Get your patient on Mefenamic Acid
Mefenamic Acid prescribing information
BOXED WARNING
Cardiovascular Risk
NSAIDs may cause an increased risk of serious cardiovascular thrombotic events, myocardial infarction, and stroke, which can be fatal. This risk may increase with duration of use. Patients with cardiovascular disease or risk factors for cardiovascular disease may be at greater risk (See WARNINGS ).
Mefenamic acid is contraindicated for the treatment of peri-operative pain in the setting of coronary artery bypass graft (CABG) surgery (See WARNINGS ).
Gastrointestinal Risk
NSAIDs cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal.
These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events (See WARNINGS ).
INDICATIONS AND USAGE
Carefully consider the potential benefits and risks of mefenamic acid and other treatment options before deciding to use mefenamic acid. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ).
Mefenamic acid is indicated:
- For relief of mild to moderate pain in patients ≥14 years of age, when therapy will not exceed one week (7 days).
- For treatment of primary dysmenorrhea.
DOSAGE AND ADMINISTRATION
Carefully consider the potential benefits and risks of mefenamic acid and other treatment options before deciding to use mefenamic acid. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ).
After observing the response to initial therapy with mefenamic acid, the dose and frequency should be adjusted to suit an individual patient's needs.
For the relief of acute pain in adults and adolescents ≥14 years of age, the recommended dose is 500 mg as an initial dose followed by 250 mg every 6 hours as needed, usually not to exceed one week. 4
For the treatment of primary dysmenorrhea, the recommended dose is 500 mg as an initial dose followed by 250 mg every 6 hours, given orally, starting with the onset of bleeding and associated symptoms. Clinical studies indicate that effective treatment can be initiated with the start of menses and should not be necessary for more than 2 to 3 days. 5
CONTRAINDICATIONS
Mefenamic acid is contraindicated in patients with known hypersensitivity to mefenamic acid.
Mefenamic acid should not be given to patients who have experienced asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients (see WARNINGS - Anaphylactoid Reactions, and PRECAUTIONS - Preexisting Asthma).
Mefenamic acid is contraindicated for the treatment of peri-operative pain in the setting of coronary artery bypass graft (CABG) surgery (see WARNINGS ).
Mefenamic acid is contraindicated in patients with acute active ulceration or chronic inflammation of either the upper or lower gastrointestinal tract.
Mefenamic acid should not be used in patients with pre-existing renal disease.
ADVERSE REACTIONS
In patients taking mefenamic acid or other NSAIDs, the most frequently reported adverse experiences occurring in approximately 1-10% of patients are:
Gastrointestinal experiences including - abdominal pain, constipation, diarrhea, dyspepsia, flatulence, gross bleeding/perforation, heartburn, nausea, GI ulcers (gastric/duodenal), vomiting, abnormal renal function, anemia, dizziness, edema, elevated liver enzymes, headaches, increased bleeding time, pruritus, rashes, tinnitus.
Additional adverse experiences reported occasionally and listed here by body system include:
Body as a whole - fever, infection, sepsis
Cardiovascular system - congestive heart failure, hypertension, tachycardia, syncope
Digestive system - dry mouth, esophagitis, gastric/peptic ulcers, gastritis, gastrointestinal bleeding, glossitis, hematemesis, hepatitis, jaundice
Hemic and lymphatic system - ecchymosis, eosinophilia, leukopenia, melena, purpura, rectal bleeding, stomatitis, thrombocytopenia
Metabolic and nutritional - weight changes
Nervous system - anxiety, asthenia, confusion, depression, dream abnormalities, drowsiness; insomnia, malaise, nervousness, paresthesia, somnolence, tremors, vertigo
Respiratory system - asthma, dyspnea
Skin and appendages - alopecia, photosensitivity, pruritus, sweat
Special senses - blurred vision
Urogenital system - cystitis, dysuria, hematuria, interstitial nephritis, oliguria/polyuria, proteinuria, renal failure
Other adverse reactions, which occur rarely are:
Body as a whole - anaphylactoid reactions, appetite changes, death
Cardiovascular system - arrhythmia, hypotension, myocardial infarction, palpitations, vasculitis
Digestive system - eructation, liver failure, pancreatitis
Hemic and lymphatic system - agranulocytosis, hemolytic anemia, aplastic anemia, lymphadenopathy, pancytopenia
Metabolic and nutritional – hyperglycemia
Nervous system - convulsions, coma, hallucinations, meningitis
Respiratory - respiratory depression, pneumonia
Skin and appendages - angioedema, toxic epidermal necrosis, erythema multiforme, exfoliative dermatitis, Stevens-Johnson syndrome, urticaria
Special senses - conjunctivitis, hearing impairment
Drug Interactions
A number of compounds are inhibitors of CYP2C9. Drug interactions studies of mefenamic acid and these compounds have not been conducted. The possibility of altered safety and efficacy should be considered when mefenamic acid is used concomitantly with these drugs.
