Get your patient on Linagliptin
Linagliptin prescribing information
1 INDICATIONS AND USAGE
Linagliptin is a dipeptidyl peptidase-4 (DPP-4) inhibitor indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus (1 )
Limitations of Use
Linagliptin tablet is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus [see Clinical Studies (14.1) ] .
Limitations of Use
Linagliptin should not be used in patients with type 1 diabetes or for the treatment of diabetic ketoacidosis, as it would not be effective in these settings.
Linagliptin has not been studied in patients with a history of pancreatitis. It is unknown whether patients with a history of pancreatitis are at an increased risk for the development of pancreatitis while using linagliptin [see Warnings and Precautions (5.1) ].
3 DOSAGE FORMS AND STRENGTHS
Linagliptin 5 mg tablets are white or off-white, round, biconvex, film-coated tablets with “S75” debossed on one side and blank on the other side.
8 USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
The limited data with linagliptin use in pregnant women are not sufficient to inform of drug-associated risk for major birth defects and miscarriage. There are risks to the mother and fetus associated with poorly controlled diabetes in pregnancy [see Clinical Considerations ].
In animal reproduction studies, no adverse developmental effects were observed when linagliptin was administered to pregnant rats during the period of organogenesis at doses similar to the maximum recommended clinical dose, based on exposure [see Data ] .
The estimated background risk of major birth defects is 6 to 10% in women with pre-gestational diabetes with a HbA1c>7 and has been reported to be as high as 20 to 25% in women with HbA1c>10. The estimated background risk of miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Clinical Considerations
Disease-associated maternal and/or embryo/fetal risk
Poorly controlled diabetes in pregnancy increases the maternal risk for diabetic ketoacidosis, pre-eclampsia, spontaneous abortions, preterm delivery, and delivery complications. Poorly controlled diabetes increases the fetal risk for major birth defects, stillbirth, and macrosomia related morbidity.
Data
Animal Data
No adverse developmental outcome was observed when linagliptin was administered to pregnant Wistar Han rats and Himalayan rabbits during the period of organogenesis at doses up to 240 mg/kg and 150 mg/kg, respectively. These doses represent approximately 943 times (rats) and 1943 times (rabbits) the 5 mg clinical dose, based on exposure. No adverse functional, behavioral, or reproductive outcome was observed in offspring following administration of linagliptin to Wistar Han rats from gestation day 6 to lactation day 21 at a dose 49 times the 5 mg clinical dose, based on exposure.
Lactation
Risk Summary
There is no information regarding the presence of linagliptin in human milk, the effects on the breastfed infant, or the effects on milk production. However, linagliptin is present in rat milk. Therefore, the developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for linagliptin tablets and any potential adverse effects on the breastfed child from linagliptin tablets or from the underlying maternal condition.
8.4 Pediatric Use
Safety and effectiveness of linagliptin in pediatric patients under 18 years of age have not been established.
8.5 Geriatric Use
In the 15 type 2 diabetes studies with linagliptin, 1085 linagliptin-treated patients were 65 years of age and older (including 131 linagliptin-treated patients 75 years of age and older). Of these 15 studies, 12 were double-blind placebo-controlled. In these 12 studies, 591 linagliptin-treated patients were 65 years of age and older (including 82 linagliptin-treated patients 75 years of age and older). In these linagliptin studies, no overall differences in safety or effectiveness of linagliptin were observed between geriatric patients and younger adult patients.
8.6 Renal Impairment
No dose adjustment is recommended for patients with renal impairment [see Clinical Pharmacology (12.3) ] .
8.7 Hepatic Impairment
No dose adjustment is recommended for patients with hepatic impairment [see Clinical Pharmacology (12.3) ] .
4 CONTRAINDICATIONS
Linagliptin is contraindicated in patients with a history of a hypersensitivity reaction to linagliptin, such as anaphylaxis, angioedema, exfoliative skin conditions, urticaria, or bronchial Linagliptin is contraindicated in patients with hypersensitivity to linagliptin or any of the excipients in linagliptin tablets, reactions such as anaphylaxis, angioedema, exfoliative skin conditions, urticaria, or bronchial hyperreactivity have occurred [see Warnings and Precautions (5.4) and Adverse Reactions (6) ] .
5 WARNINGS AND PRECAUTIONS
- Pancreatitis: There have been reports of acute pancreatitis, including fatal pancreatitis. If pancreatitis is suspected, promptly discontinue linagliptin. (5.1)
- Heart failure: Heart failure has been observed with two other members of the DPP-4 inhibitor class. Consider risks and benefits of linagliptin in patients who have known risk factors for heart failure. Monitor for signs and symptoms. (5.2)
- Hypoglycemia: When used with an insulin secretagogue (e.g., sulfonylurea (SU)) or insulin, consider lowering the dose of the insulin secretagogue or insulin to reduce the risk of hypoglycemia (5.3)
- Hypersensitivity reactions: Serious hypersensitivity reactions (e.g., anaphylaxis, angioedema, and exfoliative skin conditions) have occurred with linagliptin. If hypersensitivity reactions occur, discontinue linagliptin, treat promptly, and monitor until signs and symptoms resolve. (5.4)
- Arthralgia: Severe and disabling arthralgia has been reported in patients taking DPP-4 inhibitors. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. (5.5)
- Bullous pemphigoid: There have been reports of bullous pemphigoid requiring hospitalization. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue linagliptin (5.6) .
5.1 Pancreatitis
Acute pancreatitis, including fatal pancreatitis, has been reported in patients treated with linagliptin.
Take careful notice of potential signs and symptoms of pancreatitis. If pancreatitis is suspected, promptly discontinue linagliptin and initiate appropriate management. It is unknown whether patients with a history of pancreatitis are at increased risk for the development of pancreatitis while using linagliptin.
5.2 Heart Failure
An association between DPP-4 inhibitor treatment and heart failure has been observed in cardiovascular outcomes trials for two other members of the DPP-4 inhibitor class. These trials evaluated patients with type 2 diabetes mellitus and atherosclerotic cardiovascular disease.
Consider the risks and benefits of linagliptin prior to initiating treatment in patients at risk for heart failure, such as those with a prior history of heart failure and a history of renal impairment, and observe these patients for signs and symptoms of heart failure during therapy. Advise patients of the characteristic symptoms of heart failure and to immediately report such symptoms. If heart failure develops, evaluate and manage according to current standards of care and consider discontinuation of linagliptin.
5.3 Use with Medications Known to Cause Hypoglycemia
Insulin secretagogues and insulin are known to cause hypoglycemia. The use of linagliptin in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin was associated with a higher rate of hypoglycemia compared with placebo in clinical trials [see Adverse Reactions (6.1) ] . The use of linagliptin in combination with insulin in subjects with severe renal impairment was associated with a higher rate of hypoglycemia [see Adverse Reactions (6.1) ] . Therefore, a lower dose of the insulin secretagogue or insulin may be required to reduce the risk of hypoglycemia when used in combination with linagliptin.
5.4 Hypersensitivity Reactions
There have been postmarketing reports of serious hypersensitivity reactions in patients treated with linagliptin. These reactions include anaphylaxis, angioedema, and exfoliative skin conditions. Onset of these reactions predominantly occurred within the first 3 months after initiation of treatment with linagliptin, with some reports occurring after the first dose. If a serious hypersensitivity reaction is suspected, discontinue linagliptin, assess for other potential causes for the event, and institute alternative treatment for diabetes.
Angioedema has also been reported with other dipeptidyl peptidase-4 (DPP-4) inhibitors. Use caution in a patient with a history of angioedema to another DPP-4 inhibitor because it is unknown whether such patients will be predisposed to angioedema with linagliptin.
