Get your patient on Humatin - Paromomycin Sulfate capsule (Paromomycin Sulfate)
Humatin - Paromomycin Sulfate capsule prescribing information
INDICATIONS AND USAGE
Paromomycin sulfate is indicated for intestinal amebiasis–acute and chronic (NOTE-It is not effective in extraintestinal amebiasis); management of hepatic coma–as adjunctive therapy.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of HUMATIN™ Capsules and other antibacterial drugs, HUMATIN™ Capsules should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
DOSAGE AND ADMINISTRATION
Intestinal amebiasis: Adults and Pediatric Patients: Usual dose—25 to 35 mg/kg body weight daily, administered in three doses with meals, for five to ten days.
Management of hepatic coma:
Adults: Usual dose—4 g daily in divided doses, given at regular intervals for five to six days.
CONTRAINDICATIONS
Paromomycin sulfate is contraindicated in individuals with a history of previous hypersensitivity reactions to it. It is also contraindicated in intestinal obstruction.
ADVERSE REACTIONS
Nausea, abdominal cramps, and diarrhea have been reported in patients on doses over 3 g daily.
To report SUSPECTED ADVERSE REACTIONS, Waylis Therapeutics LLC at 844-200-7910 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
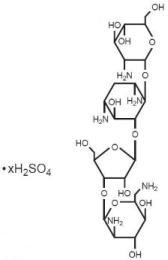
Paromomycin sulfate is a broad spectrum antibiotic produced by Streptomyces riomosus var. paromomycinus. It is a white, amorphous, stable, water-soluble product. Paromomycin sulfate is designated chemically as 0-2, 6-Diamino-2, 6-dideoxy-β -L-idopyranosyl-(1→3)-0-β -D-ribofuranosyl-(1→5)-0-[2-amino-2-deoxy-α -D-glucopyranosyl-(1→4)]-2-deoxystreptamine sulfate (salt). The molecular formula is C 23 H 45 N 5 O 14 •xH 2 SO 4 , with a molecular weight of 615.64 (base).
Its structural formula is:
Each capsule, for oral administration, contains paromomycin sulfate equivalent to 250 mg paromomycin. Each capsule also contains the following inactive ingredients: FD&C Blue # 1, D&C Red # 28, FD&C Red # 40, gelatin and titanium dioxide. The imprinting ink for the 250 mg capsule contains D&C yellow #10, FD&C blue # 1, FD&C blue # 2, FD&C red # 40, iron oxide black, pharmaceutical shellac glaze, and propylene glycol.
CLINICAL PHARMACOLOGY
The in-vitro and in-vivo antibacterial action of paromomycin closely parallels that of neomycin. It is poorly absorbed after oral administration, with almost 100% of the drug recoverable in the stool.
HOW SUPPLIED
HUMATIN™ Capsules each contain paromomycin sulfate equivalent to 250 mg paromomycin, are supplied as follows:
NDC 80725-250-01: Bottles of 100
The capsule is Dark Blue Opaque /White Opaque, imprinted with "HP 38" in black ink on the cap and on the body.