Hydrocodone Bitartrate And Aspirin - Hydrocodone Bitartrate And Aspirin tablet prescribing information
WARNING: SERIOUS AND LIFE-THREATENING RISKS FROM USE OF HYDROCODONE BITARTRATE AND ASPIRIN TABLETS
Addition, Abuse and Misuse
Because the use of Hydrocodone Bitartrate and Aspirin Tablets exposes patients and other users to the risks of opioid addiction, abuse, and misuse, which can lead to overdose and death, assess each patient’s risk prior to prescribing and reassess all patients regularly for the development of these behaviors and conditions [see Warnings ].
Life-Threatening Respiratory Depression
Serious, life-threatening, or fatal respiratory depression may occur with use of Hydrocodone Bitartrate and Aspirin Tablets, especially during initiation or following a dosage increase. To reduce the risk of respiratory depression, proper dosing and titration of Hydrocodone Bitartrate and Aspirin Tablets are essential [see Warnings ].
Accidental Ingestion
Accidental ingestion of even one dose of Hydrocodone Bitartrate and Aspirin Tablets, especially by children, can result in a fatal overdose of hydrocodone [see Warnings ].
Risks From Concomitant Use With Benzodiazepines Or Other CNS Depressants
Concomitant use of opioids with benzodiazepines or other central nervous system (CNS) depressants, including alcohol, may result in profound sedation, respiratory depression, coma, and death. Reserve concomitant prescribing of Hydrocodone Bitartrate and Aspirin Tablets and benzodiazepines or other CNS depressants for use in patients for whom alternative treatment options are inadequate [see Warnings , Precautions ; Drug Interactions ].
Neonatal Opioid Withdrawal Syndrome (NOWS)
If opioid use is required for an extended period of time in a pregnant woman, advise the patient of the risk of NOWS, which may be life-threatening if not recognized and treated. Ensure that management by neonatology experts will be available at delivery [see Warnings ].
Opioid Analgesic Risk Evaluation and Mitigation Strategy (REMS)
Healthcare providers are strongly encouraged to complete a REMS complaint education program and to counsel patients and caregivers on serious risks, safe use, and the importance of reading the Medication Guide with each prescription [see Warnings ].
Cytochrome P450 3A4 Interaction
The concomitant use of Hydrocodone Bitartrate and Aspirin Tablets with all Cytochrome P450 3A4 inhibitors may result in an increase in hydrocodone plasma concentrations, which could increase or prolong adverse reactions and may cause potentially fatal respiratory depression. In addition, discontinuation of a concomitantly used Cytochrome P450 3A4 inducer may result in an increase in hydrocodone plasma concentrations. Monitor patients receiving Hydrocodone Bitartrate and Aspirin Tablets and any Cytochrome P450 3A4 inhibitor or inducer for signs of respiratory depression or sedation [see CLINICAL PHARMACOLOGY , WARNINGS , PRECAUTIONS ; Drug Interactions ].
INDICATIONS AND USAGE
Hydrocodone Bitartrate and Aspirin Tablets are indicated for the management of pain severe enough to require an opioid analgesic and for which alternative treatments are inadequate.
Limitations of Use
Because of the risks of addiction, abuse, and misuse, with opioids, which can occur at any dosage or duration, reserve Hydrocodone Bitartrate and Aspirin Tablets for use in patients for whom alternative treatment options (e.g., non-opioid analgesics):
- have not been tolerated or are not expected to be tolerated.
- have not provided adequate analgesia or are not expected to provide adequate analgesia.
Hydrocodone Bitartrate and Aspirin Tablets should not be used for extended period of time unless the pain remains severe enough to require an opioid analgesic and for which alternative treatment options continue to be inadequate.
DOSAGE AND ADMINISTRATION
Important Dosage and Administration Instructions
Hydrocodone Bitartrate and Aspirin Tablets should be prescribed only by healthcare professionals who are knowledgeable about the use of opioids and how to mitigate the associated risks.
Use the lowest effective dosage for the shortest duration of time consistent with the individual patient treatment goals [see WARNINGS ]. Because the risk of overdose increases as opioid doses increase, reserve titration to higher doses of Hydrocodone Bitartrate with Aspirin Tablets for patients in whom lower doses are insufficiently effective and in whom the expected benefits of using higher dose opioid clearly outweigh the substantial risks.
Many acute pain conditions (e.g., the pain that occurs with a number of surgical procedures or acute musculoskeletal injuries) require no more than a few days of an opioid analgesic. Clinical guidelines on opioid prescribing for some acute pain conditions are available.
There is variability in the opioid analgesic dose and duration needed to adequately manage pain due both to the cause of pain and to individual patient factors. Initiate the dosing regimen for each patient individually, taking into account the patient’s underlying cause and severity of pain, prior analgesic treatment and response, and risk factors for addition, abuse, and misuse [see WARNINGS ].
