Get your patient on Glycopyrrolate Oral Solution - Glycopyrrolate Oral Solution liquid (Glycopyrrolate Oral Solution)
Glycopyrrolate Oral Solution - Glycopyrrolate Oral Solution liquid prescribing information
1 INDICATIONS AND USAGE
Glycopyrrolate oral solution is indicated to reduce chronic severe drooling in patients aged 3 to 16 years with neurologic conditions associated with problem drooling (e.g., cerebral palsy)
2 DOSAGE AND ADMINISTRATION
Glycopyrrolate oral solution must be measured and administered with an accurate measuring device [see Patient Counseling Information (17)].
Initiate dosing at 0.02 mg/kg orally three times daily and titrate in increments of 0.02 mg/kg every 5-7 days based on therapeutic response and adverse reactions. The maximum recommended dosage is 0.1 mg/kg three times daily not to exceed 1.5-3 mg per dose based upon weight. For greater detail, see Table 1.
During the four-week titration period, dosing can be increased with the recommended dose titration schedule while ensuring that the anticholinergic adverse events are tolerable. Prior to each increase in dose, review the tolerability of the current dose level with the patient’s caregiver.
Glycopyrrolate oral solution should be dosed at least one hour before or two hours after meals.
The presence of high fat food reduces the oral bioavailability of glycopyrrolate oral solution if taken shortly after a meal [see Clinical Pharmacology (12.3)].
| Weight | Dose Level 1 | Dose Level 2 | Dose Level 3 | Dose Level 4 | Dose Level 5 | ||||||
| kg | lbs | (~0.02 mg/kg) | (~0.04 mg/kg) | (~0.06 mg/kg) | (~0.08 mg/kg) | (~0.1 mg/kg) | |||||
| 13-17 | 27-38 | 0.3 mg | 1.5 mL | 0.6 mg | 3 mL | 0.9 mg | 4.5 mL | 1.2 mg | 6 mL | 1.5 mg | 7.5 mL |
| 18-22 | 39-49 | 0.4 mg | 2 mL | 0.8 mg | 4 mL | 1.2 mg | 6 mL | 1.6 mg | 8 mL | 2.0 mg | 10 mL |
| 23-27 | 50-60 | 0.5 mg | 2.5 mL | 1.0 mg | 5 mL | 1.5 mg | 7.5 mL | 2.0 mg | 10 mL | 2.5 mg | 12.5 mL |
| 28-32 | 61-71 | 0.6 mg | 3 mL | 1.2 mg | 6 mL | 1.8 mg | 9 mL | 2.4 mg | 12 mL | 3.0 mg | 15 mL |
| 33-37 | 72-82 | 0.7 mg | 3.5 mL | 1.4 mg | 7 mL | 2.1 mg | 10.5 mL | 2.8 mg | 14 mL | 3.0 mg | 15 mL |
| 38-42 | 83-93 | 0.8 mg | 4 mL | 1.6 mg | 8 mL | 2.4 mg | 12 mL | 3.0 mg | 15 mL | 3.0 mg | 15 mL |
| 43-47 | 94-104 | 0.9 mg | 4.5 mL | 1.8 mg | 9 mL | 2.7 mg | 13.5 mL | 3.0 mg | 15 mL | 3.0 mg | 15 mL |
| ≥48 | ≥105 | 1.0 mg | 5 mL | 2.0 mg | 10 mL | 3.0 mg | 15 mL | 3.0 mg | 15 mL | 3.0 mg | 15 mL |
3 DOSAGE FORMS AND STRENGTHS
Glycopyrrolate oral solution is available as a 1 mg/5 mL clear, cherry-flavored solution for oral administration in 16 ounce bottles.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data in pregnant women for glycopyrrolate oral solution to inform decisions concerning any drug-associated risks. In pregnant rats, daily oral administration of glycopyrrolate during organogenesis at dose exposures 2.5 to 113 times the exposure at the maximum recommended human dose (MRHD) did not result in an increased incidence of gross external or visceral defects [ see Data ]. When glycopyrrolate was administered intravenously to pregnant rabbits during organogenesis at dose exposures equivalent to up to approximately 7.8 times the exposure at the MRHD, no adverse effects on embryo-fetal development were seen. The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Animal Data
Glycopyrrolate was orally administered to pregnant rats at dosages of 50, 200, and 400 mg/kg/day during the period of organogenesis. These dosages resulted in systemic exposures (estimated AUC 0-inf values) approximately 2.5, 23, and 113 times, respectively, the estimated systemic exposure in humans at the MRHD (9 mg per day, administered in three divided doses). Glycopyrrolate had no effect on maternal survival, but significantly reduced mean maternal body weight gain over the period of dosing at all dosages evaluated. Mean fetal weight was significantly reduced in the 200 and 400 mg/kg/day dose groups. There were two litters with all resorbed fetuses in the 400 mg/kg/day dose group. There were no effects of treatment on the incidence of gross external or visceral defects. Minor treatment-related skeletal effects included reduced ossification of various bones in the 200 and 400 mg/kg/day dose groups; these skeletal effects were likely secondary to maternal toxicity.
