Ipratropium Bromide And Albuterol Sulfate - Ipratropium Bromide And Albuterol Sulfate solution prescribing information
INDICATIONS AND USAGE
Ipratropium Bromide and Albuterol Sulfate Inhalation Solution is indicated for the treatment of bronchospasm associated with COPD in patients requiring more than one bronchodilator.
DOSAGE AND ADMINISTRATION
The recommended dose of Ipratropium Bromide and Albuterol Sulfate Inhalation Solution is one 3 mL vial administered 4 times per day via nebulization with up to 2 additional 3 mL doses allowed per day, if needed. Safety and efficacy of additional doses or increased frequency of administration of Ipratropium Bromide and Albuterol Sulfate Inhalation Solution beyond these guidelines has not been studied and the safety and efficacy of extra doses of albuterol sulfate or ipratropium bromide in addition to the recommended doses of Ipratropium Bromide and Albuterol Sulfate Inhalation Solution have not been studied.
The use of Ipratropium Bromide and Albuterol Sulfate Inhalation Solution can be continued as medically indicated to control recurring bouts of bronchospasm. If a previously effective regimen fails to provide the usual relief, medical advice should be sought immediately, as this is often a sign of worsening COPD, which would require reassessment of therapy.
A Pari-LC-Plus™ nebulizer (with face mask or mouthpiece) connected to a PRONEB™ compressor was used to deliver Ipratropium Bromide and Albuterol Sulfate Inhalation Solution to each patient in one U.S. clinical study. The safety and efficacy of Ipratropium Bromide and Albuterol Sulfate Inhalation Solution delivered by other nebulizers and compressors have not been established.
Ipratropium Bromide and Albuterol Sulfate Inhalation Solution should be administered via jet nebulizer connected to an air compressor with an adequate air flow, equipped with a mouthpiece or suitable face mask.
CONTRAINDICATIONS
Ipratropium Bromide and Albuterol Sulfate Inhalation Solution is contraindicated in patients with a history of hypersensitivity to any of its components, or to atropine and its derivatives.
ADVERSE REACTIONS
Adverse reaction information concerning Ipratropium Bromide and Albuterol Sulfate Inhalation Solution was derived from the 12-week controlled clinical trial.
ADVERSE EVENTS OCCURRING IN ≥ 1% OF ≥ 1 TREATMENT GROUP(S) AND WHERE THE COMBINATION TREATMENT SHOWED THE HIGHEST PERCENTAGE | |||
Body System COSTART Term | Albuterol n (%) | Ipratropium n (%) | lpratropium Bromide and Albuterol Sulfate n (%) |
NUMBER OF PATIENTS | 761 | 754 | 765 |
N (%) Patients with AE | 327 (43.0) | 329 (43.6) | 367 (48.0) |
BODY AS A WHOLE | |||
Pain | 8 (1.1) | 4 (0.5) | 10 (1.3) |
Pain chest | 11 (1 .4) | 14 (1.9) | 20 (2.6) |
DIGESTIVE | |||
Diarrhea | 5 (0.7) | 9 (1.2) | 14 (1.8) |
Dyspepsia | 7 (0.9) | 8 (1.1) | 10 (1.3) |
Nausea | 7 (0.9) | 6 (0.8) | 11 (1.4) |
MUSCULO-SKELETAL | |||
Cramps Leg | 8 (1.1) | 6 (0.8) | 11 (1.4) |
RESPIRATORY | |||
Bronchitis | 11 (1 .4) | 13 (1.7) | 13(1.7) |
Lung Disease | 36 (4.7) | 34 (4.5) | 49 (6.4) |
Pharyngitis | 27 (3.5) | 27 (3.6) | 34 (4.4) |
Pneumonia | 7 (0.9) | 8 (1.1) | 10 (1.3) |
UROGENITAL | |||
Infection urinary tract | 3 (0.4) | 9 (1.2) | 12(1.6) |
Additional adverse reactions reported in more than 1% of patients treated with Ipratropium Bromide and Albuterol Sulfate Inhalation Solution included constipation and voice alterations.
In the clinical trial, there was a 0.3% incidence of possible allergic-type reactions, including skin rash, pruritis, and urticaria.