Respiratory depression can occur at any time during opioid therapy, especially when initiating and following dosage increase with Hydrocodone Bitartrate and Aspirin Tablets. Consider the risk when selecting an initial dose and when making dose adjustments [see WARNINGS ]. Administer Hydrocodone Bitartrate and Aspirin Tablets with food or a full glass of water to minimize GI distress.
Patient Access to Naloxone for the Emergency Treatment of Opioid Overdose
- Discuss the availability of naloxone for the emergency treatment of opioid overdose with the patient and caregiver and assess the potential need for access to naloxone, both when initiating and renewing treatment with Hydrocodone Bitartrate and Aspirin Tablets [see WARNINGS ; Life-Threatening Respiratory Depression , PRECAUTIONS ; Information for Patients/Caregivers ].
- Inform patients and caregivers about the various ways to obtain naloxone as permitted by individual state naloxone dispensing and prescribing requirements or guidelines (e.g., by prescription, directly from a pharmacist, or as part of a community-based program).
- Consider prescribing naloxone, based on the patient’s risk factors for overdose, such as concomitant use of CNS depressants, a history of opioid use disorder, or prior opioid overdose. The presence of risk factors for overdose should not prevent the proper management of pain in any given patient [see WARNINGS ; Addiction, Abuse, and Misuse ; Life-Threatening Respiratory Depression ; Life-Threatening Respiratory Depression ; Risks from Concomitant Use with Benzodiazepines or Other CNS Depressants ] .
- Consider prescribing naloxone when there are household members (including children) or other close contacts at risk for accidental ingestion or overdose .
Initial Dosage
Initiating Treatment with Hydrocodone Bitartrate and Aspirin Tablets
Initiate treatment with Hydrocodone Bitartrate and Aspirin Tablets using one (1) or two (2) tablets every four (4) to six (6) hours, as needed for pain at the lowest dose necessary to achieve adequate analgesia. Titrate the dose based upon the individual patient’s response to their initial dose of Hydrocodone Bitartrate and Aspirin Tablets.
Conversion from Other Opioids to Hydrocodone Bitartrate and Aspirin Tablets
There is inter-patient variability in the potency of opioid drugs and opioid formulations. Therefore, a conservative approach is advised when determining the total daily dosage of Hydrocodone Bitartrate and Aspirin Tablets. It is safer to underestimate a patient’s 24-hour Hydrocodone Bitartrate and Aspirin Tablets dosage than to overestimate the 24-hour Hydrocodone Bitartrate and Aspirin Tablets dosage and manage an adverse reaction due to an overdose.
Conversion from Hydrocodone Bitartrate and Aspirin Tablets to Extended-Release Hydrocodone
The relative bioavailability of Hydrocodone Bitartrate and Aspirin Tablets compared to extended-release hydrocodone is unknown, so conversion to extended-release hydrocodone may lead to increased risk of excessive sedation and respiratory depression.
Titration and Maintenance of Therapy
Individually titrate Hydrocodone Bitartrate and Aspirin Tablets to a dose that provides adequate analgesia and minimizes adverse reactions. Continually reevaluate patients receiving Hydrocodone Bitartrate and Aspirin Tablets to assess the maintenance of pain control, signs and symptoms of opioid withdrawal, and other adverse reactions, as well as reassess for the development of addiction, abuse, or misuse [see WARNINGS ]. Frequent communication is important among the prescriber, other members of the healthcare team, the patient, and the caregiver/family during periods of changing analgesic requirements, including initial titration.
If the level of pain increases after dosage stabilization, attempt to identify the source of the increased pain before increasing the Hydrocodone Bitartrate and Aspirin Tablets dosage. If after increasing the dosage, unacceptable opioid-related adverse reactions are observed (including an increase in pain after dosage increase), consider reducing the dosage [see WARNINGS ]. Adjust the dosage to obtain an appropriate balance between management of pain and opioid-related adverse reactions.
Safe Reduction or Discontinuation of Hydrocodone Bitartrate and Aspirin Tablets
Do not abruptly discontinue Hydrocodone Bitartrate and Aspirin Tablets in patients who may be physically dependent on opioids. Rapid discontinuation of opioid analgesics in patients who are physically dependent on opioids has resulted in serious withdrawal symptoms, uncontrolled pain, and suicide. Rapid discontinuation has also been associated with attempts to find other sources of opioid analgesics, which may be confused with drug-seeking for abuse. Patients may also attempt to treat their pain or withdrawal symptoms with illicit opioids, such as heroin, and other substances.
When a decision has been made to decrease the dose or discontinue therapy in an opioid-dependent patient taking Hydrocodone Bitartrate and Aspirin Tablets, there are a variety of factors that should be considered, including the total daily dose of opioid (including Hydrocodone Bitartrate and Aspirin Tablets) the patient has been taking, the duration of treatment, the type of pain being treated, and the physical and psychological attributes of the patient. It is important to ensure ongoing care of the patient and to agree on an appropriate tapering schedule and follow-up plan so that patient and provider goals and expectations are clear and realistic. When opioid analgesics are being discontinued due to a suspected substance use disorder, evaluate and treat the patient, or refer for evaluation and treatment of the substance use disorder. Treatment should include evidence-based approaches, such as medication assisted treatment of opioid use disorder. Complex patients with co-morbid pain and substance use disorders may benefit from referral to a specialist.