ACE-inhibitors
Reports suggest that NSAIDs may diminish the antihypertensive effect of ACE-inhibitors. This interaction should be given consideration in patients taking NSAIDs concomitantly with ACE-inhibitors.
Aspirin
When mefenamic acid is administered with aspirin, its protein binding is reduced, although the clearance of free mefenamic acid is not altered. The clinical significance of this interaction is not known; however, as with other NSAIDs, concomitant administration of MEFENAMIC ACID and aspirin is not generally recommended because of the potential of increased adverse effects.
Diuretics
Clinical studies, as well as post marketing observations, have shown that mefenamic acid can reduce the natriuretic effect-of furosemide and thiazides in some patients. This response has been attributed to inhibition of renal prostaglandin synthesis. During concomitant therapy of NSAIDs, the patient should be observed closely for signs of renal failure (see PRECAUTIONS, Renal Effects), as well as to assure diuretic efficacy.
Lithium
NSAIDs have produced an elevation of plasma lithium levels and a reduction in renal lithium clearance. The mean minimum lithium concentration increased 15% and the renal clearance decreased by approximately 20%. These effects have been attributed to inhibition of renal prostaglandin synthesis by the NSAID. Thus, when NSAIDs and lithium are administered concurrently, subjects should be observed carefully for signs of lithium toxicity.
Methotrexate
NSAIDs have been reported to competitively inhibit methotrexate accumulation in rabbit kidney slices. This may indicate that they could enhance the toxicity of methotrexate. Caution should be used when NSAIDs are administered concomitantly with methotrexate.
Warfarin
The effects of warfarin and NSAIDs on GI bleeding are synergistic, such that users of both drugs together have a risk of serious GI bleeding higher than users of either drug alone.
Antacids
In a single dose study (n=6), ingestion of an antacid containing 1.7-gram of magnesium hydroxide with 500-mg of mefenamic acid increased the C max and AUC of mefenamic acid by 125% and 36%, respectively. 1
DESCRIPTION
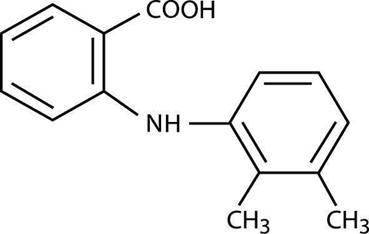
Mefenamic acid is a member of the fenamate group of nonsteroidal anti-inflammatory drugs (NSAIDs). Each green-banded, yellow capsule contains 250 mg of mefenamic acid for oral administration. Mefenamic acid is a white to greyish-white, odorless, microcrystalline powder with a melting point of 230°-231°C and water solubility of 0.004% at pH 7.1. The chemical name is N-2,3-xylylanthranilic acid. The molecular weight is 241.29. Its molecular formula is C 15 H 15 NO 2 and the structural formula of mefenamic acid is:
Each capsule also contains lactose monohydrate. The capsule shell and band contain dimethicone, D&C yellow No. 10, FD&C blue No. 1 aluminum lake, FD&C yellow No. 6, gelatin, propylene glycol, shellac, titanium dioxide and yellow iron oxide.
CLINICAL PHARMACOLOGY
Pharmacodynamics
Mefenamic acid is a non-steroidal anti-inflammatory drug (NSAID) that exhibits anti-inflammatory, analgesic, and antipyretic activities in animal models. The mechanism of action of mefenamic acid, like that of other NSAIDs, is not completely understood but may be related to prostaglandin synthetase inhibition.
Pharmacokinetics
Absorption
Mefenamic acid is rapidly absorbed after oral administration. In two 500-mg single oral dose studies, the mean extent of absorption was 30.5 mcg/hr/mL (17%CV). 1,2 The bioavailability of the capsule relative to an IV dose or an oral solution has not been studied. Following a single 1-gram oral dose, mean peak plasma levels ranging from 10-20 mcg/mL 3 have been reported. Peak plasma levels are attained in 2 to 4 hours and the elimination half-life approximates 2 hours. Following multiple doses, plasma levels are proportional to dose with no evidence of drug accumulation. In a multiple dose trial of normal adult subjects (n=6) receiving 1-gram doses of mefenamic acid four times daily, steady-state concentrations of 20 mcg/mL were reached on the second day of administration, consistent with the short half-life.
The effect of food on the rate and extent of absorption of mefenamic acid has not been studied. Concomitant ingestion of antacids containing magnesium hydroxide has been shown to significantly increase the rate and extent of mefenamic acid absorption (see PRECAUTIONS, Drug Interactions ). 1
Distribution
Mefenamic acid has been reported as being greater than 90% bound to albumin. 9 The relationship of unbound fraction to drug concentration has not been studied. The apparent volume of distribution ( Vzs s /F) estimated following a 500-mg oral dose of mefenamic acid was 1.06 L/kg. 2
Based on its physical and chemical properties, mefenamic acid is expected to be excreted in human breast milk.
