Ketorolac Tromethamine prescribing information
WARNING
Ketorolac tromethamine, a nonsteroidal anti-inflammatory drug (NSAID), is indicated for the short-term (up to 5 days in adults) management of moderately severe acute pain that requires analgesia at the opioid level. Oral ketorolac tromethamine is indicated only as continuation treatment following intravenous or intramuscular dosing of ketorolac tromethamine, if necessary. The total combined duration of use of oral ketorolac tromethamine and ketorolac tromethamine injection should not exceed 5 days.
Ketorolac tromethamine is not indicated for use in pediatric patients and it is NOT indicated for minor or chronic painful conditions. Increasing the dose of ketorolac tromethamine beyond the label recommendations will not provide better efficacy but will increase the risk of developing serious adverse events.
GASTROINTESTINAL RISK
• Ketorolac tromethamine can cause peptic ulcers, gastrointestinal bleeding and/or perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Therefore, ketorolac tromethamine is CONTRAINDICATED in patients with active peptic ulcer disease, in patients with recent gastrointestinal bleeding or perforation, and in patients with a history of peptic ulcer disease or gastrointestinal bleeding. Elderly patients are at greater risk for serious gastrointestinal events (see WARNINGS ).
CARDIOVASCULAR THROMBOTIC EVENTS
• Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use (see WARNINGS and PRECAUTIONS ).
•Ketorolac tromethamine is CONTRAINDICATED in the setting of coronary artery bypass graft (CABG) surgery (see CONTRAINDICATIONS and WARNINGS ).
RENAL RISK
• Ketorolac tromethamine is CONTRAINDICATED in patients with advanced renal impairment and in patients at risk for renal failure due to volume depletion (see WARNINGS ).
RISK OF BLEEDING
• Ketorolac tromethamine inhibits platelet function and is, therefore, CONTRAINDICATED in patients with suspected or confirmed cerebrovascular bleeding, patients with hemorrhagic diathesis, incomplete hemostasis and those at high risk of bleeding (see WARNINGS and PRECAUTIONS ).
Ketorolac tromethamine is CONTRAINDICATED as prophylactic analgesic before any major surgery.
HYPERSENSITIVITY
• Hypersensitivity reactions, ranging from bronchospasm to anaphylactic shock, have occurred and appropriate counteractive measures must be available when administering the first dose of ketorolac tromethamine injection (see CONTRAINDICATIONS and WARNINGS ). Ketorolac tromethamine is CONTRAINDICATED in patients with previously demonstrated hypersensitivity to ketorolac tromethamine or allergic manifestations to aspirin or other nonsteroidal anti-inflammatory drugs (NSAIDs).
INTRATHECAL OR EPIDURAL ADMINISTRATION
• Ketorolac tromethamine is CONTRAINDICATED for intrathecal or epidural administration due to its alcohol content.
RISK DURING LABOR AND DELIVERY
• The use of ketorolac tromethamine in labor and delivery is CONTRAINDICATED because it may adversely affect fetal circulation and inhibit uterine contractions.
CONCOMITANT USE WITH NSAIDs
• Ketorolac tromethamine is CONTRAINDICATED in patients currently receiving aspirin or NSAIDs because of the cumulative risk of inducing serious NSAID-related side effects.
SPECIAL POPULATIONS
• Dosage should be adjusted for patients 65 years or older, for patients under 50 kg (110 lbs.) of body weight (see DOSAGE AND ADMINISTRATION ) and for patients with moderately elevated serum creatinine (see WARNINGS ). Doses of ketorolac tromethamine injection are not to exceed 60 mg (total dose per day) in these patients.
DOSAGE AND ADMINISTRATION
Ketorolac Tromethamine Tablets
• Ketorolac tromethamine tablets are indicated only as continuation therapy to ketorolac tromethamine injection, and the combined duration of use of ketorolac tromethamine injection and ketorolac tromethamine tablets is not to exceed 5 (five) days, because of the increased risk of serious adverse events.
The recommended total daily dose of ketorolac tromethamine tablets (maximum 40 mg) is significantly lower than for ketorolac tromethamine injection (maximum 120 mg) (see DOSAGE AND ADMINISTRATION ).
INDICATIONS AND USAGE
Carefully consider the potential benefits and risks of ketorolac tromethamine and other treatment options before deciding to use ketorolac. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ).