There are no standard opioid tapering schedules that are suitable for all patients. Good clinical practice dictates a patient-specific plan to taper the dose of the opioid gradually. For patients on Hydrocodone Bitartrate and Aspirin Tablets who are physically opioid-dependent, initiate the taper by a small enough increment (e.g., no greater than 10% to 25% of the total daily dose) to avoid withdrawal symptoms, and proceed with dose-lowering at an interval of every 2 to 4 weeks. Patients who have been taking opioids for briefer periods of time may tolerate a more rapid taper.
It may be necessary to provide the patient with lower dosage strengths to accomplish a successful taper. Reassess the patient frequently to manage pain and withdrawal symptoms, should they emerge. Common withdrawal symptoms include restlessness, lacrimation, rhinorrhea, yawning, perspiration, chills, myalgia, and mydriasis. Other signs and symptoms also may develop, including irritability, anxiety, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhea, or increased blood pressure, respiratory rate, or heart rate. If withdrawal symptoms arise, it may be necessary to pause the taper for a period of time or raise the dose of the opioid analgesic to the previous dose, and then proceed with a slower taper. In addition, evaluate patients for any changes in mood, emergence of suicidal thoughts, or use of other substances.
When managing patients taking opioid analgesics, particularly those who have been treated for an extended period of time, and/or with high doses for chronic pain, ensure that a multimodal approach to pain management, including mental health support (if needed), is in place prior to initiating an opioid analgesic taper. A multimodal approach to pain management may optimize the treatment of chronic pain, as well as assist with the successful tapering of the opioid analgesic [see WARNINGS ; Withdrawal , DRUG ABUSE AND DEPENDENCE ].
ADVERSE REACTIONS
The following serious adverse reactions are described, or described in greater detail, in other sections:
- Addiction, Abuse, and Misuse [see WARNINGS ]
- Life-Threatening Respiratory Depression [see WARNINGS ]
- Interactions with Benzodiazepines and Other CNS Depressants [see WARNINGS ]
- Neonatal Opioid Withdrawal Syndrome [see WARNINGS ]
- Opioid-Induced Hyperalgesia and Allodynia [see WARNINGS ]
- Adrenal Insufficiency [see WARNINGS ]
- Severe Hypotension [see WARNINGS ]
- Gastrointestinal Adverse Reactions [see WARNINGS ]
- Seizures [see WARNINGS ]
- Withdrawal [see WARNINGS ]
- Coagulation Abnormalities and Bleeding [see WARNINGS ]
- Reye’s Syndrome [see WARNINGS ]
- Renal Toxicity and Hyperkalemia [see PRECAUTIONS ]
- Premature Closure of the Fetal Ductus Arteriosus [see PRECAUTIONS ]
Postmarketing Experience
The following adverse reactions associated with the use of Hydrocodone Bitartrate and Aspirin Tablets were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Many adverse reactions due to aspirin ingestion are dose-related. The following is a list of adverse reactions that have been reported in the literature [see WARNINGS ].
Body as a Whole: Fever, hypothermia, thirst.
Cardiovascular: Dysrhythmias, hypotension, tachycardia.
Central Nervous System: Agitation, cerebral edema, coma, confusion, dizziness, headache, subdural or intracranial hemorrhage, lethargy, seizures.
Fluid and Electrolyte: Dehydration, hyperkalemia, metabolic acidosis, respiratory alkalosis.
Gastrointestinal: Dyspepsia, GI bleeding, ulceration and perforation, nausea, vomiting, transient elevations of hepatic enzymes, hepatitis, Reye's Syndrome, pancreatitis.
Hematologic: Prolongation of the prothrombin time, disseminated intravascular coagulation, coagulopathy, thrombocytopenia.
Hypersensitivity: Acute anaphylaxis, angioedema, asthma, bronchospasm, laryngeal edema, urticaria.
Musculoskeletal: Rhabdomyolysis.
Metabolism: Hypoglycemia (in children), hyperglycemia.
Reproductive: Prolonged pregnancy and labor, stillbirths, lower birth weight infants, antepartum and postpartum bleeding.
Respiratory: Hyperpnea, pulmonary edema, tachypnea.
Special Senses: Hearing loss, tinnitus. Patients with high frequency hearing loss may have difficulty perceiving tinnitus. In these patients, tinnitus cannot be used as a clinical indicator of salicylism.
Urogenital: Interstitial nephritis, papillary necrosis, proteinuria, renal insufficiency and failure.
Serotonin syndrome : Cases of serotonin syndrome, a potentially life-threatening condition, have been reported during concomitant use of opioids with serotonergic drugs.
Adrenal insufficiency : Cases of adrenal insufficiency have been reported with opioid use, more often following greater than one month of use.
Anaphylaxis : Anaphylaxis has been reported with ingredients contained in Hydrocodone Bitartrate and Aspirin Tablets.
