Get your patient on Indapamide - Indapamide tablet, Film Coated (Indapamide)
Indapamide - Indapamide tablet, Film Coated prescribing information
DOSAGE AND ADMINISTRATION
Hypertension
The adult starting indapamide dose for hypertension is 1.25 mg as a single daily dose taken in the morning. If the response to 1.25 mg is not satisfactory after 4 weeks, the daily dose may be increased to 2.5 mg taken once daily. If the response to 2.5 mg is not satisfactory after 4 weeks, the daily dose may be increased to 5 mg taken once daily, but adding another antihypertensive should be considered.
Edema of Congestive Heart Failure
The adult starting indapamide dose for edema of congestive heart failure is 2.5 mg as a single daily dose taken in the morning. If the response to 2.5 mg is not satisfactory after one week, the daily dose may be increased to 5 mg taken once daily.
If the antihypertensive response to indapamide is insufficient, indapamide may be combined with other antihypertensive drugs, with careful monitoring of blood pressure. It is recommended that the usual dose of other agents be reduced by 50% during initial combination therapy. As the blood pressure response becomes evident, further dosage adjustments may be necessary.
In general, doses of 5 mg and larger have not appeared to provide additional effects on blood pressure or heart failure, but are associated with a greater degree of hypokalemia. There is minimal clinical trial experience in patients with doses greater than 5 mg once a day.
CONTRAINDICATIONS
Anuria.
Known hypersensitivity to indapamide or to other sulfonamide-derived drugs.
ADVERSE REACTIONS
Most adverse effects have been mild and transient.
The clinical adverse reactions listed in Table 1 represent data from Phase II/III placebo-controlled studies (306 patients given indapamide 1.25 mg). The clinical adverse reactions listed in Table 2 represent data from Phase II placebo-controlled studies and long-term controlled clinical trials (426 patients given indapamide 2.5 mg or 5 mg). The reactions are arranged into two groups: 1) a cumulative incidence equal to or greater than 5%; 2) a cumulative incidence less than 5%. Reactions are counted regardless of relation to drug.
Incidence ≥ 5% | Incidence < 5% 1 |
BODY AS A WHOLE | |
Headache Infection Pain Back Pain | Asthenia Flu Syndrome Abdominal Pain Chest Pain |
GASTROINTESTINAL SYSTEM | |
Constipation Diarrhea Dyspepsia Nausea | |
METABOLIC SYSTEM | |
Peripheral Edema | |
CENTRAL NERVOUS SYSTEM | |
Dizziness | Nervousness Hypertonia |
RESPIRATORY SYSTEM | |
Rhinitis | Cough Pharyngitis Sinusitis |
SPECIAL SENSES | |
Conjunctivitis | |
1 OTHER All other clinical adverse reactions occurred at an incidence of < 1%. | |
Approximately 4% of patients given indapamide 1.25 mg compared to 5% of the patients given placebo discontinued treatment in the trials of up to 8 weeks because of adverse reactions.
In controlled clinical trials of 6 to 8 weeks in duration, 20% of patients receiving indapamide 1.25 mg, 61% of patients receiving indapamide 5 mg, and 80% of patients receiving indapamide 10 mg had at least one potassium value below 3.4 mEq/L. In the indapamide 1.25 mg group, about 40% of those patients who reported hypokalemia as a laboratory adverse event returned to normal serum potassium values without intervention. Hypokalemia with concomitant clinical signs or symptoms occurred in 2% of patients receiving indapamide 1.25 mg.
Incidence ≥ 5% | Incidence < 5% |
CENTRAL NERVOUS SYSTEM/NEUROMUSCULAR | |
Headache Dizziness Fatigue, weakness, loss of energy, lethargy, tiredness, or malaise Muscle cramps or spasm, or numbness of the extremities Nervousness, tension, anxiety, irritability, or agitation | Lightheadedness Drowsiness Vertigo Insomnia Depression Blurred Vision |
GASTROINTESTINAL SYSTEM | |
Constipation Nausea Vomiting Diarrhea Gastric irritation Abdominal pain or cramps Anorexia | |
CARDIOVASCULAR SYSTEM | |
Orthostatic hypotension Premature ventricular contractions Irregular heart beat Palpitations | |
GENITOURINARY SYSTEM | |
Frequency of urination Nocturia Polyuria | |
DERMATOLOGIC/HYPERSENSITIVITY | |
Rash Hives Pruritus Vasculitis | |
OTHER | |
Impotence or reduced libido Rhinorrhea Flushing Hyperuricemia Hyperglycemia Hyponatremia Hypochloremia Increase in serum urea nitrogen (BUN) or creatinine Glycosuria Weight loss Dry mouth Tingling of extremities | |
Because most of these data are from long-term studies (up to 40 weeks of treatment), it is probable that many of the adverse experiences reported are due to causes other than the drug. Approximately 10% of patients given indapamide discontinued treatment in long-term trials because of reactions either related or unrelated to the drug.