5.5 Severe and Disabling Arthralgia
There have been postmarketing reports of severe and disabling arthralgia in patients taking DPP-4 inhibitors. The time to onset of symptoms following initiation of drug therapy varied from one day to years. Patients experienced relief of symptoms upon discontinuation of the medication. A subset of patients experienced a recurrence of symptoms when restarting the same drug or a different DPP-4 inhibitor. Consider DPP-4 inhibitors as a possible cause for severe joint pain and discontinue drug if appropriate.
5.6 Bullous Pemphigoid
Postmarketing cases of bullous pemphigoid requiring hospitalization have been reported with DPP-4 inhibitor use. In reported cases, patients typically recovered with topical or systemic immunosuppressive treatment and discontinuation of the DPP-4 inhibitor. Tell patients to report development of blisters or erosions while receiving linagliptin. If bullous pemphigoid is suspected, linagliptin should be discontinued and referral to a dermatologist should be considered for diagnosis and appropriate treatment.
6 ADVERSE REACTIONS
The following serious adverse reactions are described below or elsewhere in the prescribing information:
- Pancreatitis [see Warnings and Precautions (5.1) ]
- Heart Failure [see Warnings and Precautions (5.2) ]
- Use with Medications Known to Cause Hypoglycemia [see Warnings and Precautions (5.3) ]
- Hypersensitivity Reactions [see Warnings and Precautions (5.4) ]
- Severe and Disabling Arthralgia [see Warnings and Precautions (5.5) ]
- Bullous Pemphigoid [see Warnings and Precautions (5.6) ]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety evaluation of linagliptin 5 mg once daily in patients with type 2 diabetes is based on 14 placebo-controlled trials, 1 active-controlled study, and one study in patients with severe renal impairment. In the 14 placebo-controlled studies, a total of 3625 patients were randomized and treated with linagliptin 5 mg daily and 2176 with placebo. The mean exposure in patients treated with linagliptin across studies was 29.6 weeks. The maximum follow-up was 78 weeks.
Linagliptin 5 mg once daily was studied as monotherapy in three placebo-controlled trials of 18 and 24 weeks’ duration and in five additional placebo-controlled studies lasting ≤18 weeks. The use of linagliptin in combination with other antihyperglycemic agents was studied in six placebo-controlled trials: two with metformin (12 and 24 weeks’ treatment duration); one with a sulfonylurea (18 weeks’ treatment duration); one with metformin and sulfonylurea (24 weeks’ treatment duration); one with pioglitazone (24 weeks’ treatment duration); and one with insulin (primary endpoint at 24 weeks).
In a pooled dataset of 14 placebo-controlled clinical trials, adverse reactions that occurred in ≥2% of patients receiving linagliptin (n = 3625) and more commonly than in patients given placebo (n = 2176), are shown in Table 1. The overall incidence of adverse events with linagliptin were similar to placebo.
| Number (%) of Patients | ||
| Linagliptin 5 mg n = 3625 | Placebo n = 2176 | |
| Nasopharyngitis | 254 (7.0) | 132 (6.1) |
| Diarrhea | 119 (3.3) | 65 (3.0) |
| Cough | 76 (2.1) | 30 (1.4) |
Rates for other adverse reactions for linagliptin 5 mg vs placebo when linagliptin was used in combination with specific anti-diabetic agents were: urinary tract infection (3.1% vs 0%) and hypertriglyceridemia (2.4% vs 0%) when linagliptin was used as add-on to sulfonylurea; hyperlipidemia (2.7% vs 0.8%) and weight increased (2.3% vs 0.8%) when linagliptin was used as add-on to pioglitazone; and constipation (2.1% vs 1%) when linagliptin was used as add-on to basal insulin therapy. Other adverse reactions reported in clinical studies with treatment of linagliptin were hypersensitivity (e.g., urticaria, angioedema, localized skin exfoliation, or bronchial hyperreactivity) and myalgia.
Following 104 weeks’ treatment in a controlled study comparing linagliptin with glimepiride in which all patients were also receiving metformin, adverse reactions reported in ≥5% of patients treated with linagliptin (n = 776) and more frequently than in patients treated with a sulfonylurea (n = 775) were back pain (9.1% vs 8.4%), arthralgia (8.1% vs 6.1%), upper respiratory tract infection (8.0% vs 7.6%), headache (6.4% vs 5.2%), cough (6.1% vs 4.9%), and pain in extremity (5.3% vs 3.9%).
In the clinical trial program, pancreatitis was reported in 15.2 cases per 10,000 patient year exposure while being treated with linagliptin compared with 3.7 cases per 10,000 patient year exposure while being treated with comparator (placebo and active comparator, sulfonylurea). Three additional cases of pancreatitis were reported following the last administered dose of linagliptin.
Hypoglycemia
Table 2 summarizes the incidence of hypoglycemia in placebo-controlled studies of linagliptin. The incidence of hypoglycemia increased when linagliptin was administered with sulfonylurea or insulin.
| •Hypoglycemia requiring assistance of another person to actively administer carbohydrate, glucagon, or other resuscitative actions. | ||
| Add-on to Sulfonylurea (18 Weeks) | Placebo (N=84) | Linagliptin (N=161) |
| Hypoglycemia with plasma glucose <54 mg/dL | 1.2% | 1.9% |
| Severe• hypoglycemia (%) | 0% | 0% |
| Add-on to Metformin and Sulfonylurea (24 Weeks) | Placebo (N=263) | Linagliptin (N=792) |
| Hypoglycemia with plasma glucose <54 mg/dL | 5.3% | 8.1% |
| Severe• hypoglycemia (%) | 0.8% | 0.6% |
| Add-on to Basal Insulin (52 Weeks) | Placebo (N=630) | Linagliptin (N=631) |
| Hypoglycemia with plasma glucose <54 mg/dL | 21.6% | 19.8% |
| Severe• hypoglycemia (%) | 1.1% | 1.7% |
Use in Renal Impairment
Linagliptin was compared to placebo as add-on to pre-existing antidiabetic therapy over 52 weeks in 133 patients with severe renal impairment (estimated GFR <30 mL/min). For the initial 12 weeks of the study, background antidiabetic therapy was kept stable and included insulin, sulfonylurea, glinides, and pioglitazone. For the remainder of the trial, dose adjustments in antidiabetic background therapy were allowed.
In general, the incidence of adverse events including severe hypoglycemia was similar to those reported in other linagliptin trials. The observed incidence of hypoglycemia was higher (linagliptin, 63% compared to placebo, 49%) due to an increase in asymptomatic hypoglycemic events especially during the first 12 weeks when background glycemic therapies were kept stable. Ten linagliptin-treated patients (15%) and 11 placebo-treated patients (17%) reported at least one episode of confirmed symptomatic hypoglycemia (accompanying finger stick glucose ≤54 mg/dL). During the same time period, severe hypoglycemic events, defined as an event requiring the assistance of another person to actively administer carbohydrate, glucagon or other resuscitative actions, were reported in 3 (4.4%) linagliptin-treated patients and 3 (4.6%) placebo-treated patients. Events that were considered life-threatening or required hospitalization were reported in 2 (2.9%) patients on linagliptin and 1 (1.5%) patient on placebo.
Renal function as measured by mean eGFR and creatinine clearance did not change over 52 weeks' treatment compared to placebo.
Laboratory Tests
Changes in laboratory findings were similar in patients treated with linagliptin 5 mg compared to patients treated with placebo.
Increase in Uric Acid: Changes in laboratory values that occurred more frequently in the linagliptin group and > 1% more than in the placebo group were increases in uric acid (1.3% in the placebo group, 2.7% in the linagliptin group).
