Ibuprofen 400mg prescribing information
Cardiovascular Thrombotic Events
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use (see Warnings and Precautions ).
- Ibuprofen tablets are contraindicated in the setting of coronary artery bypass graft (CABG) surgery (see Contraindications and Warnings ).
Gastrointestinal Risk
- NSAIDS cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events. (See WARNINGS ).
INDICATIONS AND USAGE
Carefully consider the potential benefits and risks of Ibuprofen tablets and other treatment options before deciding to use ibuprofen. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ).
Ibuprofen tablets are indicated for relief of the signs and symptoms of rheumatoid arthritis and osteoarthritis.
Ibuprofen tablets are indicated for relief of mild to moderate pain.
Ibuprofen tablets are also indicated for the treatment of primary dysmenorrhea.
Controlled clinical trials to establish the safety and effectiveness of Ibuprofen tablets in children have not been conducted.
DOSAGE AND ADMINISTRATION
Carefully consider the potential benefits and risks of Ibuprofen tablets and other treatment options before deciding to use Ibuprofen tablets. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ).
After observing the response to initial therapy with Ibuprofen tablets, the dose and frequency should be adjusted to suit an individual patient’s needs. Do not exceed 3,200 mg total daily dose. If gastrointestinal complaints occur, administer Ibuprofen tablets with meals or milk.
Rheumatoid arthritis and osteoarthritis, including flare-ups of chronic disease:
Suggested Dosage : 1,200 mg to 3,200 mg daily (400 mg, 600 mg or 800 mg tid or qid). Individual patients may show a better response to 3,200 mg daily, as compared with 2,400 mg, although in well-controlled clinical trials patients on 3,200 mg did not show a better mean response in terms of efficacy. Therefore, when treating patients with 3,200 mg/day, the physician should observe sufficient increased clinical benefits to offset potential increased risk. The dose should be tailored to each patient, and may be lowered or raised depending on the severity of symptoms either at time of initiating drug therapy or as the patient responds or fails to respond. In general, patients with rheumatoid arthritis seem to require higher doses of Ibuprofen tablets than do patients with osteoarthritis.
The smallest dose of Ibuprofen tablets that yields acceptable control should be employed. A linear blood level dose-response relationship exists with single doses up to 800 mg (See CLINICAL PHARMACOLOGY for effects of food on rate of absorption).
The availability of three tablet strengths facilitates dosage adjustment. In chronic conditions, a therapeutic response to therapy with Ibuprofen tablets is sometimes seen in a few days to a week but most often is observed by two weeks. After a satisfactory response has been achieved, the patient’s dose should be reviewed and adjusted as required.
Mild to moderate pain:
400 mg every 4 to 6 hours as necessary for relief of pain. In controlled analgesic clinical trials, doses of Ibuprofen tablets greater than 400 mg were no more effective than the 400 mg dose.
Dysmenorrhea:
For the treatment of dysmenorrhea, beginning with the earliest onset of such pain, Ibuprofen tablets should be given in a dose of 400 mg every 4 hours as necessary for the relief of pain.
CONTRAINDICATIONS
Ibuprofen tablets are contraindicated in patients with known hypersensitivity to ibuprofen.
Ibuprofen tablets should not be given to patients who have experienced asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients (see WARNINGS, Anaphylactoid Reactions , and PRECAUTIONS, Preexisting Asthma ).
In the setting of coronary artery bypass graft (CABG) surgery [see Warnings ].
ADVERSE REACTIONS
The most frequent type of adverse reaction occurring with Ibuprofen tablets is gastrointestinal. In controlled clinical trials the percentage of patients reporting one or more gastrointestinal complaints ranged from 4% to 16%.
In controlled studies when Ibuprofen tablets were compared to aspirin and indomethacin in equally effective doses, the overall incidence of gastrointestinal complaints was about half that seen in either the aspirin- or indomethacin-treated patients.
Adverse reactions observed during controlled clinical trials at an incidence greater than 1% are listed in the table. Those reactions listed in Column one encompass observations in approximately 3,000 patients. More than 500 of these patients were treated for periods of at least 54 weeks.