Acute Pain in Adult Patients
Ketorolac tromethamine is indicated for the short-term (≤5 days) management of moderately severe acute pain that requires analgesia at the opioid level, usually in a postoperative setting. Therapy should always be initiated with intravenous or intramuscular dosing of ketorolac tromethamine, and oral ketorolac tromethamine is to be used only as continuation treatment, if necessary.
The total combined duration of use of ketorolac tromethamine injection and oral ketorolac tromethamine is not to exceed 5 days of use because of the potential of increasing the frequency and severity of adverse reactions associated with the recommended doses (see WARNINGS , PRECAUTIONS , DOSAGE AND ADMINISTRATION , and ADVERSE REACTIONS ). Patients should be switched to alternative analgesics as soon as possible, but ketorolac tromethamine therapy is not to exceed 5 days.
DOSAGE AND ADMINISTRATION
Carefully consider the potential benefits and risks of ketorolac tromethamine and other treatment options before deciding to use ketorolac tromethamine. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals. In adults, the combined duration of use of intravenous or intramuscular dosing of ketorolac tromethamine and oral ketorolac tromethamine is not to exceed 5 days. In adults, the use of oral ketorolac tromethamine is only indicated as continuation therapy to intravenous or intramuscular dosing of ketorolac tromethamine. See package insert for ketorolac tromethamine tablets for transition from intravenous or intramuscular dosing of ketorolac tromethamine (single- or multiple-dose) to multiple-dose oral ketorolac tromethamine.
Note: Oral formulation should not be given as an initial dose.
Use minimum effective dose for the individual patient.
Total duration of treatment in adult patients: the combined duration of use of intravenous or intramuscular dosing of ketorolac tromethamine and oral ketorolac tromethamine is not to exceed 5 days.
KETOROLAC TROMETHAMINE INJECTION
Ketorolac tromethamine injection may be used as a single or multiple doses on a regular or “as needed” schedule for the management of moderately severe, acute pain that requires analgesia at the opioid level, usually in a postoperative setting. Hypovolemia should be corrected prior to the administration of ketorolac tromethamine (see WARNINGS - Renal Effects ). Patients should be switched to alternative analgesics as soon as possible, but ketorolac tromethamine therapy is not to exceed 5 days.
When administering ketorolac tromethamine injection, the intravenous bolus must be given over no less than 15 seconds. The intramuscular administration should be given slowly and deeply into the muscle. The analgesic effect begins in ~30 minutes with maximum effect in 1 to 2 hours after dosing intravenous or intramuscular. Duration of analgesic effect is usually 4 to 6 hours.
Single-Dose Treatment: The following regimen should be limited to single administration use only
Intramuscular Dosing
• Patients < 65 years of age: One dose of 60 mg.
•Patients ≥ 65 years of age, renally impaired and/or less than 50 kg (110 lbs) of body weight: One dose of 30 mg.
Intravenous Dosing
• Patients < 65 years of age: One dose of 30 mg.
•Patients ≥ 65 years of age, renally impaired and/or less than 50 kg (110 lbs) of body weight: One dose of 15 mg.
Multiple-Dose Treatment (Intravenous or Intramuscular)
• Patients < 65 years of age:The recommended dose is 30 mg ketorolac tromethamine injection every 6 hours. The maximum daily dose for these populations should not exceed 120 mg.
• For patients ≥65 years of age, renally impaired patients (see WARNINGS ), and patients less than 50 kg (110 lbs): The recommended dose is 15 mg ketorolac tromethamine injection every 6 hours. The maximum daily dose for these populations should not exceed 60 mg.
For breakthrough pain, do not increase the dose or the frequency of ketorolac tromethamine. Consideration should be given to supplementing these regimens with low doses of opioids “as needed” unless otherwise contraindicated.
Pharmaceutical Information for Ketorolac Tromethamine Injection
Ketorolac tromethamine injection should not be mixed in a small volume (e.g., in a syringe) with morphine sulfate, meperidine hydrochloride, promethazine hydrochloride or hydroxyzine hydrochloride; this will result in precipitation of ketorolac from solution.
NOTE : Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
CONTRAINDICATIONS
(see also BOXED WARNING )
Ketorolac Tromethamine is contraindicated in patients with previously demonstrated hypersensitivity to ketorolac tromethamine.