Androgen deficiency : Cases of androgen deficiency have with use of opioids for an extended period of time [see CLINICAL PHARMACOLOGY ].
Hyperalgesia and Allodynia: Cases of hyperalgesia and allodynia have been reported with opioid therapy of any duration [see WARNINGS ].
Hypoglycemia: Cases of hypoglycemia have been reported in patients taking opioids. Most reports were in patients with at least one predisposing risk factor (e.g., diabetes).
To report SUSPECTED ADVERSE REACTIONS, contact LGM Pharma Solutions, LLC at 1-877-288-1495 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
| Inhibitors of CYP3A4 and CYP2D6 | |
| Clinical Impact: | The concomitant use of Hydrocodone Bitartrate and Aspirin Tablets and CYP3A4 inhibitors can increase the plasma concentration of hydrocodone, resulting in increased or prolonged opioid effects. These effects could be more pronounced with concomitant use of Hydrocodone Bitartrate and Aspirin Tablets and CYP2D6 and CYP3A4 inhibitors, particularly when an inhibitor is added after a stable dose of Hydrocodone Bitartrate and Aspirin Tablets is achieved [see WARNINGS ]. After stopping CYP3A4 inhibitor, as the effects of the inhibitor decline, the hydrocodone plasma concentration will decrease [see CLINICAL PHARMACOLOGY ], resulting in decreased opioid efficacy or a withdrawal syndrome in patients who had developed physical dependence to hydrocodone. |
| Intervention: | If concomitant use is necessary, consider dosage reduction of Hydrocodone Bitartrate and Aspirin Tablets until stable drug effects are achieved. Evaluate patients at frequent intervals for respiratory depression and sedation. If a CYP3A4 inhibitor is discontinued, consider increasing the Hydrocodone Bitartrate and Aspirin Tablets dosage until stable drug effects are achieved. Evaluate for signs of opioid withdrawal. |
| Examples: | Macrolide antibiotics (e.g., erythromycin), azole-antifungal agents (e.g. ketoconazole), protease inhibitors (e.g., ritonavir) |
| CYP3A4 Inducers | |
| Clinical Impact: | The concomitant use of Hydrocodone Bitartrate and Aspirin Tablets and CYP3A4 inducers can decrease the plasma concentration of hydrocodone [see CLINICAL PHARMACOLOGY ], resulting in decreased efficacy or onset of a withdrawal syndrome in patients who have developed physical dependence to hydrocodone [see WARNINGS ]. After stopping a CYP3A4 inducer, as the effects of the inducer decline, the hydrocodone plasma concentration will increase [see CLINICAL PHARMACOLOGY ], which could increase or prolong both the therapeutic effects and adverse reactions, and may cause serious respiratory depression. |
| Intervention: | If concomitant use is necessary, consider increasing the Hydrocodone Bitartrate and Aspirin Tablets dosage until stable drug effects are achieved. Evaluate for signs of opioid withdrawal. If a CYP3A4 inducer is discontinued, consider Hydrocodone Bitartrate and Aspirin Tablets dosage reduction and evaluate patients at frequent intervals for signs of respiratory depression and sedation. |
| Examples: | Rifampin, carbamazepine, phenytoin |
| Benzodiazepines and other Central Nervous System (CNS) Depressants | |
| Clinical Impact: | Due to additive pharmacologic effect, the concomitant use of benzodiazepines or other CNS depressants including alcohol, increases the risk of respiratory depression, profound sedation, coma, and death. |
| Intervention: | Reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Inform patients and caregivers of this potential interaction, educate them on the signs and symptoms of respiratory depression (including sedation). If concomitant use is warranted, consider prescribing naloxone for the emergency treatment of opioid overdose [see WARNINGS ]. |
| Examples: | Benzodiazepines and other sedatives/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, other opioids, alcohol. |
| Serotonergic Drugs | |
| Clinical Impact: | The concomitant use of opioids with other drugs that affect the serotonergic neurotransmitter system has resulted in serotonin syndrome. |
| Intervention: | If concomitant use is warranted, frequently evaluate the patient, particularly during treatment initiation and dose adjustment. Discontinue Hydrocodone Bitartrate and Aspirin Tablets if serotonin syndrome is suspected. |
| Examples: | Selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), triptans, 5-HT3 receptor antagonists, drugs that effect the serotonin neurotransmitter system (e.g., mirtazapine, trazodone, tramadol), certain muscle relaxants ( i.e. , cyclobenzaprine, metaxalone), monoamine oxidase (MAO) inhibitors (those intended to treat psychiatric disorders and also others, such as linezolid and intravenous methylene blue). |
| Monoamine Oxidase Inhibitors (MAOIs) | |
| Clinical Impact: | MAOI interactions with opioids may manifest as serotonin syndrome or opioid toxicity (e.g., respiratory depression, coma) [see WARNINGS ]. |
| Intervention: | The use of Hydrocodone Bitartrate and Aspirin Tablets is not recommended for patients taking MAOIs or within 14 days of stopping such treatment. If urgent use of an opioid is necessary, use test doses and frequent titration of small doses to treat pain while closely evaluating blood pressure and signs and symptoms of CNS and respiratory depression. |
| Examples: | Phenelzine, tranylcypromine, linezolid |
| Mixed Agonist/Antagonist and Partial Agonist Opioid Analgesics | |
| Clinical Impact: | May reduce the analgesic effect of Hydrocodone Bitartrate and Aspirin Tablets and/or precipitate withdrawal symptoms |
| Intervention: | Avoid concomitant use. |
| Examples: | Butorphanol, nalbuphine, pentazocine, buprenorphine |
| Muscle Relaxants | |
| Clinical Impact: | Hydrocodone may enhance the neuromuscular blocking action of skeletal muscle relaxants and produce an increased degree of respiratory depression. |