Still other reactions occurring less frequently than 1 in 100 were reported in controlled clinical trials and from marketing experience. These reactions have been divided into two categories: Column two of the table lists reactions with therapy with Ibuprofen tablets where the probability of a causal relationship exists: for the reactions in Column three, a causal relationship with Ibuprofen tablets has not been established.
Reported side effects were higher at doses of 3,200 mg/day than at doses of 2,400 mg or less per day in clinical trials of patients with rheumatoid arthritis. The increases in incidence were slight and still within the ranges reported in the table.
| Incidence Greater than 1% (but less than 3%) Probable Causal Relationship• | Precise Incidence Unknown (but less than 1%) Probable Causal Relationship•• | Precise Incidence Unknown (but less than 1%) Causal Relationship Unknown•• |
| GASTROINTESTINAL | Gastric or duodenal ulcer with bleeding and/or perforation, gastrointestinal hemorrhage melena, gastritis, hepatitis, jaundice, abnormal liver function tests; pancreatitis | |
| Nausea•, epigastric pain•, heartburn•, diarrhea, abdominal distress, nausea and vomiting, indigestion, constipation, abdominal cramps or pain, fullness of GI tract (bloating and flatulence) | ||
| CENTRAL NERVOUS SYSTEM | Depression, insomnia, confusion, emotional liability, somnolence, aseptic meningitis with fever and coma (see PRECAUTIONS ) | Paresthesias, hallucinations, |
| dream abnormalities, | ||
| Dizziness•, headache, nervousness | pseudotumor cerebri | |
| DERMATOLOGIC | Vesiculobullous eruptions, urticaria, erythema multiforme, Stevens- Johnson syndrome, alopecia | Toxic epidermal necrolysis, photoallergic skin reactions |
| Rash•, (including maculopapular type), pruritus | ||
| SPECIAL SENSES | Hearing loss, amblyopia (blurred and/or diminished vision, scotomata and /or changes in color vision) (see PRECAUTIONS ) | Conjunctivitis, diplopia, optic neuritis, cataracts |
| Tinnitus | ||
| HEMATOLOGIC | Neutropenia, agranulocytosis, aplastic anemia, hemolytic anemia (sometimes Coombs positive), thrombocytopenia with or without purpura, eosinophilia, decreases in hemoglobin and hematocrit (see PRECAUTIONS ) | Bleeding episodes (eg epistaxis, menorrhagia) |
| METABOLIC/ENDOCRINE | Gynecomastia, hypoglycemic reaction, acidosis | |
| Decreased appetite | ||
| CARDIOVASCULAR | Congestive heart failure in patients with marginal cardiac function, elevated blood pressure, palpitations | Arrhythmias (sinus tachycardia, sinus bradycardia) |
| Edema, fluid retention (generally responds promptly to drug discontinuation) (see PRECAUTIONS ) | ||
| ALLERGIC | Syndrome of abdominal pain, fever, chills, nausea and vomiting; anaphylaxis; bronchospasm (see CONTRAINDICATIONS ) | Serum sickness, Iupus erythematosus syndrome. Henoch-Schonlein vasculitis, angioedema |
| RENAL | Acute renal failure (see PRECAUTIONS ), decreased creatinine clearance, poliuria, azotemia, cystitis, hematuria | Renal papillary necrosis |
| MISCELLANEOUS | Dry eyes and mouth, gingival ulcer, rhinitis | |
| •Reactions occurring in 3% to 9% of patients treated with IBU (those reactions occurring in less than 3% of the patients are unmarked.) | ||
| ••Reactions are classified under “Probable Causal Relationship (PCR)” if there has been one positive rechallenge or if three or more cases occur which might be causally related. Reactions are classified under | ||
| “Causal Relationship Unknown” if seven or more events have been reported but the criteria for PCR have not been met. | ||
Post marketing Experience
The following adverse reactions have been identified during post approval use of ibuprofen. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin and Appendages: Exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), toxic epidermal necrolysis (TEN), and fixed drug eruption (FDE).
