: | Monitor patients for signs of urinary retention or reduced gastric motility when Methadone Hydrochloride Injection is used concomitantly with anticholinergic drugs. |
Anti-Retroviral Agents
Nevirapine
Based on the known metabolism of methadone, nevirapine may decrease plasma concentrations of methadone by increasing its hepatic metabolism. Opioid withdrawal syndrome has been reported in patients treated with nevirapine and methadone concomitantly. Methadone-maintained patients beginning nevirapine therapy should be monitored for evidence of withdrawal and methadone dose should be adjusted accordingly.
Efavirenz
Coadministration of efavirenz in HIV-infected methadone-maintenance patients has resulted in decreased methadone plasma concentrations of methadone associated with signs of opioid withdrawal, and necessitating increases in methadone dose.
Ritonavir and Ritonavir/Lopinavir
Reduced plasma methadone levels have been observed after administration of ritonavir alone or ritonavir/lopinavir combination. Withdrawal symptoms were however, inconsistently observed. Caution is warranted when administering methadone to patients receiving ritonavir-containing regimens in addition to other drugs known to decrease methadone plasma levels.
Zidovudine
Experimental evidence suggests that methadone increases the area under the concentration-time curve (AUC) of zidovudine with possible toxic effects.
Didanosine and Stavudine
Experimental evidence suggests that methadone decreased the AUC and peak levels for didanosine and stavudine, with a more significant decrease for didanosine. Methadone disposition was not substantially altered.
Desipramine
Blood levels of desipramine have increased with concurrent methadone therapy.
DESCRIPTION
Methadone Hydrochloride Injection USP, 10 mg/mL is an opioid analgesic for parenteral use (intravenous, intramuscular or subcutaneous use). Methadone Hydrochloride Injection USP, 10 mg/mL contains methadone hydrochloride as the active pharmaceutical ingredient.
Methadone hydrochloride is a white, crystalline material that is water-soluble. Methadone hydrochloride is chemically described as 6-(dimethylamino)-4,4-diphenyl-3-hepatanone hydrochloride. Its molecular formula is C 21 H 27 NO•HCl and it has a molecular weight of 345.91. Methadone hydrochloride has a melting point of 235°C, and a pKa of 8.25 in water at 20°C. Its octanol/water partition coefficient at pH 7.4 is 117. A solution (1:100) in water has a pH between 4.5 and 6.5.
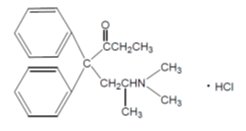
Methadone hydrochloride has the following structural formula:
Methadone Hydrochloride Injection is a sterile injectable solution available in 20 mL multiple-dose vials. Each mL of the Methadone Hydrochloride Injection contains: 10 mg of methadone hydrochloride (equivalent to 8.95 mg of methadone free base), 5 mg of chlorobutanol, as a preservative, 9 mg of sodium chloride, as tonicity agent, and hydrochloric acid and sodium hydroxide as pH adjusters, in water for injection. The pH of the sterile injectable solution is between 3.0 to 6.5.
CLINICAL PHARMACOLOGY
Mechanism of Action
Methadone hydrochloride is a mu-agonist; a synthetic opioid analgesic with multiple actions qualitatively similar to those of morphine, the most prominent of which involve the central nervous system and organs composed of smooth muscle. The principal therapeutic uses for methadone are for analgesia and for detoxification or maintenance in opioid addiction. The methadone abstinence syndrome, although qualitatively similar to that of morphine, differs in that the onset is slower, the course is more prolonged, and the symptoms are less severe.
Some data also indicate that methadone acts as an antagonist at the N-methyl-D-aspartate (NMDA) receptor. The contribution of NMDA receptor antagonism to methadone’s efficacy is unknown.
Pharmacodynamics
Effects on the Central Nervous System
Methadone produces respiratory depression by direct action on brain stem respiratory centers. The respiratory depression involves a reduction in the responsiveness of the brain stem respiratory centers to both increases in carbon dioxide tension and electrical stimulation.