| Intervention: | Because respiratory depression may be greater than otherwise expected, decrease the dosage of Hydrocodone Bitartrate and Aspirin Tablets and/or the muscle relaxant as necessary. Due to the risk of respiratory depression with concomitant use of muscle relaxants and opioids, consider prescribing naloxone for the emergency treatment of opioid overdose. |
| Diuretics | |
| Clinical Impact: | Opioids can reduce the efficacy of diuretics by inducing the release of antidiuretic hormone. The effectiveness of diuretics in patients with underlying renal or cardiovascular disease may be diminished by the concomitant administration of aspirin due to inhibition of renal prostaglandins, leading to decreased renal blood flow and salt and fluid retention. |
| Intervention: | Evaluate patients for signs of diminished diuresis and/or effects on blood pressure and increase the dosage of the diuretic as needed. |
| Anticholinergic Drugs | |
| Clinical Impact: | The concomitant use of anticholinergic drugs may increase risk of urinary retention and/or severe constipation, which may lead to paralytic ileus. |
| Intervention: | Evaluate patients for signs of urinary retention or reduced gastric motility when Hydrocodone Bitartrate and Aspirin Tablets is used concomitantly with anticholinergic drugs. |
| Anticoagulants | |
| Clinical Impact: | Aspirin may enhance the effects of anticoagulants. Concurrent use may increase the risk of bleeding. Aspirin can also displace warfarin from protein binding sites, leading to prolongation of both the prothrombin time and the bleeding time. |
| Intervention: | Follow patients for signs of bleeding. |
| Examples: | Warfarin, heparin, enoxaparin, clopidogrel, prasugrel, rivaroxaban, apixaban |
| Uricosuric Agents | |
| Clinical Impact: | Aspirin inhibits the uricosuric effects of uricosuric agents. |
| Intervention: | Avoid concomitant use. |
| Examples: | Probenecid |
| Carbonic Anhydrase Inhibitors | |
| Clinical Impact: | Concurrent use with aspirin can lead to high serum concentrations of the carbonic anhydrase inhibitor and cause toxicity due to competition at the renal tubule for secretion. |
| Intervention: | Consider reducing the dose of the carbonic anhydrase inhibitor and follow patient for any adverse effects from the carbonic anhydrase inhibitor. |
| Examples: | Acetazolamide, methazolamide |
| Methotrexate | |
| Clinical Impact: | Aspirin may enhance the toxicity of methotrexate by displacing it from its plasma protein binding sites and/or reducing its renal clearance. |
| Intervention: | Use caution if using concomitantly, especially in elderly patients or patients with renal impairment. Follow patients for methotrexate toxicity. |
| Nephrotoxic Agents | |
| Clinical Impact: | Concomitant use with aspirin may lead to additive nephrotoxicity due to the inhibition of renal prostaglandins by aspirin. Also, the plasma concentration of aspirin is increased by conditions that reduce the glomerular filtration rate or tubular secretion. |
| Intervention: | Use Hydrocodone Bitartrate and Aspirin Tablets with caution if used concomitantly with nephrotoxic agents. Closely follow the renal function of patients. |
| Examples: | Aminoglycosides, amphotericin B, systemic bacitracin, cisplatin, cyclosporine, foscarnet, or parenteral vancomycin |
| Angiotensin Converting Enzyme (ACE) Inhibitors | |
| Clinical Impact: | The hyponatremic and hypotensive effects of ACE inhibitors may be diminished by the concomitant administration of aspirin due to its indirect effect on the renin-angiotensin conversion pathway. |
| Intervention: | Use caution if using concomitantly. Follow the blood pressure and renal function of patients. |
| Examples: | Ramipril, captopril |
| Beta Blockers | |
| Clinical Impact: | The hypotensive effects of beta blockers may be diminished by the concomitant administration of aspirin due to inhibition of renal prostaglandins, leading to decreased renal blood flow, and salt and fluid retention. |
| Intervention: | Use caution if using concomitantly. Follow the blood pressure and renal function of patients. |
| Examples: | Metoprolol, propranolol |
| Hypoglycemic Agents | |
| Clinical Impact: | Aspirin may increase the serum glucose-lowering action of insulin and sulfonylureas leading to hypoglycemia. |
| Intervention: | Patients should be advised to consult a physician if any signs or symptoms of hypoglycemia occur. |
| Examples: | Insulin, glimepiride, glipizide |
| Anticonvulsants | |
| Clinical Impact: | Aspirin can displace protein-bound phenytoin and valproic acid, leading to a decrease in the total concentration of phenytoin and an increase in serum valproic acid levels. |
| Intervention: | Use caution if using concomitantly. |
| Examples: | Phenytoin, valproic acid |
| Nonsteroidal Anti-inflammatory Drugs (NSAIDs) | |
| Clinical Impact: | Concurrent use of aspirin with other NSAIDs may increase the risk of bleeding or lead to decreased renal function. Aspirin may enhance serious side effects and toxicity of ketorolac by displacing it from its plasma protein binding sites and/or reducing its renal clearance. |
| Intervention: | Avoid concomitant use. |
| Examples: | Ketoralac, ibuprofen, naproxen, diclofenac |
| Corticosteroids | |
| Clinical Impact: | In patients receiving concomitant corticosteroids and chronic use of aspirin, withdrawal of corticosteroids may result in salicylism because corticosteroids enhance renal clearance of salicylates and their withdrawal is followed by return to normal rates of renal clearance. |
| Intervention: | Avoid concomitant use. |
DESCRIPTION
Hydrocodone Bitartrate and Aspirin Tablets are immediate-release tablets for oral administration only.