Increase in Lipase: In a placebo-controlled clinical trial with linagliptin in type 2 diabetes mellitus patients with micro- or macroalbuminuria, a mean increase of 30% in lipase concentrations from baseline to 24 weeks was observed in the linagliptin arm compared to a mean decrease of 2% in the placebo arm. Lipase levels above 3 times upper limit of normal were seen in 8.2% compared to 1.7% patients in the linagliptin and placebo arms, respectively.
The clinical significance of elevations in lipase with linagliptin is unknown in the absence of potential signs and symptoms of pancreatitis [see Warnings and Precautions (5.1) ].
Vital Signs No clinically meaningful changes in vital signs were observed in patients treated with linagliptin.
6.2 Postmarketing Experience
Additional adverse reactions have been identified during postapproval use of linagliptin. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Acute pancreatitis, including fatal pancreatitis [see Indications and Usage (1) ]
- Hypersensitivity reactions including anaphylaxis, angioedema, and exfoliative skin conditions
- Severe and disabling arthralgia
- Bullous pemphigoid
- Rash
- Mouth ulceration, stomatitis
- Rhabdomyolysis
7 DRUG INTERACTIONS
Strong P-glycoprotein/CYP3A4 inducer: The efficacy of linagliptin may be reduced when administered in combination (e.g., with rifampin). Use of alternative treatments is strongly recommended. (7.1)
7.1 Inducers of P-glycoprotein or CYP3A4 Enzymes
Rifampin decreased linagliptin exposure, suggesting that the efficacy of linagliptin may be reduced when administered in combination with a strong P-gp or CYP3A4 inducer. Therefore, use of alternative treatments is strongly recommended when linagliptin is to be administered with a strong P-gp or CYP3A4 inducer [see Clinical Pharmacology (12.3) ] .
7.2 Insulin Secretagogues or Insulin
Coadministration of linagliptin with an insulin secretagogue (e.g., sulfonylurea) or insulin may require lower doses of the insulin secretagogue or insulin to reduce the risk of hypoglycemia [see Warnings and Precautions (5.3) ] .
11 DESCRIPTION
Linagliptin tablets contain, as the active ingredient, an orally-active inhibitor of the dipeptidyl peptidase-4 (DPP-4) enzyme.
Linagliptin is described chemically as 1H-Purine-2,6-dione, 8-[(3R)-3-amino-1-piperidinyl]-7-(2-butyn-1-yl)-3,7-dihydro-3-methyl-1-[(4-methyl-2-quinazolinyl)methyl]-
The empirical formula is C 25 H 28 N 8 O 2 and the molecular weight is 472.54 g/mol. The structural formula is:
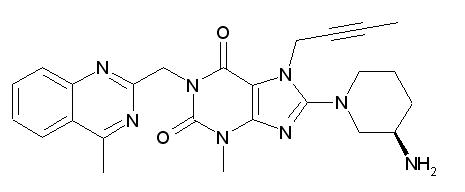
Linagliptin is a white to yellowish, not or only slightly hygroscopic solid substance. It is very slightly soluble in water (0.9 mg/mL). Linagliptin is soluble in methanol (ca. 60 mg/mL), sparingly soluble in ethanol (ca. 10 mg/mL), very slightly soluble in isopropanol (<1 mg/mL), and very slightly soluble in acetone (ca. 1 mg/mL).
Each film-coated tablet of linagliptin contains 5 mg of linagliptin free base and the following inactive ingredients: mannitol, pregelatinized starch, copovidone, and magnesium stearate. In addition, the film coating contains the following inactive ingredients: hypromellose, titanium dioxide and polyethylene glycol.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Linagliptin is an inhibitor of DPP-4, an enzyme that degrades the incretin hormones glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). Thus, linagliptin increases the concentrations of active incretin hormones, stimulating the release of insulin in a glucose-dependent manner and decreasing the levels of glucagon in the circulation. Both incretin hormones are involved in the physiological regulation of glucose homeostasis. Incretin hormones are secreted at a low basal level throughout the day and levels rise immediately after meal intake. GLP-1 and GIP increase insulin biosynthesis and secretion from pancreatic beta cells in the presence of normal and elevated blood glucose levels. Furthermore, GLP-1 also reduces glucagon secretion from pancreatic alpha-cells, resulting in a reduction in hepatic glucose output.
12.2 Pharmacodynamics
Linagliptin binds to DPP-4 in a reversible manner and thus increases the concentrations of incretin hormones. Linagliptin glucose dependently increases insulin secretion and lowers glucagon secretion, thus resulting in better regulation of glucose homeostasis. Linagliptin binds selectively to DPP-4, and selectively inhibits DPP-4 but not DPP-8 or DPP-9 activity in vitro at concentrations approximating therapeutic exposures.
Cardiac Electrophysiology
In a randomized, placebo-controlled, active-comparator, 4-way crossover study, 36 healthy subjects were administered a single oral dose of linagliptin 5 mg, linagliptin 100 mg (20 times the recommended dose), moxifloxacin, and placebo. No increase in QTc was observed with either the recommended dose of 5 mg or the 100-mg dose. At the 100-mg dose, peak linagliptin plasma concentrations were approximately 38-fold higher than the peak concentrations following a 5-mg dose.
12.3 Pharmacokinetics
The pharmacokinetics of linagliptin has been characterized in healthy subjects and patients with type 2 diabetes. After oral administration of a single 5-mg dose to healthy subjects, peak plasma concentrations of linagliptin occurred at approximately 1.5 hours post dose (T max ); the mean plasma area under the curve (AUC) was 139 nmol•h/L and maximum concentration (C max ) was 8.9 nmol/L.
Plasma concentrations of linagliptin decline in at least a biphasic manner with a long terminal half-life (>100 hours), related to the saturable binding of linagliptin to DPP-4. The prolonged elimination phase does not contribute to the accumulation of the drug. The effective half-life for accumulation of linagliptin, as determined from oral administration of multiple doses of linagliptin 5 mg, is approximately 12 hours. After once-daily dosing, steady-state plasma concentrations of linagliptin 5 mg are reached by the third dose, and C max and AUC increased by a factor of 1.3 at steady-state compared with the first dose. The intra-subject and inter-subject coefficients of variation for linagliptin AUC were small (12.6% and 28.5%, respectively). Plasma AUC of linagliptin increased in a less than dose-proportional manner in the dose range of 1 to 10 mg. The pharmacokinetics of linagliptin is similar in healthy subjects and in patients with type 2 diabetes.
Absorption
The absolute bioavailability of linagliptin is approximately 30%. A high-fat meal reduced C max by 15% and increased AUC by 4%; this effect is not clinically relevant. Linagliptin may be administered with or without food.
Distribution
The mean apparent volume of distribution at steady state following a single intravenous dose of linagliptin 5 mg to healthy subjects is approximately 1110 L, indicating that linagliptin extensively distributes to the tissues. Plasma protein binding of linagliptin is concentration-dependent, decreasing from about 99% at 1 nmol/L to 75%-89% at ≥30 nmol/L, reflecting saturation of binding to DPP-4 with increasing concentration of linagliptin. At high concentrations, where DPP-4 is fully saturated, 70% to 80% of linagliptin remains bound to plasma proteins and 20% to 30% is unbound in plasma. Plasma binding is not altered in patients with renal or hepatic impairment.
Elimination
Linagliptin has a terminal half-life of about 200 hours at steady-state, though the accumulation half-life is about 11 hours. Renal clearance at steady-state was approximately 70 mL/min.
Metabolism
Following oral administration, the majority (about 90%) of linagliptin is excreted unchanged, indicating that metabolism represents a minor elimination pathway. A small fraction of absorbed linagliptin is metabolized to a pharmacologically inactive metabolite, which shows a steady-state exposure of 13.3% relative to linagliptin.