Methadone causes miosis, even in total darkness. Pinpoint pupils are a sign of opioid overdose but are not pathognomonic (e.g., pontine lesions of hemorrhagic or ischemic origins may produce similar findings). Marked mydriasis rather than miosis may be seen due to hypoxia in overdose situations.
Some NMDA receptor antagonists have been shown to produce neurotoxic effects in animals.
Effects on the Gastrointestinal Tract and Other Smooth Muscle
Methadone causes a reduction in motility associated with an increase in smooth muscle tone in the antrum of the stomach and duodenum. Digestion of food in the small intestine is delayed and propulsive contractions are decreased. Propulsive peristaltic waves in the colon are decreased, while tone is increased to the point of spasm, resulting in constipation. Other opioid-induced effects may include a reduction in biliary and pancreatic secretions, spasm of sphincter of Oddi, transient elevations in serum amylase, and opioid-induced esophageal dysfunction (OIED).
Effects on the Cardiovascular System
Methadone produces peripheral vasodilation, which may result in orthostatic hypotension or syncope. Manifestations of histamine release and/or peripheral vasodilation may include pruritus, flushing, red eyes, sweating, and/or orthostatic hypotension.
Effects on the Endocrine System
Opioids inhibit the secretion of adrenocorticotropic hormone (ACTH), cortisol, and luteinizing hormone (LH) in humans. They also stimulate prolactin, growth hormone (GH) secretion, and pancreatic secretion of insulin and glucagon.
Use of opioids for an extended period of time may influence the hypothalamic-pituitary-gonadal axis, leading to androgen deficiency that may manifest as low libido, impotence, erectile dysfunction, amenorrhea, or infertility. The causal role of opioids in the clinical syndrome of hypogonadism is unknown because the various medical, physical, lifestyle, and psychological stressors that may influence gonadal hormone levels have not been adequately controlled for in studies conducted to date.
Effects on the Immune System
Opioids have been shown to have a variety of effects on components of the immune system in in vitro and animal models. The clinical significance of these findings is unknown. Overall, the effects of opioids appear to be modestly immunosuppressive.
Concentration–Efficacy Relationships
The minimum effective analgesic concentration will vary widely among patients, especially among patients who have been previously treated with opioid agonists. The minimum effective analgesic concentration of methadone for any individual patient may increase over time due to an increase in pain, the development of a new pain syndrome, and/or the development of analgesic tolerance.
Concentration–Adverse Reaction Relationships
There is a relationship between increasing methadone plasma concentration and increasing frequency of dose-related opioid adverse reactions such as nausea, vomiting, CNS effects, and respiratory depression. In opioid-tolerant patients, the situation may be altered by the development of tolerance to opioid-related adverse reactions.
Pharmacokinetics
Absorption
Methadone Hydrochloride Injection is intended for parenteral (intravenous, subcutaneous and intramuscular) administration. Methadone pharmacokinetics following subcutaneous and intramuscular administration have not been systematically studied and differences among the various parenteral routes have not been well characterized. As with many drugs, absorption into the systemic circulation may vary with subcutaneous and intramuscular administration.
Distribution
Methadone is a lipophilic drug and the steady state volume of distribution ranges between 2 L/kg to 6 L/kg. In plasma, methadone is predominantly bound to α 1 -acid glycoprotein (85% to 90%). Methadone is secreted in saliva, breast milk, amniotic fluid and umbilical cord plasma.
Elimination
Metabolism
Methadone is primarily metabolized by N-demethylation to an inactive metabolite, 2-ethylidene-1,5-dimethyl-3,3-diphenylpyrrolidene (EDDP). Cytochrome P450 enzymes, primarily CYP3A4, CYP2B6, CYP2C19, CYP2C9 and CYP2D6, are responsible for conversion of methadone to EDDP and other inactive metabolites, which are excreted mainly in urine.