Ketorolac tromethamine is contraindicated in patients with active peptic ulcer disease, in patients with recent gastrointestinal bleeding or perforation and in patients with a history of peptic ulcer disease or gastrointestinal bleeding.
Ketorolac tromethamine should not be given to patients who have experienced asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients (see WARNINGS - Anaphylactoid Reactions , and PRECAUTIONS – Pre-existing Asthma ).
Ketorolac tromethamine is contraindicated as prophylactic analgesic before any major surgery.
Ketorolac tromethamine is contraindicated in the setting of coronary artery bypass graft (CABG) surgery (see WARNINGS ).
Ketorolac tromethamine is contraindicated in patients with advanced renal impairment or in patients at risk for renal failure due to volume depletion (see WARNINGS for correction of volume depletion).
Ketorolac tromethamine is contraindicated in labor and delivery because, through its prostaglandin synthesis inhibitory effect, it may adversely affect fetal circulation and inhibit uterine musculature, thus increasing the risk of uterine hemorrhage.
Ketorolac tromethamine inhibits platelet function and is, therefore, contraindicated in patients with suspected or confirmed cerebrovascular bleeding, hemorrhagic diathesis, incomplete hemostasis and those at high risk of bleeding (see WARNINGS and PRECAUTIONS ).
Ketorolac tromethamine is contraindicated in patients currently receiving aspirin or NSAIDs because of the cumulative risks of inducing serious NSAID-related adverse events.
The concomitant use of ketorolac tromethamine and probenecid is contraindicated.
The concomitant use of ketorolac tromethamine and pentoxifylline is contraindicated.
Ketorolac tromethamine injection is contraindicated for neuraxial (epidural or intrathecal) administration due to its alcohol content.
ADVERSE REACTIONS
Adverse reaction rates increase with higher doses of ketoralac tromethamine. Practitioners should be alert for the severe complications of treatment with ketorolac tromethamine, such as G.I. ulceration, bleeding and perforation, postoperative bleeding, acute renal failure, anaphylactic and anaphylactoid reactions and liver failure (see Boxed WARNING , WARNINGS , PRECAUTIONS , and DOSAGE AND ADMINISTRATION ). These NSAID-related complications can be serious in certain patients for whom ketorolac tromethamine is indicated, especially when the drug is used inappropriately.
In patients taking ketorolac tromethamine or other NSAIDs in clinical trials, the most frequently reported adverse experiences in approximately 1% to 10% of patients are:
| Gastrointestinal (GI) experiences including: | ||
| abdominal pain | constipation/diarrhea | dyspepsia |
| flatulence | GI fullness | GI ulcers(gastric/duodenal) |
| gross bleeding/perforation | heartburn | nausea• |
| stomatitis | vomiting | |
| Other experiences: | ||
| abnormal renal function | anemia | dizziness |
| drowsiness | edema | elevated liver enzymes |
| headaches• | hypertension | increased bleeding time |
| injection site pain | pruritus | purpura |
| rashes | tinnitus | sweating |
• Incidence greater than 10%
Additional adverse experiences reported occasionally (<1% in patients taking ketorolac tromethamine or other NSAIDs in clinical trials) include:
Body as a Whole: fever, infections, sepsis
Cardiovascular: congestive heart failure, palpitation, pallor, tachycardia, syncope
Dermatologic: alopecia, photosensitivity, urticaria
Gastrointestinal: anorexia, dry mouth, eructation, esophagitis, excessive thirst, gastritis, glossitis, hematemesis, hepatitis, increased appetite, jaundice, melena, rectal bleeding
Hemic and Lymphatic: ecchymosis, eosinophilia, epistaxis, leukopenia, thrombocytopenia
Metabolic and Nutritional: weight change