Excretion
Following administration of an oral [ 14 C]-linagliptin dose to healthy subjects, approximately 85% of the administered radioactivity was eliminated via the enterohepatic system (80%) or urine (5%) within 4 days of dosing.
Specific Populations
Renal Impairment
An open-label pharmacokinetic study evaluated the pharmacokinetics of linagliptin 5 mg in male and female patients with varying degrees of chronic renal impairment. The study included 6 healthy subjects with normal renal function (creatinine clearance [CrCl] ≥80 mL/min), 6 patients with mild renal impairment (CrCl 50 to <80 mL/min), 6 patients with moderate renal impairment (CrCl 30 to <50 mL/min), 10 patients with type 2 diabetes mellitus and severe renal impairment (CrCl <30 mL/min), and 11 patients with type 2 diabetes mellitus and normal renal function. Creatinine clearance was measured by 24-hour urinary creatinine clearance measurements or estimated from serum creatinine based on the Cockcroft-Gault formula.
Under steady-state conditions, linagliptin exposure in patients with mild renal impairment was comparable to healthy subjects.
In patients with moderate renal impairment under steady-state conditions, mean exposure of linagliptin increased (AUC τ,ss by 71% and C max by 46%) compared with healthy subjects. This increase was not associated with a prolonged accumulation half-life, terminal half-life, or an increased accumulation factor. Renal excretion of linagliptin was below 5% of the administered dose and was not affected by decreased renal function.
Patients with type 2 diabetes mellitus and severe renal impairment showed steady-state exposure approximately 40% higher than that of patients with type 2 diabetes mellitus and normal renal function (increase in AUC τ,ss by 42% and C max by 35%). For both type 2 diabetes mellitus groups, renal excretion was below 7% of the administered dose.
These findings were further supported by the results of population pharmacokinetic analyses.
Hepatic Impairment
In patients with mild hepatic impairment (Child-Pugh class A), steady-state exposure (AUC τ,ss ) of linagliptin was approximately 25% lower and C max,ss was approximately 36% lower than in healthy subjects. In patients with moderate hepatic impairment (Child-Pugh class B), AUC ss of linagliptin was about 14% lower and C max,ss was approximately 8% lower than in healthy subjects. Patients with severe hepatic impairment (Child-Pugh class C) had comparable exposure of linagliptin in terms of AUC 0-24 and approximately 23% lower C max compared with healthy subjects. Reductions in the pharmacokinetic parameters seen in patients with hepatic impairment did not result in reductions in DPP-4 inhibition.
Body Mass Index (BMI)/Weight
No dose adjustment is necessary based on BMI/weight. BMI/weight had no clinically meaningful effect on the pharmacokinetics of linagliptin based on a population pharmacokinetic analysis.
Gender
No dose adjustment is necessary based on gender. Gender had no clinically meaningful effect on the pharmacokinetics of linagliptin based on a population pharmacokinetic analysis.
Geriatric
Age did not have a clinically meaningful impact on the pharmacokinetics of linagliptin based on a population pharmacokinetic analysis.
Pediatric
Studies characterizing the pharmacokinetics of linagliptin in pediatric patients have not yet been performed.
Race
No dose adjustment is necessary based on race. Race had no clinically meaningful effect on the pharmacokinetics of linagliptin based on available pharmacokinetic data, including subjects of White, Hispanic, Black, and Asian racial groups.
Drug Interactions
In vitro Assessment of Drug Interactions
Linagliptin is a weak to moderate inhibitor of CYP isozyme CYP3A4, but does not inhibit other CYP isozymes and is not an inducer of CYP isozymes, including CYP1A2, 2A6, 2B6, 2C8, 2C9, 2C19, 2D6, 2E1, and 4A11.
Linagliptin is a P-glycoprotein (P-gp) substrate, and inhibits P-gp mediated transport of digoxin at high concentrations. Based on these results and in vivo drug interaction studies, linagliptin is considered unlikely to cause interactions with other P-gp substrates at therapeutic concentrations.
In vivo Assessment of Drug Interactions
Strong inducers of CYP3A4 or P-gp (e.g., rifampin) decrease exposure to linagliptin to subtherapeutic and likely ineffective concentrations [see Drug Interactions (7) ] . In vivo studies indicated evidence of a low propensity for causing drug interactions with substrates of CYP3A4, CYP2C9, CYP2C8, P-gp and organic cationic transporter (OCT).
| Coadministered Drug | Dosing of Coadministered Drug• | Dosing of Linagliptin• | Geometric Mean Ratio (ratio with/without coadministered drug) No effect = 1.0 | |
|---|---|---|---|---|
| AUC † | C max | |||
| •Multiple dose (steady-state) unless otherwise noted ••For information regarding clinical recommendations [see Drug Interactions (7.1) ]. #Single dose † AUC = AUC(0 to 24 hours) for single-dose treatments and AUC = AUC(TAU) for multiple-dose treatments QD = once daily BID = twice daily TID = three times daily | ||||
| Metformin | 850 mg TID | 10 mg QD | 1.20 | 1.03 |
| Glyburide | 1.75 mg # | 5 mg QD | 1.02 | 1.01 |
| Pioglitazone | 45 mg QD | 10 mg QD | 1.13 | 1.07 |
| Ritonavir | 200 mg BID | 5 mg # | 2.01 | 2.96 |
| Rifampin•• | 600 mg QD | 5 mg QD | 0.60 | 0.56 |
| Coadministered Drug | Dosing of Coadministered Drug• | Dosing of Linagliptin• | Geometric Mean Ratio (ratio with/without coadministered drug) No effect = 1.0 | ||
|---|---|---|---|---|---|
| AUC † | C max | ||||
| •Multiple dose (steady-state) unless otherwise noted #Single dose † AUC=AUC(INF) for single-dose treatments and AUC = AUC(TAU) for multiple-dose treatments ••AUC=AUC(0-168) and C max =E max for pharmacodynamic end points INR = International Normalized Ratio PT = Prothrombin Time QD = once daily TID = three times daily | |||||
| Metformin | 850 mg TID | 10 mg QD | metformin | 1.01 | 0.89 |
| Glyburide | 1.75 mg # | 5 mg QD | glyburide | 0.86 | 0.86 |
| Pioglitazone | 45 mg QD | 10 mg QD | pioglitazone metabolite M-III metabolite M-IV | 0.94 0.98 1.04 | 0.86 0.96 1.05 |
| Digoxin | 0.25 mg QD | 5 mg QD | digoxin | 1.02 | 0.94 |
| Simvastatin | 40 mg QD | 10 mg QD | simvastatin simvastatin acid | 1.34 1.33 | 1.10 1.21 |
| Warfarin | 10 mg # | 5 mg QD | R-warfarin S-warfarin INR PT | 0.99 1.03 0.93•• 1.03•• | 1.00 1.01 1.04•• 1.15•• |
| Ethinylestradiol and levonorgestrel | ethinylestradiol 0.03 mg and levonorgestrel 0.150 mg QD | 5 mg QD | ethinylestradiol levonorgestrel | 1.01 1.09 | 1.08 1.13 |
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Linagliptin did not increase the incidence of tumors in male and female rats in a 2-year study at doses of 6, 18, and 60 mg/kg. The highest dose of 60 mg/kg is approximately 418 times the clinical dose of 5 mg/day based on AUC exposure. Linagliptin did not increase the incidence of tumors in mice in a 2-year study at doses up to 80 mg/kg (males) and 25 mg/kg (females), or approximately 35 and 270 times the clinical dose based on AUC exposure. Higher doses of linagliptin in female mice (80 mg/kg) increased the incidence of lymphoma at approximately 215 times the clinical dose based on AUC exposure.
