Get your patient on Loxicom (Meloxicam)
Loxicom prescribing information
Warning: Repeated use of meloxicam in cats has been associated with acute renal failure and death. Do not administer additional injectable or oral meloxicam to cats. See Contraindications , Warnings , and Precautions for detailed information.
Indications:
Loxicom oral suspension is indicated for the control of pain and inflammation associated with osteoarthritis in dogs.
Dosage and Administration:
Always provide client information sheet with prescription. Carefully consider the potential benefits and risks of Loxicom and other treatment options before deciding to use Loxicom. Use the lowest effective dose for the shortest duration consistent with individual response. Loxicom oral suspension should be administered initially at 0.09 mg/lb (0.2 mg/kg) body weight only on the first day of treatment.
For all treatments after day 1, Loxicom oral suspension should be administered once daily at a dose of 0.045 mg/lb (0.1 mg/kg). The provided syringes are calibrated to deliver the daily maintenance dose in pounds.
Contraindications:
Dogs with known hypersensitivity to meloxicam should not receive Loxicom oral suspension. Do not use Loxicom oral suspension in cats. Acute renal failure and death have been associated with the use of meloxicam in cats.
Adverse Reactions:
Field safety was evaluated in 306 dogs. Based on the results of two studies, GI abnormalities (vomiting, soft stools, diarrhea, and inappetance) were the most common adverse reactions associated with the administration of meloxicam. The following table lists adverse reactions and the numbers of dogs that experienced them during the studies. Dogs may have experienced more than one episode of the adverse reaction during the study.
| Adverse Reactions Observed During Two Field Studies | ||
| Clinical Observation | Meloxicam (n=157) | Placebo (n=149) |
| Vomiting | 40 | 23 |
| Diarrhea/Soft Stool | 19 | 11 |
| Bloody Stool | 1 | 0 |
| Inappetance | 5 | 1 |
| Bleeding gums after dental procedure | 1 | 0 |
| Lethargy/Swollen Carpus | 1 | 0 |
| Epiphora | 1 | 0 |
In foreign suspected adverse drug reaction (SADR) reporting over a 9 year period, incidences of adverse reactions related to meloxicam administration included: auto-immune hemolytic anemia (1 dog), thrombocytopenia (1 dog), polyarthritis (1 dog), nursing puppy lethargy (1 dog), and pyoderma (1 dog).
Description:
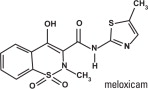
Meloxicam is a non-steroidal anti-inflammatory drug (NSAID) of the oxicam class. Each milliliter of Loxicom ® oral suspension contains meloxicam equivalent to 0.5 milligrams and sodium benzoate (1.5 milligrams) as a preservative. The chemical name for Meloxicam is 4-Hydroxy-2-methyl-N- (5-methyl-2-thiazolyl)-2H-1,2-benzothiazine-3-carboxamide-1, 1-dioxide. The formulation is a yellowish viscous suspension.
Clinical Pharmacology:
Meloxicam has nearly 100% bioavailability when administered orally with food. The terminal elimination half-life after a single dose is estimated to be approximately 24 hrs (+/-30%) regardless of route of administration. There is no evidence of statistically significant gender differences in drug pharmacokinetics. Drug bioavailability, volume of distribution, and total systemic clearance remain constant up to 5 times the recommended dose for use in dogs. However, there is some evidence of enhanced drug accumulation and terminal elimination half-life prolongation when dogs are dosed for 45 days or longer. Peak drug concentrations can be expected to occur within about 7.5 hrs after oral administration. Corresponding peak concentration is approximately 0.464 mcg/mL following a 0.2 mg/kg oral dose. The drug is 97% bound to canine plasma proteins.
How Supplied:
Loxicom oral suspension 0.5 mg/mL: 15 mL and 30 mL bottles with 1 mL and 3 mL measuring syringes.
NDC 55529-042-13 - 0.5 mg/mL - 15 mL
NDC 55529-042-14 - 0.5 mg/mL - 30 mL
Storage: Store at controlled room temperature 68-77°F (20-25°C). Excursions permitted between 59°F and 86°F (15°C and 30°C). Brief exposure to temperature up to 104°F (40°C) may be tolerated provided the mean kinetic temperature does not exceed 77°F (25°C); however such exposure should be minimized.
Approved by FDA under ANADA # 200-786
Made in the UK.
Manufactured by: Norbrook Laboratories Limited Newry, BT35 6QQ, Co. Down, Northern Ireland
Loxicom ® is a registered trademark of Norbrook Laboratories Limited
U.S. Patent No. 9,399,013
TAKE TIME

OBSERVE LABEL
DIRECTIONS
Rev. 02/26
624670I02
Norbrook ®