Nervous System: abnormal dreams, abnormal thinking, anxiety, asthenia, confusion, depression, euphoria, extrapyramidal symptoms, hallucinations, hyperkinesis, inability to concentrate, insomnia, nervousness, paresthesia, somnolence, stupor, tremors, vertigo, malaise
Reproductive, female: infertility
Respiratory: asthma, cough, dyspnea, pulmonary edema, rhinitis
Special Senses: abnormal taste, abnormal vision, blurred vision, hearing loss
Urogenital: cystitis, dysuria, hematuria, increased urinary frequency, interstitial nephritis, oliguria/polyuria, proteinuria, renal failure, urinary retention
Other rarely observed reactions (reported from postmarketing experience in patients taking ketorolac tromethamine or other NSAIDs) are:
Body as a Whole: angioedema, death, hypersensitivity reactions such as anaphylaxis, anaphylactoid reaction, laryngeal edema, tongue edema (see WARNINGS ), myalgia
Cardiovascular: arrhythmia, bradycardia, chest pain, flushing, hypotension, myocardial infarction, vasculitis
Dermatologic: exfoliative dermatitis, erythema multiforme, Lyell's syndrome, bullous reactions including Stevens-Johnson syndrome and toxic epidermal necrolysis, and fixed drug eruption (FDE)
Gastrointestinal: acute pancreatitis, liver failure, ulcerative stomatitis, exacerbation of inflammatory bowel disease (ulcerative colitis, Crohns disease)
Hemic and Lymphatic: agranulocytosis, aplastic anemia, hemolytic anemia, lymphadenopathy, pancytopenia, post operative wound hemorrhage (rarely requiring blood transfusion - see Boxed WARNING , WARNINGS , and PRECAUTIONS )
Metabolic and Nutritional: hyperglycemia, hyperkalemia, hyponatremia
Nervous System: aseptic meningitis, convulsions, coma, psychosis
Respiratory: bronchospasm, respiratory depression, pneumonia
Special Senses: conjunctivitis
Urogenital: flank pain with or without hematuria and/or azotemia, hemolytic uremic syndrome
Postmarketing Surveillance Study
A large postmarketing observational, nonrandomized study, involving approximately 10,000 patients receiving ketorolac tromethamine, demonstrated that the risk of clinically serious gastrointestinal (GI) bleeding was dose-dependent (see Tables 3A and 3B ). This was particularly true in elderly patients who received an average daily dose greater than 60 mg/day of ketorolac tromethamine (see Table 3A ).
Table 3. Incidence of Clinically Serious G.I. Bleeding as Related to Age, Total Daily Dose, and History of G.I. Perforation, Ulcer, Bleeding (PUB) after up to 5 Days of Treatment with Ketorolac Tromethamine Injection
A. Adult Patients without History of PUB | |||||||
Age of Patients | Total Daily Dose of Ketorolac Tromethamine Injection | ||||||
≤ 60 mg | >60 to 90 mg | >90 to 120 mg | >120 mg | ||||
<65 years of age | 0.4% | 0.4% | 0.9% | 4.6% | |||
≥65 years of age | 1.2% | 2.8% | 2.2% | 7.7% | |||
B. Adult Patients with History of PUB | |||||||
Age of Patients | Total Daily Dose of Ketorolac Tromethamine Injection | ||||||
≤ 60 mg | >60 to 90 mg | >90 to 120 mg | >120 mg | ||||
<65 years of age | 2.1% | 4.6% | 7.8% | 15.4% | |||
≥65 years of age | 4.7% | 3.7% | 2.8% | 25.0 % | |||
DESCRIPTION
Ketorolac Tromethamine Injection, USP is a member of the pyrrolo-pyrrole group of nonsteroidal anti-inflammatory drugs (NSAIDs). The chemical name for ketorolac tromethamine is (±)-5-benzoyl-2,3-dihydro-1H-pyrrolizine-1-carboxylic acid, compound with 2-amino-2-(hydroxymethyl)-1,3-propanediol (1:1), and the structural formula is presented in Figure 1.
FIGURE 1
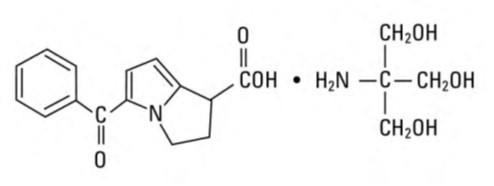
C 15 H 13 NO 3 • C 4 H 11 NO 3
Ketorolac tromethamine is a racemic mixture of [-]S and [+]R ketorolac tromethamine. Ketorolac tromethamine may exist in three crystal forms. All forms are equally soluble in water. Ketorolac tromethamine has a pKa of 3.5 and an n-octanol/water partition coefficient of 0.26. The molecular weight of ketorolac tromethamine is 376.40.