Each Hydrocodone Bitartrate and Aspirin Tablet, 5 mg/500 mg contains:
Hydrocodone Bitartrate ……………….. 5 mg
Aspirin ……………………………................ 500 mg
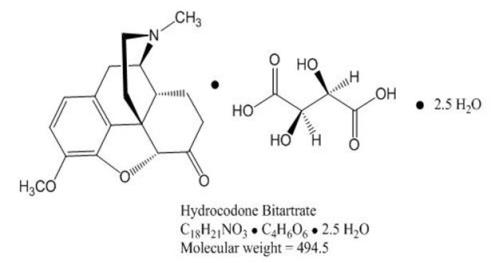
Hydrocodone Bitartrate is an opioid agonist and occurs as fine, white crystals or as a crystalline powder. It is affected by light. It is soluble in water, slightly soluble in alcohol, and insoluble in ether and chloroform. The chemical name is: 4,5α-epoxy-3-methoxy-17-methylmorphinan-6-one tartrate (1:1) hydrate (2:5). It has the following structural formula:
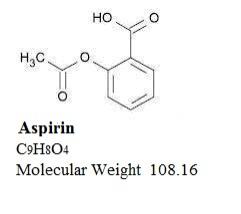
Aspirin, acetylsalicylic acid, is a nonsteroidal anti-inflammatory drug which is an odorless white, needle-like crystalline or powdery substance. The aspirin component is 2-(acetyloxy)-, Benzoic acid. When exposed to moisture, aspirin hydrolyzes into salicylic and acetic acids, and gives off a vinegary-odor. It is highly lipid soluble and slightly soluble in water; freely soluble in alcohol; soluble in chloroform and in ether; sparingly soluble in absolute ether. Its structure is as follows:
In addition, each tablet contains the following inactive ingredients: Microcrystalline Cellulose, Anhydrous Lactose, Corn Starch, Hypromellose, Crospovidone, Stearic Acid, Talc, Colloidal Silicon Dioxide.
CLINICAL PHARMACOLOGY
Mechanism of Action
Hydrocodone is full opioid agonist with relative selectivity for the mu-opioid (μ) receptor, although it can interact with other opioid receptors at higher doses. The principal therapeutic action of hydrocodone is analgesia. Like all full opioid agonists, there is no ceiling effect for analgesia with hydrocodone. Clinically, dosage is titrated to provide adequate analgesia and may be limited by adverse reactions, including respiratory and CNS depression.
The precise mechanism of the analgesic action is unknown. However, specific CNS opioid receptors for endogenous compounds with opioid-like activity have been identified throughout the brain and spinal cord and are thought to play a role in the analgesic effects of this drug.
Aspirin is a nonsteroidal anti-inflammatory drug. Aspirin (acetylsalicylic acid) is a more potent inhibitor of both prostaglandin synthesis and platelet aggregation than other salicylic acid derivatives. The differences in activity between aspirin and salicylic acid are thought to be due to the acetyl group on the aspirin molecule. This acetyl group is responsible for the inactivation of cyclo-oxygenase via acetylation.
Aspirin (acetylsalicylic acid) works by inhibiting the body's production of prostaglandins, including prostaglandins involved in inflammation. Prostaglandins cause pain sensations by stimulating muscle contractions and dilating blood vessels throughout the body. In the CNS, aspirin works on the hypothalamus heat-regulating center to reduce fever, however, other mechanisms may be involved.