Hypokalemia with concomitant clinical signs or symptoms occurred in 3% of patients receiving indapamide 2.5 mg q.d. and 7% of patients receiving indapamide 5 mg q.d. In long-term controlled clinical trials comparing the hypokalemic effects of daily doses of indapamide and hydrochlorothiazide, however, 47% of patients receiving indapamide 2.5 mg, 72% of patients receiving indapamide 5 mg, and 44% of patients receiving hydrochlorothiazide 50 mg had at least one potassium value (out of a total of 11 taken during the study) below 3.5 mEq/L. In the indapamide 2.5 mg group, over 50% of those patients returned to normal serum potassium values without intervention.
In clinical trials of 6 to 8 weeks, the mean changes in selected values were as shown in the tables below.
| Mean Changes from Baseline after 8 Weeks of Treatment - 1.25 mg | |||||
|---|---|---|---|---|---|
| Serum Electrolytes (mEq/L) | Serum Uric Acid (mg/dL) | BUN (mg/dL) | |||
| Potassium | Sodium | Chloride | |||
Indapamide 1.25 mg (n=255 to 257) | -0.28 | -0.63 | -2.60 | 0.69 | 1.46 |
Placebo (n=263 to 266) | 0.00 | -0.11 | -0.21 | 0.06 | 0.06 |
No patients receiving indapamide 1.25 mg experienced hyponatremia considered possibly clinically significant (< 125 mEq/L).
Indapamide had no adverse effects on lipids.
| Mean Changes from Baseline after 40 Weeks of Treatment - 2.5 mg and 5 mg | |||||
|---|---|---|---|---|---|
| Serum Electrolytes (mEq/L) | Serum Uric Acid (mg/dL) | BUN (mg/dL) | |||
| Potassium | Sodium | Chloride | |||
Indapamide 2.5 mg (n=76) | -0.4 | -0.6 | -3.6 | 0.7 | -0.1 |
Indapamide 5 mg (n=81) | -0.6 | -0.7 | -5.1 | 1.1 | 1.4 |
The following reactions have been reported with clinical usage of indapamide: jaundice (intrahepatic cholestatic jaundice), hepatitis, pancreatitis, and abnormal liver function tests. These reactions were reversible with discontinuance of the drug.
Also reported are erythema multiforme, Stevens-Johnson Syndrome, bullous eruptions, purpura, photosensitivity, fever, pneumonitis, anaphylactic reactions, agranulocytosis, leukopenia, thrombocytopenia, and aplastic anemia. Other adverse reactions reported with antihypertensive/diuretics are necrotizing angiitis, respiratory distress, sialadenitis, xanthopsia.
Postmarketing Experience
Eye Disorders
Choroidal effusion, acute myopia, and angle-closure glaucoma (frequency not known). 1
1 Module 2.5 SER-indapamide-choroidal effusion-acute myopia and angle-closure glaucoma-Jul2020.
To report SUSPECTED ADVERSE REACTIONS, contact ANI Pharmaceuticals, Inc. at 1-855-204-1431 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Other Antihypertensives
Indapamide may add to or potentiate the action of other antihypertensive drugs. In limited controlled trials that compared the effect of indapamide combined with other antihypertensive drugs with the effect of the other drugs administered alone, there was no notable change in the nature or frequency of adverse reactions associated with the combined therapy.
Lithium
See WARNINGS .
Post-Sympathectomy Patient
The antihypertensive effect of the drug may be enhanced in the post-sympathectomized patient.
Norepinephrine
Indapamide, like the thiazides, may decrease arterial responsiveness to norepinephrine, but this diminution is not sufficient to preclude effectiveness of the pressor agent for therapeutic use.