Additional information derived from the published literature on the use of albuterol sulfate and ipratropium bromide singly or in combination includes precipitation or worsening of narrow-angle glaucoma, acute eye pain, blurred vision, mydriasis, paradoxical bronchospasm, wheezing, exacerbation of COPD symptoms, drowsiness, aching, flushing, upper respiratory tract infection, palpitations, taste perversion, elevated heart rate, sinusitis, back pain, sore throat and metabolic acidosis. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
DESCRIPTION
The active components in Ipratropium Bromide and Albuterol Sulfate Inhalation Solution are albuterol sulfate and ipratropium bromide. Albuterol sulfate is a salt of racemic albuterol and a relatively selective β 2-adrenergic bronchodilator chemically described as α 1-[( tert -Butylamino) methyl]-4-hydroxy- m -xylene- α,α′-diol sulfate (2:1) (salt). It has a molecular weight of 576.7 and the empirical formula is (C 13 H 21 NO 3 ) 2 •H 2 SO 4 . It is a white crystalline powder, soluble in water and slightly soluble in ethanol. The World Health Organization’s recommended name for albuterol base is salbutamol.
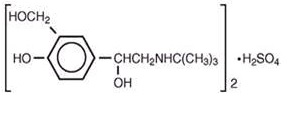
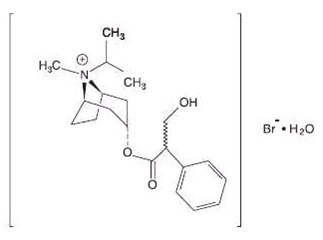
Ipratropium bromide is an anticholinergic bronchodilator chemically described as 8-azoniabicyclo[3.2.1]-octane,3-(3-hydroxy-1-oxo-2-phenylpropoxy)-8-methyl-8-(1-methylethyl)-, bromide, monohydrate (endo,syn)-, (±)-; a synthetic quaternary ammonium compound, chemically related to atropine. It has a molecular weight of 430.4 and the empirical formula is C 20 H 30 BrNO 3 •H 2 O. It is a white crystalline substance, freely soluble in water and lower alcohols, and insoluble in lipophilic solvents such as ether, chloroform, and fluorocarbons.
Each 3 mL vial of Ipratropium Bromide and Albuterol Sulfate Inhalation Solution contains 3 mg (0.1%) of albuterol sulfate (equivalent to 2.5 mg (0.083%) of albuterol base) and 0.5 mg (0.017%) of ipratropium bromide in an isotonic, sterile, aqueous solution containing sodium chloride and hydrochloric acid to adjust to pH 4.
Ipratropium Bromide and Albuterol Sulfate Inhalation Solution is a clear, colorless solution. It does not require dilution prior to administration by nebulization. For Ipratropium Bromide and Albuterol Sulfate Inhalation Solution, like all other nebulized treatments, the amount delivered to the lungs will depend on patient factors, the jet nebulizer utilized, and compressor performance. Using the Pari-LC-Plus™ nebulizer (with face mask or mouthpiece) connected to a PRONEB™ compressor system, under in vitro conditions, the mean delivered dose from the mouth piece (% nominal dose) was approximately 46% of albuterol and 42% of ipratropium bromide at a mean flow rate of 3.6 L/min. The mean nebulization time was 15 minutes or less. Ipratropium Bromide and Albuterol Sulfate Inhalation Solution should be administered from jet nebulizers at adequate flow rates, via face masks or mouthpieces (see DOSAGE AND ADMINISTRATION ).
CLINICAL PHARMACOLOGY
Ipratropium Bromide and Albuterol Sulfate Inhalation Solution is a combination of the β 2-adrenergic bronchodilator, albuterol sulfate, and the anticholinergic bronchodilator, ipratropium bromide.
Mechanism of Action
Albuterol sulfate
The prime action of β-adrenergic drugs is to stimulate adenyl cyclase, the enzyme that catalyzes the formation of cyclic-3’,5’-adenosine monophosphate (cAMP) from adenosine triphosphate (ATP). The cAMP thus formed mediates the cellular responses. In vitro studies and in vivo pharmacologic studies have demonstrated that albuterol has a preferential effect on β 2-adrenergic receptors compared with isoproterenol. While it is recognized that β 2-adrenergic receptors are the predominant receptors in bronchial smooth muscle, recent data indicated that 10% to 50% of the β-receptors in the human heart may be β 2-receptors. The precise function of these receptors, however, is not yet established. Albuterol has been shown in most controlled clinical trials to have more effect on the respiratory tract, in the form of bronchial smooth muscle relaxation, than isoproterenol at comparable doses while producing fewer cardiovascular effects. Controlled clinical studies and other clinical experience have shown that inhaled albuterol, like other β-adrenergic agonist drugs, can produce a significant cardiovascular effect in some patients.