Pharmacodynamics
Aspirin affects platelet aggregation by irreversibly inhibiting prostaglandin cyclo-oxygenase. This effect lasts for the life of the platelet and prevents the formation of the platelet aggregating factor thromboxane A2. Nonacetylated salicylates do not inhibit this enzyme and have no effect on platelet aggregation. At somewhat higher doses, aspirin reversibly inhibits the formation of prostaglandin I2 (prostacyclin), which is an arterial vasodilator and inhibits platelet aggregation.
At higher doses aspirin is an effective anti-inflammatory agent, partially due to inhibition of inflammatory mediators via cyclo-oxygenase inhibition in peripheral tissues. In vitro studies suggest that other mediators of inflammation may also be suppressed by aspirin administration, although the precise mechanism of action has not been elucidated. It is this nonspecific suppression of cyclo-oxygenase activity in peripheral tissues following large doses that leads to its primary side effect of gastric irritation. [see ADVERSE REACTIONS ].
Effects on the Central Nervous System
Hydrocodone produces respiratory depression by direct action on brain stem respiratory centers. The respiratory depression involves a reduction in the responsiveness of the brain stem respiratory centers to both increases in carbon dioxide tension and electrical stimulation.
Hydrocodone causes miosis, even in total darkness. Pinpoint pupils are a sign of opioid overdose but are not pathognomonic (e.g., pontine lesions of hemorrhagic or ischemic origins may produce similar findings). Marked mydriasis rather than miosis may be seen due to hypoxia in overdose situations.
Effects on the Gastrointestinal Tract and Other Smooth Muscle
Hydrocodone causes a reduction in motility associated with an increase in smooth muscle tone in the antrum of the stomach and duodenum. Digestion of food in the small intestine is delayed and propulsive contractions are decreased. Propulsive peristaltic waves in the colon are decreased, while tone may be increased to the point of spasm, resulting in constipation. Other opioid-induced effects may include a reduction in biliary and pancreatic secretions, spasm of sphincter of Oddi, and transient elevations in serum amylase.
Aspirin can produce gastrointestinal injury (lesions, ulcers) through a mechanism that is not yet completely understood, but may involve a reduction in eicosanoid synthesis by the gastric mucosa. Decreased production of prostaglandins may compromise the defenses of the gastric mucosa and the activity of substances involved in tissue repair and ulcer healing.
Effects on the Cardiovascular System
Hydrocodone produces peripheral vasodilation which may result in orthostatic hypotension or syncope. Manifestations of histamine release and/or peripheral vasodilation may include pruritus, flushing, red eyes, sweating, and/or orthostatic hypotension.
Effects on the Endocrine System
Opioids inhibit the secretion of adrenocorticotropic hormone (ACTH), cortisol, and luteinizing hormone (LH) in humans [see ADVERSE REACTIONS ]. They also stimulate prolactin, growth hormone (GH) secretion, and pancreatic secretion of insulin and glucagon.
Use of opioids for an extended period of time may influence the hypothalamic-pituitary-gonadal axis, leading to androgen deficiency that may manifest as symptoms as low libido, impotence, erectile dysfunction, amenorrhea, or infertility. The casual role of opioids in the syndrome of hypogonadism is unknown because the various medical, physical, lifestyle, and psychological stressors that may influence gonadal hormone levels have not been adequately controlled for in studies conducted to date [see ADVERSE REACTIONS ].
Effects on the Immune System
Opioids have been shown to have a variety of effects on components of the immune system. The clinical significance of these findings is unknown. Overall, the effects of opioids appear to be modestly immunosuppressive.
Concentration-Efficacy Relationships
The minimum effective analgesic concentration will vary widely among patients, especially among patients who have been previously treated with agonist opioid agonists. The minimum effective analgesic concentration of hydrocodone for any individual patient may increase over time due to an increase in pain, the development of a new pain syndrome, and/or the development of analgesic tolerance [see DOSAGE AND ADMINISTRATION ].
Concentration-Adverse Reaction Relationships
There is a relationship between increasing hydrocodone plasma concentration and increasing frequency of dose-related opioid adverse reactions such as nausea, vomiting, CNS effects, and respiratory depression. In opioid-tolerant patients, the situation may be altered by the development of tolerance to opioid-related adverse reactions [see DOSAGE AND ADMINISTRATION ].
The dose of Hydrocodone Bitartrate and Aspirin Tablets must be individualized because the effective analgesic dose for some patients will be too high to be tolerated by other patients [see DOSAGE AND ADMINISTRATION ].
Pharmacokinetics
The behavior of the individual components is described below.
Hydrocodone
Absorption:
Following a 10 mg oral dose of hydrocodone administered to five adult male subjects, the mean peak concentration was 23.6 ± 5.2 ng/mL. Maximum serum levels were achieved at 1.3 ± 0.3 hours and the half-life was determined to be 3.8 ± 0.3 hours.
