Get your patient on Maxidex - Dexamethasone for Suspension (Dexamethasone)
Maxidex - Dexamethasone for Suspension prescribing information
INDICATIONS AND USAGE
Steroid responsive inflammatory conditions of the palpebral and bulbar conjunctiva, cornea, and anterior segment of the globe, such as allergic conjunctivitis, acne rosacea, superficial punctate keratitis, herpes zoster keratitis, iritis, cyclitis, selected infective conjunctivitides when the inherent hazard of steroid use is accepted to obtain an advisable diminution in edema and inflammation; corneal injury from chemical, radiation, or thermal burns, or penetration of foreign bodies.
DOSAGE AND ADMINISTRATION
SHAKE WELL BEFORE USING. One or two drops topically in the conjunctival sac(s). In severe disease, drops may be used hourly, being tapered to discontinuation as the inflammation subsides. In mild disease, drops may be used up to four to six times daily.
Not more than one bottle should be prescribed initially, and the prescription should not be refilled without further evaluation as outlined in PRECAUTIONS above.
CONTRAINDICATIONS
Contraindicated in acute, untreated bacterial infections; mycobacterial ocular infections; epithelial herpes simplex (dendritic keratitis); vaccinia, varicella, and most other viral diseases of the cornea and conjunctiva; fungal disease of ocular structures; and in those persons who have shown hypersensitivity to any component of this preparation.
ADVERSE REACTIONS
Glaucoma with optic nerve damage, visual acuity and field defects; cataract formation; secondary ocular infection following suppression of host response; and perforation of the globe may occur.
Clinical Studies Experience
In clinical studies with MAXIDEX, the most frequently reports adverse reactions were ocular discomfort occurring in approximately 10% of the patients and eye irritation occurring in approximately 1% of the patients. All other adverse reactions from these studies occurred with a frequency less than 1%, including keratitis, conjunctivitis, dry eye, photophobia, blurred vision, eye pruritis, foreign body sensation, increased lacrimation, abnormal ocular sensation, eyelid margin crusting, and ocular hyperemia.
Postmarketing Experience
Additional adverse reactions identified from post-marketing use include corneal erosion, dizziness, eye pain, eyelid ptosis, headache, hypersensitivity reactions, and mydriasis. Frequencies cannot be estimated from the available data.
The following additional adverse reactions have been reported with dexamethasone use:
Cushing's syndrome and adrenal suppression may occur after use of dexamethasone in excess of the listed dosing instructions in predisposed patients, including children and patients treated with CYP3A4 inhibitors.
DESCRIPTION
MAXIDEX (dexamethasone ophthalmic suspension) 0.1% is an adrenocortical steroid prepared as a sterile topical ophthalmic suspension. The active ingredient is represented by the chemical structure:
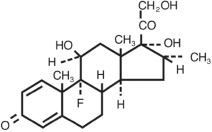
Chemical name: Pregna-1,4-diene-3,20-dione,9-fluoro-11,17,21-trihydroxy-16-methyl-,(11β,16α)-.
Each mL of MAXIDEX (dexamethasone ophthalmic suspension) 0.1% contains: Active: dexamethasone 0.1%. Preservative: benzalkonium chloride 0.01%. Vehicle: hypromellose 0.5%. Inactives: citric acid and/or sodium hydroxide (to adjust pH), dibasic sodium phosphate, edetate disodium, polysorbate 80, purified water, and sodium chloride.
CLINICAL PHARMACOLOGY
Dexamethasone suppresses the inflammatory response to a variety of agents and it probably delays or slows healing.
Clinical Studies Experience
In clinical studies with MAXIDEX, the most frequently reports adverse reactions were ocular discomfort occurring in approximately 10% of the patients and eye irritation occurring in approximately 1% of the patients. All other adverse reactions from these studies occurred with a frequency less than 1%, including keratitis, conjunctivitis, dry eye, photophobia, blurred vision, eye pruritis, foreign body sensation, increased lacrimation, abnormal ocular sensation, eyelid margin crusting, and ocular hyperemia.
HOW SUPPLIED
MAXIDEX ® (dexamethasone ophthalmic suspension) 0.1% in plastic dispensers:
5 mL NDC 82667-600-05