Drug Interactions
ACE-inhibitors: Reports suggest that NSAIDs may diminish the antihypertensive effect of ACE-inhibitors. This interaction should be given consideration in patients taking NSAIDs concomitantly with ACE-inhibitors.
DESCRIPTION
Ibuprofen Tablets, USP contain the active ingredient Ibuprofen, USP which is (±) -2 - ( p - isobutylphenyl) propionic acid. Ibuprofen is a white powder with a melting point of 74° C to 77° C and is very slightly soluble in water (<1 mg/mL) and readily soluble in organic solvents such as ethanol and acetone. The structural formula is represented below: 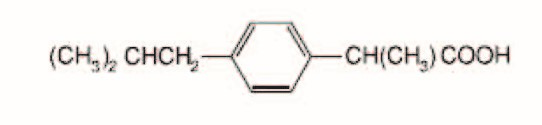
Ibuprofen, a nonsteroidal anti-inflammatory drug (NSAID), is available in 400 mg, 600 mg, and 800 mg tablets for oral administration. Inactive ingredients: carnauba wax, colloidal silicon dioxide, croscarmellose sodium, microcrystalline cellulose, polysorbate 80, povidone, pregelatinized starch, sodium starch glycolate, stearic acid, hypromellose, polyethylene glycol and propylene glycol.
CLINICAL PHARMACOLOGY
Ibuprofen tablets contain ibuprofen which possesses analgesic and antipyretic activities. Its mode of action, like that of other NSAIDs, is not completely understood, but may be related to prostaglandin synthetase inhibition.
In clinical studies in patients with rheumatoid arthritis and osteoarthritis, Ibuprofen tablets have been shown to be comparable to aspirin in controlling pain and inflammation and to be associated with a statistically significant reduction in the milder gastrointestinal side effects (see ADVERSE REACTIONS ). Ibuprofen may be well tolerated in some patients who have had gastrointestinal side effects with aspirin, but these patients when treated with Ibuprofen tablets should be carefully followed for signs and symptoms of gastrointestinal ulceration and bleeding. Although it is not definitely known whether ibuprofen causes less peptic ulceration than aspirin, in one study involving 885 patients with rheumatoid arthritis treated for up to one year, there were no reports of gastric ulceration with ibuprofen whereas frank ulceration was reported in 13 patients in the aspirin group (statistically significant p<.001).
Gastroscopic studies at varying doses show an increased tendency toward gastric irritation at higher doses. However, at comparable doses, gastric irritation is approximately half that seen with aspirin. Studies using 51 Cr-tagged red cells indicate that fecal blood loss associated with Ibuprofen tablets in doses up to 2,400 mg daily did not exceed the normal range, and was significantly less than that seen in aspirin-treated patients.
In clinical studies in patients with rheumatoid arthritis, Ibuprofen has been shown to be comparable to indomethacin in controlling the signs and symptoms of disease activity and to be associated with a statistically significant reduction of the milder gastrointestinal (see ADVERSE REACTIONS ) and CNS side effects.
Ibuprofen may be used in combination with gold salts and/or corticosteroids.
Controlled studies have demonstrated that Ibuprofen is a more effective analgesic than propoxyphene for the relief of episiotomy pain, pain following dental extraction procedures, and for the relief of the symptoms of primary dysmenorrhea.
In patients with primary dysmenorrhea, Ibuprofen has been shown to reduce elevated levels of prostaglandin activity in the menstrual fluid and to reduce resting and active intrauterine pressure, as well as the frequency of uterine contractions. The probable mechanism of action is to inhibit prostaglandin synthesis rather than simply to provide analgesia.
Pharmacodynamics
In a healthy volunteer study, ibuprofen 400 mg given once daily, administered 2 hours prior to immediate-release aspirin (81 mg) for 6 days, showed an interaction with the antiplatelet activity of aspirin as measured by % serum thromboxane B2 (TxB2) inhibition at 24 hours following the day-6 aspirin dose [53%]. An interaction was still observed, but minimized, when ibuprofen 400 mg given once-daily was administered as early as 8 hours prior to the immediate-release aspirin dose [90.7%]. However, there was no interaction with the antiplatelet activity of aspirin when ibuprofen 400 mg, given once daily, was administered 2 hours after (but not concomitantly, 15 min, or 30 min after) the immediate-release aspirin dose [99.2%].