Ipratropium bromide
Ipratropium bromide is an anticholinergic (parasympatholytic) agent, which blocks the muscarinic receptors of acetylcholine, and, based on animal studies, appears to inhibit vagally mediated reflexes by antagonizing the action of acetylcholine, the transmitter agent released from the vagus nerve. Anticholinergics prevent the increases in intracellular concentration of cyclic guanosine monophosphate (cGMP), resulting from the interaction of acetylcholine with the muscarinic receptors of bronchial smooth muscle.
Ipratropium Bromide and Albuterol Sulfate Inhalation Solution
Ipratropium Bromide and Albuterol Sulfate Inhalation Solution is expected to maximize the response to treatment in patients with chronic obstructive pulmonary disease (COPD) by reducing bronchospasm through two distinctly different mechanisms: sympathomimetic (albuterol sulfate) and anticholinergic / parasympatholytic (ipratropium bromide). Simultaneous administration of both an anticholinergic and a β 2-sympathomimetic is designed to produce greater bronchodilation effects than when either drug is utilized alone at its recommended dosage.
Pharmacokinetics
Albuterol sulfate
Albuterol sulfate is longer acting than isoproterenol in most patients by any route of administration, because it is not a substrate for the cellular uptake processes for catecholamines nor for the metabolism of catechol-O-methyl transferase. Instead the drug is conjugatively metabolized to albuterol 4’-O - sulfate.
Ipratropium bromide
The bronchodilation following inhalation of ipratropium is primarily a local, site-specific effect, not a systemic one. Much of an inhaled dose is swallowed as shown by fecal excretion studies. Following nebulization of a 1 mg dose to healthy volunteers, a mean of 4% of the dose was excreted unchanged in the urine.
Ipratropium bromide is minimally (0% to 9% in vitro ) bound to plasma albumin and α 1–acid glycoproteins. It is partially metabolized to inactive ester hydrolysis products. Following intravenous administration, approximately one-half is excreted unchanged in the urine. The half-life of elimination is about 1.6 hours after intravenous administration. Ipratropium bromide that reaches the systemic circulation is reportedly removed by the kidneys rapidly at a rate that exceeds the glomerular filtration rate. The pharmacokinetics of Ipratropium Bromide and Albuterol Sulfate Inhalation Solution or ipratropium bromide have not been studied in the elderly and in patients with hepatic or renal insufficiency (see PRECAUTIONS ).
Ipratropium Bromide and Albuterol Sulfate Inhalation Solution
In a double-blind, double period, crossover study, 15 male and female subjects were administered single doses of Ipratropium Bromide and Albuterol Sulfate Inhalation Solution or albuterol sulfate inhalation solution at two times the recommended single doses as two inhalations separated by 15 minutes. The total nebulized dose of albuterol sulfate from both treatments was 6.0 mg and the total dose of ipratropium bromide from Ipratropium Bromide and Albuterol Sulfate Inhalation Solution was 1.0 mg. Peak albuterol plasma concentrations occurred at 0.8 hours after dosing for both treatments. The mean peak albuterol concentration following administration of albuterol sulfate alone was 4.86 (± 2.65) ng/mL and it was 4.65 (± 2.92) ng/mL for Ipratropium Bromide and Albuterol Sulfate Inhalation Solution. Mean AUC values for the two treatments were 26.6 (± 15.2) ng•hr/mL (albuterol sulfate alone) versus 24.2 (± 14.5) ng•hr/mL (Ipratropium Bromide and Albuterol Sulfate Inhalation Solution). The mean t 1/2 values were 7.2 (± 1.3) hours (albuterol sulfate alone) and 6.7 (± 1.7) hours (Ipratropium Bromide and Albuterol Sulfate Inhalation Solution). A mean of 8.4 (± 8.9)% of the albuterol dose was excreted unchanged in urine following administration of two vials of Ipratropium Bromide and Albuterol Sulfate Inhalation Solution which is similar to 8.8 (± 7.3)% that was obtained from albuterol sulfate inhalation solution. There were no statistically significant differences in the pharmacokinetics of albuterol between the two treatments. For ipratropium, a mean of 3.9 (± 5.1)% of the ipratropium bromide dose was excreted unchanged in the urine following two vials of Ipratropium Bromide and Albuterol Sulfate Inhalation Solution, which is comparable with previously reported data.