Excretion
Elimination of methadone is mediated by extensive biotransformation, followed by renal and fecal excretion. After single intravenous dose administration the plasma clearance of methadone ranged between 3 L/h to 10 L/h and the terminal half-life (t ½ ) ranged between 8 to 59 hours. Methadone has been known to persist in the liver and other tissues. Slow release from the liver and other tissues may prolong the duration of methadone action despite low plasma concentrations.
Specific Populations
Use During Pregnancy
There are no pharmacokinetic studies of parenteral methadone in pregnancy. The disposition of oral methadone has been studied in approximately 30 pregnant patients in 2 nd and 3 rd trimesters. Elimination of methadone was significantly changed in pregnancy. Total body clearance of methadone was increased in pregnant patients compared to the same patients postpartum or to non-pregnant opioid-dependent women. The terminal half-life of methadone is decreased during second and third trimesters. The decrease in plasma half-life and increased clearance of methadone resulting in lower methadone trough levels during pregnancy can lead to withdrawal symptoms in some pregnant patients. The dosage may need to be increased or the dosing interval decreased in pregnant patients receiving methadone (see DOSAGE AND ADMINISTRATION ).
Hepatic Impairment
Methadone pharmacokinetics have not been extensively evaluated in patients with hepatic insufficiency. Methadone is metabolized in the liver and patients with liver impairment may be at risk of accumulating methadone after multiple dosing.
Renal Impairment
Methadone pharmacokinetics have not been extensively evaluated in patients with renal insufficiency. Unchanged methadone and its metabolites are excreted in urine to a variable degree. Methadone is a basic (pKa = 9.2) compound and the luminal pH of the urinary tract can affect its extraction from plasma. Urine acidification has been shown to increase renal elimination of methadone. Forced diuresis, peritoneal dialysis, hemodialysis, or charcoal hemoperfusion have not been established as beneficial for increasing methadone or metabolite elimination.
Sex
The pharmacokinetics of methadone have not been evaluated for sex specificity.
Race
The pharmacokinetics of methadone have not been evaluated for race specificity.
Age
Geriatric Population:
The pharmacokinetics of methadone have not been evaluated in geriatric population.
Pediatric Population:
The pharmacokinetics of methadone have not been evaluated in pediatric population.
Drug Interaction Studies
Cytochrome P450 Interactions
Methadone undergoes hepatic N-demethylation by cytochrome P450 (CYP) isoforms, principally CYP3A4, CYP2B6, CYP2C19, CYP2C9 and CYP2D6. Co-administration of methadone with CYP inducers may result in more rapid metabolism and potential for decreased effects of methadone, whereas administration with CYP inhibitors may reduce metabolism and potentiate methadone’s effects. Although antiretroviral drugs such as efavirenz, nelfinavir, nevirapine, ritonavir, lopinavir+ritonavir combination are known to inhibit some CYPs, they are shown to reduce the plasma levels of methadone, possibly due to CYP induction activity (see PRECAUTIONS: Drug Interactions ).
Cytochrome P450 Inducers
The following drug interactions were reported following co-administration of methadone with known inducers of cytochrome P450 enzymes:
Rifampin
In patients well-stabilized on methadone, concomitant administration of rifampin resulted in a marked reduction in serum methadone levels and a concurrent appearance of withdrawal symptoms.
Phenytoin
In a pharmacokinetic study with patients on methadone maintenance therapy, phenytoin administration (250 mg twice daily initially for 1 day followed by 300 mg daily for 3 to 4 days) resulted in an approximately 50% reduction in methadone exposure and withdrawal symptoms occurred concurrently. Upon discontinuation of phenytoin, the incidence of withdrawal symptoms decreased and methadone exposure increased to a level comparable to that prior to phenytoin administration.
St. John’s Wort, Phenobarbital, Carbamazepine
Administration of methadone with other CYP3A4 inducers may result in withdrawal symptoms.