Linagliptin was not mutagenic or clastogenic with or without metabolic activation in the Ames bacterial mutagenicity assay, a chromosomal aberration test in human lymphocytes, and an in vivo micronucleus assay.
In fertility studies in rats, linagliptin had no adverse effects on early embryonic development, mating, fertility, or bearing live young up to the highest dose of 240 mg/kg (approximately 943 times the clinical dose based on AUC exposure).
14 CLINICAL STUDIES
14.1 Glycemic Control Trials
Linagliptin has been studied as monotherapy and in combination with metformin, sulfonylurea, pioglitazone, and insulin. Linagliptin has also been studied in patients with type 2 diabetes and severe chronic renal impairment.
In patients with type 2 diabetes, treatment with linagliptin produced clinically significant improvements in hemoglobin A1c (A1C), fasting plasma glucose (FPG), and 2-hour post-prandial glucose (PPG) compared with placebo.
Monotherapy
A total of 730 patients with type 2 diabetes participated in 2 double-blind, placebo-controlled studies, one of 18 weeks’ and another of 24 weeks’ duration, to evaluate the efficacy and safety of linagliptin monotherapy. In both monotherapy studies, patients currently on an antihyperglycemic agent discontinued the agent and underwent a diet, exercise, and drug washout period of about 6 weeks that included an open-label placebo run-in during the last 2 weeks. Patients with inadequate glycemic control (A1C 7% to 10%) after the washout period were randomized; patients not currently on antihyperglycemic agents (off therapy for at least 8 weeks) with inadequate glycemic control (A1C 7% to 10%) were randomized after completing the 2-week, open-label, placebo run-in period. In the 18-week study, only patients ineligible for metformin were recruited. In the 18-week study, 76 patients were randomized to placebo and 151 to linagliptin 5 mg; in the 24-week study, 167 patients were randomized to placebo and 336 to linagliptin 5 mg. Patients who failed to meet specific glycemic goals during the 18-week study received rescue therapy with pioglitazone and/or insulin; metformin rescue therapy was used in the 24-week trial.
Treatment with linagliptin 5 mg daily provided statistically significant improvements in A1C, FPG, and 2-hour PPG compared with placebo (Table 5). In the 18-week study, 12% of patients receiving linagliptin 5 mg and 18% who received placebo required rescue therapy. In the 24-week study, 10.2% of patients receiving linagliptin 5 mg and 20.9% of patients receiving placebo required rescue therapy. The improvement in A1C compared with placebo was not affected by gender, age, race, prior antihyperglycemic therapy, baseline BMI, or a standard index of insulin resistance (HOMA-IR). As is typical for trials of agents to treat type 2 diabetes, the mean reduction in A1C with linagliptin appears to be related to the degree of A1C elevation at baseline. In these 18- and 24-week studies, the changes from baseline in A1C were -0.4% and -0.4%, respectively, for those given linagliptin, and 0.1% and 0.3%, respectively, for those given placebo. Change from baseline in body weight did not differ significantly between the groups.
| •Full analysis population using last observation on study ••18-week study: Placebo, n=68; linagliptin, n=136 24-week study: Placebo, n=147; linagliptin, n=306 •••18-week study. HbA1c: ANCOVA model included treatment, reason for metformin intolerance and number of prior oral anti-diabetic medicine(s) (OADs) as class-effects, as well as baseline HbA1c as continuous covariates. FPG: ANCOVA model included treatment, reason for metformin intolerance and number of prior OADs as class-effects, as well as baseline HbA1c and baseline FPG as continuous covariates. 24-week study. HbA1c: ANCOVA model included treatment and number of prior OADs as class-effects, as well as baseline HbA1c as continuous covariates. FPG: ANCOVA model included treatment and number of prior OADs as class-effects, as well as baseline HbA1c and baseline FPG as continuous covariates. PPG: ANCOVA model included treatment and number of prior OADs as class-effects, as well as baseline HbA1c and baseline postprandial glucose after two hours as covariate. | ||||
| 18-Week Study | 24-Week Study | |||
| Linagliptin 5 mg | Placebo | Linagliptin 5 mg | Placebo | |
| A1C (%) | ||||
| Number of patients | n = 147 | n = 73 | n = 333 | n = 163 |
| Baseline (mean) | 8.1 | 8.1 | 8.0 | 8.0 |
| Change from baseline (adjusted mean•••) | -0.4 | 0.1 | -0.4 | 0.3 |
| Difference from placebo (adjusted mean) (95% CI) | -0.6 (-0.9, -0.3) | -- | -0.7 (-0.9, -0.5) | -- |
| Patients [n (%)] achieving A1C <7%•• | 32 (23.5) | 8 (11.8) | 77 (25) | 17 (12) |
| FPG (mg/dL) | ||||
| Number of patients | n = 138 | n = 66 | n = 318 | n = 149 |
| Baseline (mean) | 178 | 176 | 164 | 166 |
| Change from baseline (adjusted mean•••) | -13 | 7 | -9 | 15 |
| Difference from placebo (adjusted mean) (95% CI) | -21 (-31, -10) | -- | -23 (-30, -16) | -- |
| 2-hour PPG (mg/dL) | ||||
| Number of patients | Data not available | Data not available | n = 67 | n = 24 |
| Baseline (mean) | -- | -- | 258 | 244 |
| Change from baseline (adjusted mean•••) | -- | -- | -34 | 25 |
| Difference from placebo (adjusted mean) (95% CI) | -- | -- | -58 (-82, -34) | -- |
Add-on Combination Therapy with Metformin
A total of 701 patients with type 2 diabetes participated in a 24-week, randomized, double-blind, placebo-controlled study designed to assess the efficacy of linagliptin in combination with metformin. Patients already on metformin (n = 491) at a dose of at least 1,500 mg per day were randomized after completing a 2-week, open-label, placebo run-in period. Patients on metformin and another antihyperglycemic agent (n = 207) were randomized after a run-in period of approximately 6 weeks on metformin (at a dose of at least 1,500 mg per day) in monotherapy. Patients were randomized to the addition of either linagliptin 5 mg or placebo, administered once daily. Patients who failed to meet specific glycemic goals during the studies were treated with glimepiride rescue.
In combination with metformin, linagliptin provided statistically significant improvements in A1C, FPG, and 2-hour PPG compared with placebo (Table 6). Rescue glycemic therapy was used in 7.8% of patients treated with linagliptin 5 mg and in 18.9% of patients treated with placebo. A similar decrease in body weight was observed for both treatment groups.