In another study, where immediate-release aspirin 81 mg was administered once daily with ibuprofen 400 mg given three times daily (1, 7, and 13 hours post-aspirin dose) for 10 consecutive days, the mean % serum thromboxane B2 (TxB2) inhibition suggested no interaction with the antiplatelet activity of aspirin [98.3%]. However, there were individual subjects with serum TxB2 inhibition below 95%, with the lowest being 90.2%.
When a similarly designed study was conducted with enteric-coated aspirin, where healthy subjects were administered enteric-coated aspirin 81 mg once daily for 6 days and ibuprofen 400 mg three times daily (2, 7 and 12 h post-aspirin dose) for 6 days, there was an interaction with the antiplatelet activity at 24 hours following the day-6 aspirin dose [67%]. [ See Precautions/Drug Interactions ].
Pharmacokinetics
The ibuprofen in Ibuprofen tablets is rapidly absorbed. Peak serum ibuprofen levels are generally attained one to two hours after administration. With single doses up to 800 mg, a linear relationship exists between amount of drug administered and the integrated area under the serum drug concentration vs time curve. Above 800 mg, however, the area under the curve increases less than proportional to increases in dose. There is no evidence of drug accumulation or enzyme induction.
The administration of Ibuprofen tablets either under fasting conditions or immediately before meals yields quite similar serum ibuprofen concentration-time profiles. When Ibuprofen is administered immediately after a meal, there is a reduction in the rate of absorption but no appreciable decrease in the extent of absorption. The bioavailability of the drug is minimally altered by the presence of food.
A bioavailability study has shown that there was no interference with the absorption of ibuprofen when given in conjunction with an antacid containing both aluminum hydroxide and magnesium hydroxide.
Ibuprofen is rapidly metabolized and eliminated in the urine. The excretion of ibuprofen is virtually complete 24 hours after the last dose. The serum half-life is 1.8 to 2.0 hours.
Studies have shown that following ingestion of the drug, 45% to 79% of the dose was recovered in the urine within 24 hours as metabolite A (25%), (+)-2-[ p -(2hydroxymethyl-propyl) phenyl] propionic acid and metabolite B (37%), (+)-2-[ p -(2carboxypropyl) phenyl]propionic acid; the percentages of free and conjugated ibuprofen were approximately 1% and 14%, respectively.
HOW SUPPLIED
Ibuprofen Tablets, USP are available in the following strengths:
The 400 mg tablets are film-coated clear, round shaped, white tablets, debossed “C5” on one side and plain on the other side.
They are available as follows:
Bottles of 100 tablets – NDC 0904-7587-59
Bottles of 500 tablets – NDC 0904-7587-40 (PACKAGE NOT CHILD-RESISTANT)
The 600 mg tablets are film -coated clear, oval-shaped, white tablets, debossed “C7” on one side and plain on the other side.
They are available as follows:
Bottles of 100 tablets – NDC 0904-7588-59
Bottles of 500 tablets – NDC 0904-7588-40 (PACKAGE NOT CHILD-RESISTANT)
The 800 mg tablets are film -coated clear, oval-shaped, white tablets, debossed “C11” on one side and plain on the other side.
They are available as follows:
Bottles of 100 tablets – NDC 0904-7592-59
Bottles of 500 tablets – NDC 0904-7592-40 (PACKAGE NOT CHILD-RESISTANT)
Store at 20º to 25ºC (68º to 77ºF); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Avoid excessive heat 40°C (104°F).
Manufactured by:
Contract Pharmacal Corp.
165 Oser Avenue
Hauppauge, NY 11788 USA
Toll Free Number: (800) 231-4670
Distributed by:
MAJOR® PHARMACEUTICALS
Indianapolis, IN 46268
Rev. 08/25