Get your patient on Kit For The Preparation Of Technetium Tc 99m Medronate - Technetium Tc 99m Medronate injection, Powder, Lyophilized, For Solution (Technetium Tc 99m Medronate)
Kit For The Preparation Of Technetium Tc 99m Medronate - Technetium Tc 99m Medronate injection, Powder, Lyophilized, For Solution prescribing information
INDICATIONS AND USAGE
Technetium Tc 99m medronate may be used as a bone imaging agent to delineate areas of altered osteogenesis.
DOSAGE AND ADMINISTRATION
After preparation with oxidant-free sodium pertechnetate Tc 99m injection the suggested dose range of technetium Tc 99m medronate injection in the average patient (70 kg) is 370 MBq to 740 MBq (10 mCi to 20 mCi) given intravenously. Imaging post injection is optimal at 1 to 4 hours.
The patient dose should be measured by a suitable radioactivity calibration system immediately prior to administration.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. Shielding should be utilized when preparing the technetium Tc 99m medronate injection.
Radiation Dosimetry
The effective half-life was assumed to be the physical half-life for all calculated values. The estimated radiation absorbed doses to an average patient (70 kg) from an intravenous injection of a maximum dose of 740 MBq (20 mCi) of technetium Tc 99m medronate are shown in Table 4 .
Absorbed Radiation Dose | |||
Organ | mGy/740 MBq | rads/20 mCi | |
Total Body | 1.3 | 0.13 | |
Bone Total | 7.0 | 0.70 | |
Red Marrow | 5.6 | 0.56 | |
Kidneys | 8.0 | 0.80 | |
Liver | 0.6 | 0.06 | |
Bladder Wall | 2-hr. void | 26.0 | 2.60 |
4.8-hr. void | 62.0 | 6.20 | |
Ovaries | 2-hr. void | 2.4 | 0.24 |
4.8-hr. void | 3.4 | 0.34 | |
Testes | 2-hr. void | 1.6 | 0.16 |
4.8-hr. void | 2.2 | 0.22 | |
Method of Calculation: “S” Absorbed Dose per Unit Cumulated Activity for Selected Radionuclides and Organs, MIRD Pamphlet No. 11, 1975
CONTRAINDICATIONS
None known.
ADVERSE REACTIONS
Several adverse reactions due to technetium Tc 99m medronate have been reported. These were usually hypersensitivity reactions characterized by itching, various skin rashes, hypotension, chills, nausea and vomiting. There have also been rare cases of dizziness and asthenia associated with the use of technetium Tc 99m medronate.
Drug Interactions
The biodistribution of technetium Tc 99m medronate may be altered in the presence of high levels of certain cations (iron, calcium, and aluminum). This may result in reduced uptake of radionuclide in the skeleton and increased extraosseal uptake, which may potentially degrade imaging quality. In patients with high levels of these cations caused by concomitant medications, particularly patients receiving iron infusions, consider performing an imaging study with technetium Tc 99m medronate injection once the cation levels have normalized (e.g., after 3 to 5 half-lives of the cation). (See WARNINGS .)
DESCRIPTION
Each reaction vial contains a sterile, nonpyrogenic, nonradioactive lyophilized mixture of 20 mg medronic acid, 1 mg ascorbic acid, 0.13 mg (minimum) stannous fluoride, SnF 2 and 0.38 mg total tin, maximum (as stannous fluoride, SnF 2 ).
The pH is adjusted with sodium hydroxide or hydrochloric acid to 6.5 (6.3 to 6.7) prior to lyophilization. The vial does not contain a preservative. The contents of the vial are lyophilized and sealed under nitrogen at the time of manufacture.
The pH of the reconstituted product is 5.4 to 6.8.
The structure of medronic acid is given below:
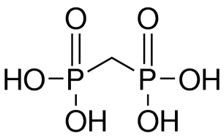
The precise structure of technetium Tc 99m medronate is unknown at this time.
When sterile, pyrogen-free sodium pertechnetate Tc 99m injection is added to the vial, the diagnostic agent technetium Tc 99m medronate is formed for administration by intravenous injection.
CLINICAL PHARMACOLOGY
During the initial 24 hours following intravenous injection of technetium Tc 99m medronate, about 50 percent of the dose is retained in the skeleton, and about 50 percent is excreted in the urine. A minimum amount of uptake has been observed in soft-tissue organs, most notably the kidneys.
Clearance of radioactivity from the blood is quite rapid, with about 10 percent of the injected dose remaining at one hour, and less than 5 and 2 percent at two and four hours, respectively. The rapid blood clearance (T ½: 38 to 75 minutes) provides bone to non-osseous tissue ratios favoring early imaging.
Following intravenous administration of technetium Tc 99m medronate, skeletal uptake occurs as a function of blood flow to bone and bone efficiency in extracting the complex. Bone mineral crystals are generally considered to be hydroxyapatite, and the complex appears to have an affinity for the hydroxyapatite crystals in bone.
Deposition of radioactivity in bone is rapid and appears to be related to osteogenic activity as well as the aforementioned skeletal blood perfusion. Skeletal uptake is bilaterally uniform, with larger concentrations in the axial structure and in the long bones. Increased accumulation of radioactivity may be seen, generally, in any bone disease state in which there is increased osteogenesis or a localized increase in osseous blood perfusion; consequently, bone imaging agents generally are not effective in detecting chronic bone diseases.
HOW SUPPLIED
Kit for the Preparation of Technetium Tc 99m Medronate is supplied as kits of 10 reaction vials (NDC 65857-505-10).
Each reaction vial contains a sterile, nonpyrogenic, nonradioactive lyophilized mixture of 20 mg medronic acid, 1 mg ascorbic acid, 0.13 mg (minimum) stannous fluoride, SnF 2 and 0.38 mg total tin, maximum (as stannous fluoride, SnF 2 ).
The pH is adjusted with sodium hydroxide or hydrochloric acid prior to lyophilization. The vial does not contain a preservative. The contents of the vial are lyophilized and sealed under nitrogen at the time of manufacture. The pH of the reconstituted product is 5.4 to 6.8.
Kit Contents 10 sterile reaction vials 20 pressure-sensitive labels for technetium Tc 99m medronate 1 package insert
Storage
Store the product as supplied at 20° to 25°C (68° to 77°F). After reconstitution store refrigerated at 2° to 8°C (36° to 46°F).
Do not use and discard radiolabeled technetium Tc 99m medronate 6 hours after reconstitution.