Cytochrome P450 Inhibitors
Voriconazole
Voriconazole can inhibit the activity of CYP3A4, CYP2C9, and CYP2C19. Repeat dose administration of oral voriconazole (400 mg every 12 hours for 1 day, then 200 mg every 12 hours for 4 days) increased the peak plasma concentration (C max ) and AUC of (R)-methadone by 31% and 47%, respectively, in subjects receiving a methadone maintenance dose (30 mg to 100 mg daily. The C max and AUC of (S)-methadone increased by 65% and 103%, respectively. Increased plasma concentrations of methadone have been associated with toxicity including QT prolongation. Frequent monitoring for adverse events and toxicity related to methadone is recommended during co-administration. Dose reduction of methadone may be needed (see PRECAUTIONS: Drug Interactions ).
Antiretroviral Drugs
Although antiretroviral drugs such as efavirenz, nelfinavir, nevirapine, ritonavir, telaprevir, lopinavir+ritonavir combination are known to inhibit some CYPs, they are shown to reduce the plasma levels of methadone, possibly due to CYP induction activity.
Abacavir, amprenavir, darunavir+ritonavir, efavirenz, nelfinavir, nevirapine, ritonavir, telaprevir, lopinavir+ritonavir, saquinavir+ritonavir, tipranvir+ritonavir combination
Co-administration of these anti-retroviral agents resulted in increased clearance or decreased plasma levels of methadone (see PRECAUTIONS: Drug Interactions ).
Didanosine and Stavudine
Methadone decreased the AUC and peak levels for didanosine and stavudine, with a more significant decrease for didanosine. Methadone disposition was not substantially altered (see PRECAUTIONS: Drug Interactions ).
Zidovudine
Methadone increased the AUC of zidovudine which could result in toxic effects (see PRECAUTIONS: Drug Interactions ).
Pharmacokinetics in Special Populations
Pregnancy
There are no pharmacokinetic studies of parenteral methadone in pregnancy. The disposition of oral methadone has been studied in approximately 30 pregnant patients in 2 nd and 3 rd trimesters. Elimination of methadone was significantly changed in pregnancy. Total body clearance of methadone was increased in pregnant patients compared to the same patients postpartum or to non-pregnant opioid-dependent women. The terminal half-life of methadone is decreased during second and third trimesters. The decrease in plasma half-life and increased clearance of methadone resulting in lower methadone trough levels during pregnancy can lead to withdrawal symptoms in some pregnant patients. The dosage may need to be increased or the dosing interval decreased in pregnant patients receiving methadone (see DOSAGE AND ADMINISTRATION ).
HOW SUPPLIED
Methadone Hydrochloride Injection USP, 200 mg/20 mL (10 mg/mL) is a clear, colorless solution and is available in:
NDC 67457-217-20 20 mL Multiple-Dose Vials: One vial per carton
Store at 20°C to 25°C (68°F to 77°F), with excursions permitted between 15°C to 30°C (59°F to 86°F). [See USP Controlled Room Temperature.]
Protect from light. Store in carton until contents have been used.
Manufactured for: Mylan Institutional LLC Morgantown, WV 26505 U.S.A.
Manufactured by: Alcami Corporation Charleston, SC 29405 U.S.A.
XXXXXXX Revised: 12/2025 MI:MTHDIJ:RX12
Mechanism of Action
Methadone hydrochloride is a mu-agonist; a synthetic opioid analgesic with multiple actions qualitatively similar to those of morphine, the most prominent of which involve the central nervous system and organs composed of smooth muscle. The principal therapeutic uses for methadone are for analgesia and for detoxification or maintenance in opioid addiction. The methadone abstinence syndrome, although qualitatively similar to that of morphine, differs in that the onset is slower, the course is more prolonged, and the symptoms are less severe.
Some data also indicate that methadone acts as an antagonist at the N-methyl-D-aspartate (NMDA) receptor. The contribution of NMDA receptor antagonism to methadone’s efficacy is unknown.