| •Full analysis population using last observation on study ••Linagliptin 5 mg + Metformin, n=485; Placebo + Metformin, n=163 •••HbA1c: ANCOVA model included treatment and number of prior oral OADs as class-effects, as well as baseline HbA1c as continuous covariates. FPG: ANCOVA model included treatment and number of prior OADs as class-effects, as well as baseline HbA1c and baseline FPG as continuous covariates. PPG: ANCOVA model included treatment and number of prior OADs as class-effects, as well as baseline HbA1c and baseline postprandial glucose after two hours as covariate. | ||
| Linagliptin 5 mg + Metformin | Placebo + Metformin | |
| A1C (%) | ||
| Number of patients | n = 513 | n = 175 |
| Baseline (mean) | 8.1 | 8.0 |
| Change from baseline (adjusted mean•••) | -0.5 | 0.15 |
| Difference from placebo + metformin (adjusted mean) (95% CI) | -0.6 (-0.8, -0.5) | -- |
| Patients [n (%)] achieving A1C <7%•• | 127 (26.2) | 15 (9.2) |
| FPG (mg/dL) | ||
| Number of patients | n = 495 | n = 159 |
| Baseline (mean) | 169 | 164 |
| Change from baseline (adjusted mean•••) | -11 | 11 |
| Difference from placebo + metformin (adjusted mean) (95% CI) | -21 (-27, -15) | -- |
| 2-hour PPG (mg/dL) | ||
| Number of patients | n = 78 | n = 21 |
| Baseline (mean) | 270 | 274 |
| Change from baseline (adjusted mean•••) | -49 | 18 |
| Difference from placebo + metformin (adjusted mean) (95% CI) | -67 (-95, -40) | -- |
Initial Combination Therapy with Metformin
A total of 791 patients with type 2 diabetes mellitus and inadequate glycemic control on diet and exercise participated in the 24-week, randomized, double-blind, portion of this placebo-controlled factorial study designed to assess the efficacy of linagliptin as initial therapy with metformin. Patients on an antihyperglycemic agent (52%) underwent a drug washout period of 4 weeks’ duration. After the washout period and after completing a 2-week single-blind placebo run-in period, patients with inadequate glycemic control (A1C ≥7.0% to ≤10.5%) were randomized. Patients with inadequate glycemic control (A1C ≥7.5% to <11.0%) not on antihyperglycemic agents at study entry (48%) immediately entered the 2-week, single-blind, placebo run-in period and then were randomized. Randomization was stratified by baseline A1C (<8.5% vs ≥8.5%) and use of a prior oral antidiabetic drug (none vs monotherapy). Patients were randomized in a 1:2:2:2:2:2 ratio to either placebo or one of 5 active-treatment arms. Approximately equal numbers of patients were randomized to receive initial therapy with 5 mg of linagliptin once daily, 500 mg or 1,000 mg of metformin twice daily, or 2.5 mg of linagliptin twice daily in combination with 500 mg or 1,000 mg of metformin twice daily. Patients who failed to meet specific glycemic goals during the study were treated with sulfonylurea, thiazolidinedione, or insulin rescue therapy.
Initial therapy with the combination of linagliptin and metformin provided significant improvements in A1C and fasting plasma glucose (FPG) compared to placebo, to metformin alone, and to linagliptin alone (Table 7).
The adjusted mean treatment difference in A1C from baseline to week 24 (LOCF) was -0.5% (95% CI -0.7, -0.3; p<0.0001) for linagliptin 2.5 mg/metformin 1,000 mg twice daily compared to metformin 1,000 twice daily; -1.1% (95% CI -1.4, -0.9; p<0.0001) for linagliptin 2.5 mg/metformin 1,000 mg twice daily compared to linagliptin 5 mg once daily; -0.6% (95% CI -0.8, -0.4; p<0.0001) for linagliptin 2.5 mg/metformin 500 mg twice daily compared to metformin 500 mg twice daily; and -0.8% (95% CI -1.0, -0.6; p<0.0001) for linagliptin 2.5 mg/metformin 500 mg twice daily compared to linagliptin 5 mg once daily.
Lipid effects were generally neutral. No meaningful change in body weight was noted in any of the 6 treatment groups.
| •Total daily dose of linagliptin is equal to 5 mg ••Full analysis population using last observation on study •••Metformin 500 mg twice daily, n=140; Linagliptin 2.5 mg twice daily + Metformin 500 twice daily, n=136; Metformin 1,000 mg twice daily, n=137; Linagliptin 2.5 mg twice daily + Metformin 1,000 mg twice daily, n=138 ••••HbA1c: ANCOVA model included treatment and number of prior OADs as class-effects, as well as baseline HbA1c as continuous covariates. FPG: ANCOVA model included treatment and number of prior OADs as class-effects, as well as baseline HbA1c and baseline FPG as continuous covariates. | ||||||
| Placebo | Linagliptin 5 mg Once Daily• | Metformin 500 mg Twice Daily | Linagliptin 2.5 mg Twice Daily• + Metformin 500 mg Twice Daily | Metformin 1,000 mg Twice Daily | Linagliptin 2.5 mg Twice Daily• + Metformin 1,000 mg Twice Daily | |
| A1C (%) | ||||||
| Number of patients | n = 65 | n = 135 | n = 141 | n = 137 | n = 138 | n = 140 |
| Baseline (mean) | 8.7 | 8.7 | 8.7 | 8.7 | 8.5 | 8.7 |
| Change from baseline (adjusted mean••••) | 0.1 | -0.5 | -0.6 | -1.2 | -1.1 | -1.6 |
| Difference from placebo (adjusted mean) (95% CI) | -- | -0.6 (-0.9, -0.3) | -0.8 (-1.0, -0.5) | -1.3 (-1.6, -1.1) | -1.2 (-1.5, -0.9) | -1.7 (-2.0, -1.4) |
| Patients [n (%)] achieving A1C <7%••• | 7 (10.8) | 14 (10.4) | 26 (18.6) | 41 (30.1) | 42 (30.7) | 74 (53.6) |
| Patients (%) receiving rescue medication | 29.2 | 11.1 | 13.5 | 7.3 | 8.0 | 4.3 |
| FPG (mg/dL) | ||||||
| Number of patients | n = 61 | n = 134 | n = 136 | n = 135 | n = 132 | n = 136 |
| Baseline (mean) | 203 | 195 | 191 | 199 | 191 | 196 |
| Change from baseline (adjusted mean••••) | 10 | -9 | -16 | -33 | -32 | -49 |
| Difference from placebo (adjusted mean) (95% CI) | -- | -19 (-31, -6) | -26 (-38, -14) | -43 (-56, -31) | -42 (-55, -30) | -60 (-72, -47) |
Active-Controlled Study vs Glimepiride in Combination with Metformin
The efficacy of linagliptin was evaluated in a 104-week, double-blind, glimepiride-controlled, non-inferiority study in patients with type 2 diabetes with insufficient glycemic control despite metformin therapy. Patients being treated with metformin only entered a run-in period of 2 weeks’ duration, whereas patients pretreated with metformin and one additional antihyperglycemic agent entered a run-in treatment period of 6 weeks’ duration with metformin monotherapy (dose of ≥1,500 mg/day) and washout of the other agent. After an additional 2-week placebo run-in period, those with inadequate glycemic control (A1C 6.5% to 10%) were randomized 1:1 to the addition of linagliptin 5 mg once daily or glimepiride. Randomization was stratified by baseline HbA1c (<8.5% vs ≥8.5%), and the previous use of antidiabetic drugs (metformin alone vs metformin plus one other OAD). Patients receiving glimepiride were given an initial dose of 1 mg/day and then electively titrated over the next 12 weeks to a maximum dose of 4 mg/day as needed to optimize glycemic control. Thereafter, the glimepiride dose was to be kept constant, except for down-titration to prevent hypoglycemia.