Elimination:
Metabolism
Hydrocodone exhibits a complex pattern of metabolism including O-demethylation, N-demethylation and 6-keto reduction to the corresponding 6-α- and 6-β-hydroxymetabolites. CYP3A4 mediated N-demethylation to norhydrocodone is the primary metabolic pathway of hydrocodone with a lower contribution from CYP2D6 mediated O-demethylation to hydromorphone. Hydromorphone is formed from the O-demethylation of hydrocodone and may contribute to the total analgesic effect of hydrocodone. Therefore, the formation of these and related metabolites can, in theory, be affected by other drugs [see PRECAUTIONS ; Drug Interactions ]. N-demethylation of hydrocodone to form norhydrocodone via CYP3A4 while O-demethylation of hydrocodone to hydromorphone is predominantly catalyzed by CYP2D6 and to a lesser extent by an unknown low affinity CYP enzyme.
Excretion
Hydrocodone and its metabolites are eliminated primarily in the kidneys.
Aspirin
Absorption:
In general, immediate release aspirin is well and completely absorbed from the gastrointestinal (GI) tract. Following absorption, aspirin is hydrolyzed to salicylic acid with peak plasma levels of salicylic acid occurring within 1-2 hours of dosing [see Pharmacokinetics — Metabolism ]. The rate of absorption from the GI tract is dependent upon the dosage form, the presence or absence of food, gastric pH (the presence or absence of GI antacids or buffering agents), and other physiologic factors.
Distribution:
Salicylic acid is widely distributed to all tissues and fluids in the body including the central nervous system (CNS), breast milk, and fetal tissues. The highest concentrations are found in the plasma, liver, renal cortex, heart, and lungs. The protein binding of salicylate is concentration-dependent, i.e., nonlinear. At low concentrations (<100 micrograms/milliliter ([micro]g/mL)), approximately 90 percent of plasma salicylate is bound to albumin while at higher concentrations (>400 [micro]g/mL), only about 75 percent is bound.
Elimination:
Metabolism
Aspirin is rapidly hydrolyzed in the plasma to salicylic acid such that plasma levels of aspirin are essentially undetectable 1-2 hours after dosing. Salicylic acid is primarily conjugated in the liver to form salicyluric acid, a phenolic glucuronide, an acyl glucuronide, and a number of minor metabolites. Salicylic acid has a plasma half-life of approximately 6 hours. Salicylate metabolism is saturable and total body clearance decreases at higher serum concentrations due to the limited ability of the liver to form both salicyluric acid and phenolic glucuronide. Following toxic doses (10-20 grams (g)), the plasma half-life may be increased to over 20 hours.
Excretion
The elimination of salicylic acid follows zero order pharmacokinetics; ( i.e., the rate of drug elimination is constant in relation to plasma concentration). Renal excretion of unchanged drug depends upon urine pH. As urinary pH rises above 6.5, the renal clearance of free salicylate increases from <5 percent to >80 percent. Following therapeutic doses, approximately 10 percent is found excreted in the urine as salicylic acid, 75 percent as salicyluric acid, and 10 percent phenolic and 5 percent acyl glucuronides of salicylic acid.
HOW SUPPLIED
Hydrocodone Bitartrate and Aspirin Tablets are supplied as oval, bisected white tablets containing 5 mg Hydrocodone Bitartrate and 500 mg of Aspirin. Each tablet is debossed with “MLB” bisect “125” on one side and “5-500” on the other side.
Bottles of 100 tablets – NDC 79739-7182-1
Store at 25°C (77°F); excursions permitted to 15°-30°C (59°-86°F). [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).
Protect from moisture.
Store Hydrocodone Bitartrate and Aspirin Tablets securely and dispose of properly [see PRECAUTIONS ; Information for Patients/Caregivers ]
Manufactured by: LGM Pharma Solutions, LLC, Irvine, CA 92614
7182-PD
Rev 01/2024
Mechanism of Action
Hydrocodone is full opioid agonist with relative selectivity for the mu-opioid (μ) receptor, although it can interact with other opioid receptors at higher doses. The principal therapeutic action of hydrocodone is analgesia. Like all full opioid agonists, there is no ceiling effect for analgesia with hydrocodone. Clinically, dosage is titrated to provide adequate analgesia and may be limited by adverse reactions, including respiratory and CNS depression.
The precise mechanism of the analgesic action is unknown. However, specific CNS opioid receptors for endogenous compounds with opioid-like activity have been identified throughout the brain and spinal cord and are thought to play a role in the analgesic effects of this drug.
Aspirin is a nonsteroidal anti-inflammatory drug. Aspirin (acetylsalicylic acid) is a more potent inhibitor of both prostaglandin synthesis and platelet aggregation than other salicylic acid derivatives. The differences in activity between aspirin and salicylic acid are thought to be due to the acetyl group on the aspirin molecule. This acetyl group is responsible for the inactivation of cyclo-oxygenase via acetylation.
Aspirin (acetylsalicylic acid) works by inhibiting the body's production of prostaglandins, including prostaglandins involved in inflammation. Prostaglandins cause pain sensations by stimulating muscle contractions and dilating blood vessels throughout the body. In the CNS, aspirin works on the hypothalamus heat-regulating center to reduce fever, however, other mechanisms may be involved.