DESCRIPTION
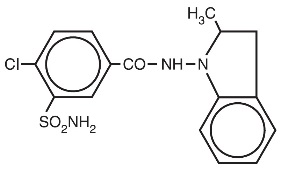
Indapamide is an oral antihypertensive/diuretic. Its molecule contains both a polar sulfamoyl chlorobenzamide moiety and a lipid-soluble methylindoline moiety. It differs chemically from the thiazides in that it does not possess the thiazide ring system and contains only one sulfonamide group. The chemical name of indapamide is 4-Chloro- N -(2-methyl-1-indolinyl)-3-sulfamoylbenzamide, and its molecular weight is 365.84. The compound is a weak acid, pK a =8.8, and is soluble in aqueous solutions of strong bases. It is a white to yellow-white crystalline (tetragonal) powder.
Each tablet, for oral administration, contains 1.25 mg or 2.5 mg of indapamide USP and the following inactive ingredients: corn starch, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, talc, and titanium dioxide. Additionally, the 1.25 mg product contains FD&C yellow #6 aluminum lake.
CLINICAL PHARMACOLOGY
Indapamide is the first of a new class of antihypertensive/diuretics, the indolines. The oral administration of 2.5 mg (two 1.25 mg tablets) of indapamide to male subjects produced peak concentrations of approximately 115 ng/mL of the drug in the blood within 2 hours. The oral administration of 5 mg (two 2.5 mg tablets) of indapamide to healthy male subjects produced peak concentrations of approximately 260 ng/mL of the drug in the blood within 2 hours. A minimum of 70% of a single oral dose is eliminated by the kidneys and an additional 23% by the gastrointestinal tract, probably including the biliary route. The half-life of indapamide in whole blood is approximately 14 hours.
Indapamide is preferentially and reversibly taken up by the erythrocytes in the peripheral blood. The whole blood/plasma ratio is approximately 6:1 at the time of peak concentration and decreases to 3.5:1 at 8 hours. From 71% to 79% of the indapamide in plasma is reversibly bound to plasma proteins.
Indapamide is an extensively metabolized drug, with only about 7% of the total dose administered, recovered in the urine as unchanged drug during the first 48 hours after administration. The urinary elimination of 14 C-labeled indapamide and metabolites is biphasic with a terminal half-life of excretion of total radioactivity of 26 hours.
In a parallel design double-blind, placebo-controlled trial in hypertension, daily doses of indapamide between 1.25 mg and 10 mg produced dose-related antihypertensive effects. Doses of 5 mg and 10 mg were not distinguishable from each other although each was differentiated from placebo and 1.25 mg indapamide. At daily doses of 1.25 mg, 5 mg and 10 mg, a mean decrease of serum potassium of 0.28, 0.61 and 0.76 mEq/L, respectively, was observed and uric acid increased by about 0.69 mg/100 mL.
In other parallel design, dose-ranging clinical trials in hypertension and edema, daily doses of indapamide between 0.5 mg and 5 mg produced dose related effects. Generally, doses of 2.5 mg and 5 mg were not distinguishable from each other although each was differentiated from placebo and from 0.5 mg or 1 mg indapamide. At daily doses of 2.5 mg and 5 mg a mean decrease of serum potassium of 0.5 and 0.6 mEq/Liter, respectively, was observed and uric acid increased by about 1 mg/100 mL.
At these doses, the effects of indapamide on blood pressure and edema are approximately equal to those obtained with conventional doses of other antihypertensive/diuretics.
In hypertensive patients, daily doses of 1.25 mg, 2.5 mg and 5 mg of indapamide have no appreciable cardiac inotropic or chronotropic effect. The drug decreases peripheral resistance with little or no effect on cardiac output, rate or rhythm. Chronic administration of indapamide to hypertensive patients has little or no effect on glomerular filtration rate or renal plasma flow.
Indapamide had an antihypertensive effect in patients with varying degrees of renal impairment, although in general, diuretic effects declined as renal function decreased.
In a small number of controlled studies, indapamide taken with other antihypertensive drugs such as hydralazine, propranolol, guanethidine, and methyldopa, appeared to have the additive effect typical of thiazide-type diuretics.
HOW SUPPLIED
Indapamide Tablets USP are available containing 1.25 mg or 2.5 mg of indapamide USP.
The 1.25 mg tablets are orange, round, film coated tablets debossed ‘ANI’ over ‘510’ on one side and plain on the other side. They are available in bottles of 100 tablets (NDC 62559-510-01).
The 2.5 mg tablets are white, round, film coated tablets debossed ‘ANI’ over ‘511’ on one side and plain on the other side. They are available in bottles of 100 tablets (NDC 62559-511-01).
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Keep container tightly closed.
Avoid excessive heat.
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
Manufactured by: ANI Pharmaceuticals, Inc. Baudette, MN 56623 
9957 Rev 05/25