After 52 and 104 weeks, linagliptin and glimepiride both had reductions from baseline in A1C (52 weeks: -0.4% for linagliptin, -0.6% for glimepiride; 104 weeks: -0.2% for linagliptin, -0.4% for glimepiride) from a baseline mean of 7.7% (Table 8). The mean difference between groups in A1C change from baseline was 0.2% with 2-sided 97.5% confidence interval (0.1%, 0.3%) for the intent-to-treat population using last observation carried forward. These results were consistent with the completers analysis.
| •p<0.0001 vs glimepiride; † p=0.0012 vs glimepiride ••Full analysis population using last observation on study •••Hypoglycemic incidence included both asymptomatic events (not accompanied by typical symptoms and plasma glucose concentration of ≤70 mg/dL) and symptomatic events with typical symptoms of hypoglycemia and plasma glucose concentration of ≤70 mg/dL. ••••HbA1c: ANCOVA model included treatment and number of prior OADs as class-effects, as well as baseline HbA1c as continuous covariates. FPG: ANCOVA model included treatment and number of prior OADs as class-effects, as well as baseline HbA1c and baseline FPG as continuous covariates. Hypoglycemia incidence (%): Cochran-Mantel-Haenszel test was performed on the patient population contained in the treated set, to compare the proportion of patients with hypoglycemic events between patients treated with linagliptin and patients treated with glimepiride. | ||||
| Week 52 | Week 104 | |||
| Linagliptin 5 mg + Metformin | Glimepiride + Metformin (mean Glimepiride dose 3 mg) | Linagliptin 5 mg + Metformin | Glimepiride + Metformin (mean Glimepiride dose 3 mg) | |
| A1C (%) | ||||
| Number of patients | n = 764 | n = 755 | n = 764 | n = 755 |
| Baseline (mean) | 7.7 | 7.7 | 7.7 | 7.7 |
| Change from baseline (adjusted mean••••) | -0.4 | -0.6 | -0.2 | -0.4 |
| Difference from glimepiride (adjusted mean) (97.5% CI) | 0.2 (0.1, 0.3) | -- | 0.2 (0.1, 0.3) | -- |
| FPG (mg/dL) | ||||
| Number of patients | n = 733 | n = 725 | n = 733 | n = 725 |
| Baseline (mean) | 164 | 166 | 164 | 166 |
| Change from baseline (adjusted mean••••) | -8• | -15 | -2 † | -9 |
| Hypoglycemia incidence (%)••• | ||||
| Number of patients | n = 776 | n = 775 | n = 776 | n = 775 |
| Incidence•••• | 5.3 • | 31.1 | 7.5 • | 36.1 |
Patients treated with linagliptin had a mean baseline body weight of 86 kg and were observed to have an adjusted mean decrease in body weight of 1.1 kg at 52 weeks and 1.4 kg at 104 weeks. Patients on glimepiride had a mean baseline body weight of 87 kg and were observed to have an adjusted mean increase from baseline in body weight of 1.4 kg at 52 weeks and 1.3 kg at 104 weeks (treatment difference p<0.0001 for both timepoints).
Add-On Combination Therapy with Pioglitazone
A total of 389 patients with type 2 diabetes participated in a 24-week, randomized, double-blind, placebo-controlled study designed to assess the efficacy of linagliptin in combination with pioglitazone. Therapy was stopped in patients on oral antihyperglycemic therapy for a period of 6 weeks (4 weeks followed by a 2-week, open-label, placebo run-in period). Drug-naïve patients entered directly into the 2-week placebo run-in period. After the run-in period, patients were randomized to receive either linagliptin 5 mg or placebo, both in addition to pioglitazone 30 mg daily. Patients who failed to meet specific glycemic goals during the studies were treated with metformin rescue. Glycemic endpoints measured were A1C and FPG.
In initial combination with pioglitazone 30 mg, linagliptin 5 mg provided statistically significant improvements in A1C and FPG compared to placebo with pioglitazone (Table 9). Rescue therapy was used in 7.9% of patients treated with linagliptin 5 mg/pioglitazone 30 mg and 14.1% of patients treated with placebo/pioglitazone 30 mg. Patient weight increased in both groups during the study with an adjusted mean change from baseline of 2.3 kg and 1.2 kg in the linagliptin 5 mg/pioglitazone 30 mg and placebo/pioglitazone 30 mg groups, respectively (p = 0.0141).
| •Full analysis population using last observation on study ••HbA1c: ANCOVA model included treatment and number of prior OADs as class-effects, as well as baseline HbA1c as continuous covariates. FPG: ANCOVA model included treatment and number of prior OADs as class-effects, as well as baseline HbA1c and baseline FPG as continuous covariates. | ||
| Linagliptin 5 mg + Pioglitazone | Placebo + Pioglitazone | |
| A1C (%) | ||
| Number of patients | n = 252 | n = 128 |
| Baseline (mean) | 8.6 | 8.6 |
| Change from baseline (adjusted mean••) | -1.1 | -0.6 |
| Difference from placebo + pioglitazone (adjusted mean) (95% CI) | -0.5 (-0.7, -0.3) | -- |
| Patients [n (%)] achieving A1C <7% | 108 (42.9) | 39 (30.5) |
| FPG (mg/dL) | ||
| Number of patients | n = 243 | n = 122 |
| Baseline (mean) | 188 | 186 |
| Change from baseline (adjusted mean••) | -33 | -18 |
| Difference from placebo + pioglitazone (adjusted mean) (95% CI) | -14 (-21, -7) | -- |
Add-On Combination with Sulfonylureas
A total of 245 patients with type 2 diabetes participated in an 18-week, randomized, double-blind, placebo-controlled study designed to assess the efficacy of linagliptin in combination with sulfonylurea (SU). Patients on sulfonylurea monotherapy (n = 142) were randomized after completing a 2-week, single-blind, placebo run-in period. Patients on a sulfonylurea plus one additional oral antihyperglycemic agent (n = 103) were randomized after a wash-out period of 4 weeks and a 2-week, single-blind, placebo run-in period. Patients were randomized to the addition of linagliptin 5 mg or to placebo, each administered once daily. Patients who failed to meet specific glycemic goals during the studies were treated with metformin rescue. Glycemic endpoints measured included A1C and FPG.
In combination with a sulfonylurea, linagliptin provided statistically significant improvements in A1C compared with placebo following 18 weeks’ treatment; the improvements in FPG observed with linagliptin were not statistically significant compared with placebo (Table 10). Rescue therapy was used in 7.6% of patients treated with linagliptin 5 mg and 15.9% of patients treated with placebo. There was no significant difference between linagliptin and placebo in body weight.
| SU = sulfonylurea •Full analysis population using last observation on study ••Linagliptin 5 mg + SU, n=156; Placebo + SU, n=82 •••HbA1c: ANCOVA model included treatment and number of prior OADs as class-effects, as well as baseline HbA1c as continuous covariates. FPG: ANCOVA model included treatment and number of prior OADs as class-effects, as well as baseline HbA1c and baseline FPG as continuous covariates | ||
| Linagliptin 5 mg + SU | Placebo + SU | |
| A1C (%) | ||
| Number of patients | n = 158 | n = 82 |
| Baseline (mean) | 8.6 | 8.6 |
| Change from baseline (adjusted mean•••) | -0.5 | -0.1 |
| Difference from placebo + SU (adjusted mean) (95% CI) | -0.5 (-0.7, -0.2) | -- |
| Patients [n (%)] achieving A1C <7%•• | 23 (14.7) | 3 (3.7) |
| FPG (mg/dL) | ||
| Number of patients | n = 155 | n = 78 |
| Baseline (mean) | 180 | 171 |
| Change from baseline (adjusted mean•••) | -8 | -2 |
| Difference from placebo + SU (adjusted mean) (95% CI) | -6 (-17, 4) | -- |
Add-On Combination Therapy with Metformin and a Sulfonylurea
A total of 1058 patients with type 2 diabetes participated in a 24-week, randomized, double-blind, placebo-controlled study designed to assess the efficacy of linagliptin in combination with a sulfonylurea and metformin. The most common sulfonylureas used by patients in the study were: glimepiride (31%), glibenclamide (26%), and gliclazide (26%, not available in the United States). Patients on a sulfonylurea and metformin were randomized to receive linagliptin 5 mg or placebo, each administered once daily. Patients who failed to meet specific glycemic goals during the study were treated with pioglitazone rescue. Glycemic endpoints measured included A1C and FPG.