Ketorolac Tromethamine Injection, USP is available for intravenous (IV) or intramuscular (IM) administration as: 15 mg in 1 mL (1.5%) and 30 mg in 1 mL (3%) in sterile solution; 60 mg in 2 mL (3%) of ketorolac tromethamine in sterile solution is available for intramuscular administration only. The solutions contain 10% (w/v) alcohol, USP, and 6.68 mg, 4.35 mg, and 8.70 mg, respectively, of sodium chloride in sterile water. The pH range is 6.9 to 7.9 and is adjusted with sodium hydroxide and/or hydrochloric acid. The sterile solutions are clear and slightly yellow in color.
CLINICAL PHARMACOLOGY
Pharmacodynamics
Ketorolac tromethamine is a nonsteroidal anti-inflammatory drug (NSAID) that exhibits analgesic activity in animal models. The mechanism of action of ketorolac, like that of other NSAIDs, is not completely understood but may be related to prostaglandin synthetase inhibition. The biological activity of ketorolac tromethamine is associated with the S-form.
Ketorolac tromethamine possesses no sedative or anxiolytic properties.
The peak analgesic effect of ketorolac tromethamine occurs within 2 to 3 hours and is not statistically significantly different over the recommended dosage range of ketorolac tromethamine.The greatest difference between large and small doses of ketorolac tromethamine is in the duration of analgesia.
Pharmacokinetics
Ketorolac tromethamine is a racemic mixture of [-]S- and [+]R-enantiomeric forms, with the S-form having analgesic activity.
Comparison of Intravenous, Intramuscular and Oral Pharmacokinetics
The pharmacokinetics of ketorolac tromethamine, following intravenous, intramuscular, and oral doses of ketorolac tromethamine are compared in Table 1. In adults, the extent of bioavailability following administration of the oral and intramuscular forms of ketorolac tromethamine was equal to that following an intravenous bolus.
Table 1. Table of Approximate Average Pharmacokinetic Parameters (Mean±SD) Following Oral, Intramuscular and Intravenous Doses of Ketorolac Tromethamine
Pharmacokinetic Parameters (units) | Oral † | Intramuscular• | Intravenous Bolus ‡ | |||
| 10 mg | 15 mg | 30 mg | 60 mg | 15 mg | 30 mg | |
Bioavailability (extent) | 100% | |||||
T max 1 (min) | 44±34 | 33±21 •• | 44±29 | 33±21 •• | 1.1±0.7 •• | 2.9±1.8 |
C max 2 (mcg/mL) [Single-dose] | 0.87± 0.22 | 1.14± 0.32•• | 2.42±0.68 | 4.55±1.27 •• | 2.47± 0.51 •• | 4.65± 0.96 |
C max (mcg/mL) [steady state qid] | 1.05± 0.26 •• | 1.56± 0.44 •• | 3.11±0.87 •• | N/A ††| | 3.09± 1.17 •• | 6.85± 2.61 |
C min 3 (mcg/mL) [steady state qid] | 0.29± 0.07 •• | 0.47± 0.13 •• | 0.93±0.26 •• | N/A | 0.61± 0.21 •• | 1.04± 0.35 |
C avg 4 (mcg/mL) [steady state qid] | 0.59± 0.2 •• | 0.94± 0.29 •• | 1.88±0.59 •• | N/A | 1.09± 0.3 •• | 2.17± 0.59 |
V ß 5 (L/kg) | 0.175±0.039 | 0.210±0.044 | ||||
% Dose metabolized = <50 % Dose excreted in feces = 6
% Dose excreted in urine = 91 % Plasma protein binding = 99
† Derived from oral pharmacokinetic studies in 77 normal fasted volunteers
• Derived from intramuscular pharmacokinetic studies in 54 normal volunteers
‡ Derived from intravenous pharmacokinetic studies in 24 normal volunteers
†† Not applicable because 60 mg is only recommended as a single dose
•• Mean value was simulated from observed plasma concentration data and standard deviation was simulated from percent coefficient of variation for observed C max and T max data
1 Time-to-peak plasma concentration
2 Peak plasma concentration
3 Trough plasma concentration
4 Average plasma concentration
5 Volume of distribution
Linear Kinetics
In adults, following administration of single oral, intramuscular or intravenous doses of ketorolac tromethamine in the recommended dosage ranges, the clearance of the racemate does not change. This implies that the pharmacokinetics of ketorolac tromethamine in adults, following single or multiple intramuscular, intravenous, or recommended oral doses of ketorolac tromethamine, are linear. At the higher recommended doses, there is a proportional increase in the concentrations of free and bound racemate.