CLINICAL STUDIES
In a 12-week, randomized, double-blind, positive-control, crossover study of albuterol sulfate, ipratropium bromide and Ipratropium Bromide and Albuterol Sulfate Inhalation Solution, 863 COPD patients were evaluated for bronchodilator efficacy comparing Ipratropium Bromide and Albuterol Sulfate Inhalation Solution with albuterol sulfate and ipratropium bromide alone.
Ipratropium Bromide and Albuterol Sulfate Inhalation Solution demonstrated significantly better changes in FEV 1 , as measured from the baseline to peak response, when compared with either albuterol sulfate or ipratropium bromide.
Ipratropium Bromide and Albuterol Sulfate Inhalation Solution was also shown to have the rapid onset associated with albuterol sulfate, with a mean time to peak FEV 1 of 1.5 hours, and the extended duration associated with ipratropium bromide with a duration of 15% response in FEV 1 of 4.3 hours.
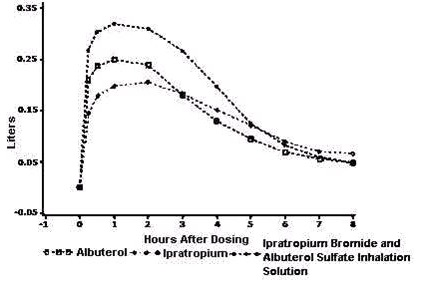
This study demonstrated that each component of Ipratropium Bromide and Albuterol Sulfate Inhalation Solution contributed to the improvement in pulmonary function, especially during the first 4 to 5 hours after dosing, and that Ipratropium Bromide and Albuterol Sulfate Inhalation Solution was significantly more effective than albuterol sulfate or ipratropium bromide alone.
HOW SUPPLIED
Ipratropium Bromide and Albuterol Sulfate Inhalation Solution is supplied as a 3 mL sterile solution for nebulization in sterile low-density polyethylene unit-dose vials. Store vials in pouch until time of use. Supplied in cartons as listed below.
NDC 62135-831-84: 6 foil pouches, each containing 5 vials, total 30 vials per carton
Storage and Handling
PROTECT FROM LIGHT. The unit-dose vial should remain stored in the protective foil pouch until time of use.
Store between 2° and 25° C (36° and 77° F). Protect from light.
Rx Only
Manufactured for: Chartwell RX, LLC.
Congers, NY 10920
INSTRUCTIONS FOR USE
FOLLOW THESE DIRECTIONS FOR USE OF YOUR NEBULIZER / COMPRESSOR OR THE DIRECTIONS GIVEN BY YOUR HEALTHCARE PROVIDER. A TYPICAL EXAMPLE IS SHOWN NEARBY.
- Remove one vial from the foil pouch. Place remaining vials back into the pouch for storage.
- Twist the cap completely off the vial and squeeze the contents into the nebulizer reservoir (Figure 1).
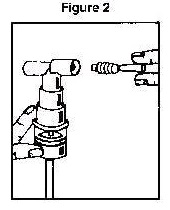
3. Connect the nebulizer to the mouthpiece or face mask (Figure 2).
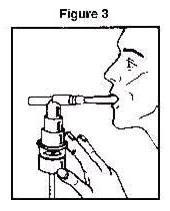
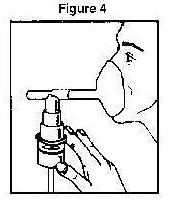
4. Connect the nebulizer to the compressor 5. Sit in a comfortable, upright position; place the mouthpiece in your mouth (Figure 3) or put on the face mask (Figure 4); and turn on the compressor.
6. Breathe as calmly, deeply and evenly as possible through your mouth until no more mist is formed in the nebulizer chamber (about 5 - 15 minutes). At this point, the treatment is finished. 7. Clean the nebulizer (see manufacturer's instructions).
L71928
Revised: 01/2024