Metabolism
Mefenamic acid is metabolized by cytochrome P450 enzyme CYP2C9 to 3-hydroxymethyl mefenamic acid (Metabolite I). Further oxidation to a 3-carboxymefenamic acid (Metabolite II) may occur. 10 The activity of these metabolites has not been studied. The metabolites may undergo glucuronidation and mefenamic acid is also glucuronidated directly. A peak plasma level approximating 20 mcg/mL was observed at 3 hours for the hydroxy metabolite and its glucuronide (n=6) after a single 1-gram dose. Similarly, a peak plasma level of 8 mcg/mL was observed at 6-8 hours for the carboxy metabolite and its glucuronide. 3
Excretion
Approximately fifty-two percent of a mefenamic acid dose is excreted into the urine primarily as glucuronides of mefenamic acid (6%), 3-hydroxymefenamic acid (25%) and 3-carboxymefenamic acid (21%). The fecal route of elimination accounts for up to 20% of the dose, mainly in the form of unconjugated 3-carboxymefenamic acid. 3
The elimination half-life of mefenamic acid is approximately two hours. Half-lives of metabolites I and II have not been precisely reported, but appear to be longer than the parent compound. 3 The metabolites may accumulate in patients with renal or hepatic failure. The mefenamic acid glucuronide may bind irreversibly to plasma proteins. Because both renal and hepatic excretion are significant pathways of elimination, dosage adjustments in patients with renal or hepatic dysfunction may be necessary. Mefenamic acid should not be administered to patients with preexisting renal disease or in patients with significantly impaired renal function.
| PK Parameters | Normal Healthy Adults (18-45 yr) | |
| Value | CV | |
| T ma x (hr) | 2 | 66 |
| Oral clearance (L/hr) | 21.23 | 38 |
| Apparent volume of distribution; Vz/F (L/kg) | 1.06 | 60 |
| Half-life; t ½ (hrs) | 2 to 4 | NA |
Special Populations
Pediatric
Mefenamic acid has not been adequately investigated in pediatric patients less than 14 years of age.
A study in 17 preterm infants administered 2 mg/kg indicated that the half-life was about five times as long as adults, consistent with the low activity of metabolic enzymes in newborn infants. The mean C max in this study was 4 mcg/mL (range 2.9-6.1). The mean time to maximum concentration (T max ) was 8 hours (range 2-18 hrs). 11
Race
Pharmacokinetic differences due to race have not been identified.
Hepatic Insufficiency
Mefenamic acid pharmacokinetics have not been studied in patients with hepatic dysfunction. As hepatic metabolism is a significant pathway of mefenamic acid elimination, patients with acute and chronic hepatic disease may require reduced doses of mefenamic acid compared to patients with normal hepatic function.
Renal Insufficiency
Mefenamic acid pharmacokinetics have not been investigated in subjects with renal insufficiency. Given that mefenamic acid, its metabolites and conjugates are primarily excreted by the kidneys, the potential exists for mefenamic acid metabolites to accumulate. Mefenamic acid should not be administered to patients with pre-existing renal disease or in patients with significantly impaired renal function.
CLINICAL STUDIES
In controlled, double-blind, clinical trials, mefenamic acid was evaluated for the treatment of primary spasmodic dysmenorrhea. The parameters used in determining efficacy included pain assessment by both patient and investigator; the need for concurrent analgesic medication; and evaluation of change in frequency and severity of symptoms characteristic of spasmodic dysmenorrhea. Patients received either mefenamic acid, 500 mg (2 capsules) as an initial dose of 250 mg every 6 hours, or placebo at onset of bleeding or of pain, whichever began first. After three menstrual cycles, patients were crossed over to the alternate treatment for an additional three cycles. Mefenamic acid was significantly superior to placebo in all parameters, and both treatments (drug and placebo) were equally tolerated.
HOW SUPPLIED
Mefenamic acid is available as 250 mg green-banded, yellow capsules, imprinted with "BP" on the cap and "629" on the body in green ink. Bottles of 30, NDC# 0276-0509-30.
Dispense in a tight container as defined in the USP.
Storage
Store at 20°-25°C (68°-77°F); excursions permitted to 15°-30°C (59°-86°F) [See USP Controlled Room Temperature].
Mefenamic Acid PubMed™ news
- Journal Article • 2026 MayPharmaceuticals, personal care-products and current-use pesticides: a review of the available data from European seas.
- Journal Article • 2026 MayResponse of microbial interactions in hybrid biofilm system with low organic loading to micropollutant removal.
- Journal Article • 2026 AprAcute pharmaceutical poisoning as a cause of seizure events: a database analysis (2011-2023).
- Journal Article • 2026 AprEfficacy of Plant-Derived Therapies for Primary Dysmenorrhea: A Systematic Review and Meta-Analysis of Randomized Controlled Trials.
- Journal Article • 2026 AprSynthesis and Structure Elucidation of New NSAID Diphosphine Ruthenium(II) Complexes as Potential Anticancer Agents: DNA/BSA Binding and Cytotoxicity Assays.