Distribution
The mean apparent volume (V ß ) of ketorolac tromethamine following complete distribution was approximately 13 liters. This parameter was determined from single-dose data. The ketorolac tromethamine racemate has been shown to be highly protein bound (99%). Nevertheless, plasma concentrations as high as 10 mcg/mL will only occupy approximately 5% of the albumin binding sites. Thus, the unbound fraction for each enantiomer will be constant over the therapeutic range. A decrease in serum albumin, however, will result in increased free drug concentrations.
Ketorolac tromethamine is excreted in human milk (see PRECAUTIONS - Nursing Mothers ).
Metabolism
Ketorolac tromethamine is largely metabolized in the liver. The metabolic products are hydroxylated and conjugated forms of the parent drug. The products of metabolism, and some unchanged drug, are excreted in the urine.
Excretion
The principal route of elimination of ketorolac and its metabolites is renal. About 92% of a given dose is found in the urine, approximately 40% as metabolites and 60% as unchanged ketorolac. Approximately 6% of a dose is excreted in the feces. A single-dose study with 10 mg ketorolac tromethamine (n = 9) demonstrated that the S-enantiomer is cleared approximately two times faster than the R-enantiomer and that the clearance was independent of the route of administration. This means that the ratio of S/R plasma concentrations decreases with time after each dose. There is little or no inversion of the R- to S-form in humans. The clearance of the racemate in normal subjects, elderly individuals and in hepatically and renally impaired patients is outlined in Table 2(see CLINICAL PHARMACOLOGY - Kinetics in Special Populations ).
The half-life of the ketorolac tromethamine S-enantiomer was approximately 2.5 hours (SD ± 0.4) compared with 5 hours (SD ± 1.7) for the R-enantiomer. In other studies, the half-life for the racemate has been reported to lie within the range of 5 to 6 hours.
Accumulation
Ketorolac tromethamine administered as an intravenous bolus, every 6 hours, for 5 days, to healthy subjects (n = 13), showed no significant difference in C max on Day 1 and Day 5. Trough levels averaged 0.29 mcg/mL (SD ± 0.13) on Day 1 and 0.55 mcg/mL (SD ± 0.23) on Day 6. Steady state was approached after the fourth dose.
Accumulation of ketorolac tromethamine has not been studied in special populations (geriatric, pediatric, renal failure patients, or hepatic disease patients).
Kinetics in Special Populations
Geriatric Patients
Based on single-dose data only, the half-life of the ketorolac tromethamine racemate increased from 5 to 7 hours in the elderly (65 to 78 years) compared with young healthy volunteers (24 to 35 years) (see Table 2 ). There was little difference in the C max for the two groups (elderly, 2.52 mcg/mL ± 0.77; young, 2.99 mcg/mL ± 1.03) (see PRECAUTIONS - Geriatric Use ).
Pediatric Patients
Limited information is available regarding the pharmacokinetics of dosing of ketorolac tromethamine in the pediatric population. Following a single intravenous bolus dose of 0.5 mg/kg in 10 children 4 to 8 years old, the half-life was 5.8 ± 1.6 hours, the average clearance was 0.042 ± 0.01 L/hr/kg, the volume of distribution during the terminal phase (V β ) was 0.34 ± 0.12 L/kg and the volume of distribution at steady state (V ss ) was 0.26 ± 0.08 L/kg. The volume of distribution and clearance of ketorolac in pediatric patients was higher than those observed in adult subjects (see Table 1 ). There are no pharmacokinetic data available for administration of ketorolac tromethamine by the intramuscular route in pediatric patients.
Renal Insufficiency
Based on single-dose data only, the mean half-life of ketorolac tromethamine in renally impaired patients is between 6 and 19 hours and is dependent on the extent of the impairment. There is poor correlation between creatinine clearance and total ketorolac tromethamine clearance in the elderly and populations with renal impairment (r = 0.5).
In patients with renal disease, the AUC ∞ of each enantiomer increased by approximately 100% compared with healthy volunteers. The volume of distribution doubles for the S-enantiomer and increases by 1/5th for the R-enantiomer. The increase in volume of distribution of ketorolac tromethamine implies an increase in unbound fraction.