In combination with a sulfonylurea and metformin, linagliptin provided statistically significant improvements in A1C and FPG compared with placebo (Table 11). In the entire study population (patients on linagliptin in combination with sulfonylurea and metformin), a mean reduction from baseline relative to placebo in A1C of -0.6% and in FPG of -13 mg/dL was seen. Rescue therapy was used in 5.4% of patients treated with linagliptin 5 mg and in 13% of patients treated with placebo. Change from baseline in body weight did not differ significantly between the groups.
| SU = sulfonylurea •Full analysis population using last observation on study ••Linagliptin 5 mg + Metformin + SU, n=742; Placebo + Metformin + SU, n=247 •••HbA1c: ANCOVA model included treatment as class-effects and baseline HbA1c as continuous covariates. FPG: ANCOVA model included treatment as class-effects, as well as baseline HbA1c and baseline FPG as continuous covariates. | ||
| Linagliptin 5 mg + Metformin + SU | Placebo + Metformin + SU | |
| A1C (%) | ||
| Number of patients | n = 778 | n = 262 |
| Baseline (mean) | 8.2 | 8.1 |
| Change from baseline (adjusted mean•••) | -0.7 | -0.1 |
| Difference from placebo (adjusted mean) (95% CI) | -0.6 (-0.7, -0.5) | -- |
| Patients [n (%)] achieving A1C <7%•• | 217 (29.2) | 20 (8.1) |
| FPG (mg/dL) | ||
| Number of patients | n = 739 | n = 248 |
| Baseline (mean) | 159 | 163 |
| Change from baseline (adjusted mean•••) | -5 | 8 |
| Difference from placebo (adjusted mean) (95% CI) | -13 (-18, -7) | -- |
Add-On Combination Therapy with Insulin A total of 1261 patients with type 2 diabetes inadequately controlled on basal insulin alone or basal insulin in combination with oral drugs participated in a randomized, double-blind placebo-controlled trial designed to evaluate the efficacy of linagliptin as add-on therapy to basal insulin over 24 weeks. Randomization was stratified by baseline HbA1c (<8.5% vs ≥8.5%), renal function impairment status (based on baseline eGFR), and concomitant use of oral antidiabetic drugs (none, metformin only, pioglitazone only, metformin + pioglitazone). Patients with a baseline A1C of > 7% and < 10% were included in the study including 709 patients with renal impairment (eGFR <90 mL/min), most of whom (n=575) were categorized as mild renal impairment (eGFR 60 to <90 mL/min). Patients entered a 2 week placebo run-in period on basal insulin (e.g., insulin glargine, insulin detemir, or NPH insulin) with or without metformin and/or pioglitazone background therapy. Following the run-in period, patients with inadequate glycemic control were randomized to the addition of either 5 mg of linagliptin or placebo, administered once daily. Patients were maintained on a stable dose of insulin prior to enrollment, during the run-in period, and during the first 24 weeks of treatment. Patients who failed to meet specific glycemic goals during the double-blind treatment period were rescued by increasing background insulin dose.
Linagliptin used in combination with insulin (with or without metformin and/or pioglitazone), provided statistically significant improvements in A1C and FPG compared to placebo (Table 12) after 24 weeks of treatment. The mean total daily insulin dose at baseline was 42 units for patients treated with linagliptin and 40 units for patients treated with placebo. Background baseline diabetes therapy included use of: insulin alone (16.1%), insulin combined with metformin only (75.5%), insulin combined with metformin and pioglitazone (7.4%), and insulin combined with pioglitazone only (1%). The mean change from baseline to Week 24 in the daily dose of insulin was +1.3 IU in the placebo group and +0.6 IU in the linagliptin group. The mean change in body weight from baseline to Week 24 was similar in the two treatment groups.
| • Full analysis population using last observation carried forward (LOCF) method on study •• Linagliptin + Insulin, n=595; Placebo + Insulin, n=593 •••HbA1c: ANCOVA model included treatment, categorical renal function impairment status and concomitant OADs as class-effects, as well as baseline HbA1c as continuous covariates. FPG: ANCOVA model included treatment, categorical renal function impairment status and concomitant OADs as class-effects, as well as baseline HbA1c and baseline FPG as continuous covariates. | ||
| Linagliptin 5 mg + Insulin | Placebo + Insulin | |
| A1C (%) | ||
| Number of patients | n = 618 | n = 617 |
| Baseline (mean) | 8.3 | 8.3 |
| Change from baseline (adjusted mean•••) | -0.6 | 0.1 |
| Difference from placebo (adjusted mean) (95% CI) | -0.7 (-0.7, -0.6) | -- |
| Patients [n (%)] achieving A1C <7%•• | 116 (19.5) | 48 (8.1) |
| FPG (mg/dL) | ||
| Number of patients | n = 613 | n = 608 |
| Baseline (mean) | 147 | 151 |
| Change from baseline (adjusted mean•••) | -8 | 3 |
| Difference from placebo (adjusted mean) (95% CI) | -11 (-16, -6) | -- |
The difference between treatment with linagliptin and placebo in terms of adjusted mean change from baseline in HbA1c after 24 weeks was comparable for patients with no renal impairment (eGFR ≥90 mL/min, n=539), with mild renal impairment (eGFR 60 to <90 mL/min, n= 565), or with moderate renal impairment (eGFR 30 to <60 mL/min, n=124).
Renal Impairment
A total of 133 patients with type 2 diabetes participated in a 52 week, double-blind, randomized, placebo-controlled trial designed to evaluate the efficacy and safety of linagliptin in patients with both type 2 diabetes and severe chronic renal impairment. Participants with an estimated (based on the four variables modified diet in renal disease [MDRD] equation) GFR value of <30 mL/min were eligible to participate in the study. Randomization was stratified by baseline HbA1c (≤8% and >8%) and background antidiabetic therapy (insulin or any combination with insulin, SU or glinides as monotherapy and pioglitazone or any other antidiabetics excluding any other DPP-4 inhibitors). For the initial 12 weeks of the study, background antidiabetic therapy was kept stable and included insulin, sulfonylurea, glinides, and pioglitazone. For the remainder of the trial, dose adjustments in antidiabetic background therapy were allowed. At baseline in this trial, 62.5% of patients were receiving insulin alone as background diabetes therapy, and 12.5% were receiving sulfonylurea alone.
After 12 weeks of treatment, linagliptin 5 mg provided statistically significant improvement in A1C compared to placebo, with an adjusted mean change of -0.6% compared to placebo (95% confidence interval -0.9, -0.3) based on the analysis using last observation carried forward (LOCF). With adjustments in antidiabetic background therapy after the initial 12 weeks, efficacy was maintained for 52 weeks, with an adjusted mean change from baseline in A1C of -0.7% compared to placebo (95% confidence interval -1.0, -0.4) based on analysis using LOCF.
16 HOW SUPPLIED/STORAGE AND HANDLING
Linagliptin tablets are available as white or off-white, round, biconvex, film-coated tablets containing 5 mg of linagliptin. Linagliptin tablets are debossed with “S75” on one side and blank on the other side.
They are supplied as follows: Bottles of 30 (NDC 48792-7865-1) If repackaging is required, dispense in a tight container as defined in USP.
Storage
Store at 25°C (77°F); excursions permitted to 15°-30°C (59°-86°F) [see USP Controlled Room Temperature]. Store in a safe place out of reach of children.
12.1 Mechanism of Action
Linagliptin is an inhibitor of DPP-4, an enzyme that degrades the incretin hormones glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). Thus, linagliptin increases the concentrations of active incretin hormones, stimulating the release of insulin in a glucose-dependent manner and decreasing the levels of glucagon in the circulation. Both incretin hormones are involved in the physiological regulation of glucose homeostasis. Incretin hormones are secreted at a low basal level throughout the day and levels rise immediately after meal intake. GLP-1 and GIP increase insulin biosynthesis and secretion from pancreatic beta cells in the presence of normal and elevated blood glucose levels. Furthermore, GLP-1 also reduces glucagon secretion from pancreatic alpha-cells, resulting in a reduction in hepatic glucose output.