The AUC ∞ -ratio of the ketorolac tromethamine enantiomers in healthy subjects and patients remained similar, indicating there was no selective excretion of either enantiomer in patients compared to healthy subjects (see WARNINGS - Renal Effects ).
Hepatic Insufficiency
There was no significant difference in estimates of half-life, AUC ∞ and C max , in 7 patients with liver disease compared to healthy volunteers (see PRECAUTIONS - Hepatic Effects and Table 2 ).
Race
Pharmacokinetic differences due to race have not been identified.
Table 2. The Influence of Age, Liver and Kidney Function, on the Clearance and Terminal Half-life of Ketorolac Tromethamine (Intramuscular 1 and Oral 2 ) in Adult Populations
Total Clearance [in L/h/kg] 3 | Terminal Half-life [in hours] | |||
Type of Subjects | Intramuscular Mean (range) | Oral Mean (range) | Intramuscular Mean (range) | Oral Mean (range) |
Normal Subjects Intramuscular (n = 54) mean age = 32, range = 18-60 Oral (n=77) mean age=32, range = 20-60 | 0.023 (0.010-0.046) | 0.025 (0.013-0.050) | 5.3 (3.5-9.2) | 5.3 (2.4-9) |
Healthy Elderly Subjects Intramuscular (n = 13), Oral (n = 12) mean age = 72, range = 65-78 | 0.019 (0.013-0.034) | 0.024 (0.018-0.034) | 7 (4.7-8.6) | 6.1 (4.3-7.6) |
Patients with Hepatic Dysfunction Intramuscular and Oral (n = 7) mean age = 51, range = 43-64 | 0.029 (0.013-0.066) | 0.033 (0.019-0.051) | 5.4 (2.2-6.9) | 4.5 (1.6-7.6) |
Patients with Renal Impairment Intramuscular (n = 25), Oral (n = 9) serum creatinine = 1.9 to 5.0 mg/dL mean age (IM) = 54, range 35-71 mean age (Oral)=57, range=39 - 70 | 0.015 (0.005-0.043) | 0.016 (0.007-0.052) | 10.3 (5.9-19.2) | 10.8 (3.4-18.9) |
Renal Dialysis Patients Intramuscular and Oral (n = 9) mean age = 40, range=27-63 | 0.016 (0.003-0.036) | --- | 13.6 (8.0-39.1) | --- |
1 Estimated from 30 mg single intramuscular doses of ketorolac tromethamine
2 Estimated from 10 mg single oral doses of ketorolac tromethamine
3 Liters/hours/kilogram
Intravenous-Administration: In normal subjects (n=37), the total clearance of 30 mg intravenous-administered ketorolac tromethamine was 0.030 (0.017-0.051) L/h/kg. The terminal half-life was 5.6 (4.0-7.9) hours. (See Kinetics in Special Populations for use of intravenous dosing of ketorolac tromethamine in pediatric patients.)
CLINICAL STUDIES
Adult Patients
In a postoperative study, where all patients received morphine by a PCA device, patients treated with ketorolac tromethamine intravenous as fixed intermittent boluses (e.g., 30 mg initial dose followed by 15 mg every 3 hours), required significantly less morphine (26%) than the placebo group. Analgesia was significantly superior, at various postdosing pain assessment times, in the patients receiving ketorolac tromethamine intravenous plus PCA morphine as compared to patients receiving PCA-administered morphine alone.
HOW SUPPLIED
Ketorolac Tromethamine Injection, USP is supplied as follows:
NDC 83270-157-01, 15 mg/mL, 1 mL pre-filled syringe, in carton of 1’s
NDC 83270-158-01, 30 mg/mL, 1 mL pre-filled syringe, in carton of 1’s
•NDC 83270-159-01, 60 mg/2 mL, 2 mL pre-filled syringe, in carton of 1’s
•FOR INTRAMUSCULAR USE ONLY
Each pre-filled syringe is supplied with an individually wrapped BD SafetyGlide™ Needle (22Guage x 1½ inch).
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Protect from light.
Retain in carton until time of use. Discard unused portion.
Manufactured by: Onesource Specialty Pharma Limited, Sterile Product Division, Opp IIM, Bilekahalli, Bannerghatta Road, Bengaluru, Karnataka, India-560076
Revised: 09/2025