Glycopyrrolate was intravenously administered to pregnant rabbits at dosages of 0.1, 0.5, and 1.0 mg/kg/day during the period of organogenesis. These dosages resulted in systemic exposures (estimated AUC 0-inf values) approximately 0.8, 4.6, and 7.8 times, respectively, the estimated systemic exposure in humans at the MRHD. Glycopyrrolate did not affect maternal survival under the conditions of this study. Mean maternal body weight gain and mean food consumption over the period of dosing were lower than the corresponding control value in the 0.5 and 1.0 mg/kg/day treatment groups. There were no effects of treatment on fetal parameters, including fetal survival, mean fetal weight, and the incidence of external, visceral, or skeletal defects.
Female rats that were pregnant or nursing were orally dosed with glycopyrrolate daily at dosages of 0, 50, 200, or 400 mg/kg/day, beginning on day 7 of gestation, and continuing until day 20 of lactation. These dosages resulted in systemic exposures (estimated AUC 0-inf values) approximately 2.5, 23, and 113 times, respectively, the estimated systemic exposure in humans at the MRHD (9 mg per day, administered in three divided doses). Mean body weight of pups in all treatment groups was reduced compared to the control group during the period of nursing, but eventually recovered to be comparable to the control group, post-weaning. No other notable delivery or litter parameters were affected by treatment in any group, including no effects on mean duration of gestation or mean numbers of live pups per litter. No treatment-related effects on survival or adverse clinical signs were observed in pups. There were no effects of maternal treatment on behavior, learning, memory, or reproductive function of pups.
8.2 Lactation
Risk Summary
There are no data on the presence of glycopyrrolate or its metabolites in human milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for glycopyrrolate oral solution and any potential adverse effects on the breastfed infant from glycopyrrolate oral solution or from the underlying maternal condition.
8.4 Pediatric Use
Glycopyrrolate oral solution was evaluated for chronic severe drooling in patients aged 3-16 years with neurologic conditions associated with problem drooling. Glycopyrrolate oral solution has not been studied in subjects under the age of 3 years.
8.5 Geriatric Use
Clinical studies of glycopyrrolate oral solution did not include subjects aged 65 and over.
8.6 Renal Impairment
Because glycopyrrolate is largely renally eliminated, glycopyrrolate oral solution should be used with caution in patients with renal impairment [see Clinical Pharmacology (12.3)].
4 CONTRAINDICATIONS
Glycopyrrolate oral solution is contraindicated in:
- Patients with medical conditions that preclude anticholinergic therapy (e.g., glaucoma, paralytic ileus, unstable cardiovascular status in acute hemorrhage, severe ulcerative colitis, toxic megacolon complicating ulcerative colitis, myasthenia gravis).
- Patients taking solid oral dosage forms of potassium chloride. The passage of potassium chloride tablets through the gastrointestinal (GI) tract may be arrested or delayed with coadministration of glycopyrrolate oral solution.
5 WARNINGS AND PRECAUTIONS
5.1 Constipation or Intestinal Pseudo-obstruction
Constipation is a common dose-limiting adverse reaction which sometimes leads to glycopyrrolate discontinuation [see Adverse Reactions (6.1)]. Assess patients for constipation, particularly within 4-5 days of initial dosing or after a dose increase. Intestinal pseudo-obstruction has been reported and may present as abdominal distention, pain, nausea or vomiting.
5.2 Incomplete Mechanical Intestinal Obstruction
Diarrhea may be an early symptom of incomplete mechanical intestinal obstruction, especially in patients with ileostomy or colostomy. If incomplete mechanical intestinal obstruction is suspected, discontinue treatment with glycopyrrolate oral solution and evaluate for intestinal obstruction.
5.3 High Ambient Temperatures
In the presence of high ambient temperature, heat prostration (fever and heat stroke due to decreased sweating) can occur with the use of anticholinergic drugs such as glycopyrrolate oral solution. Advise patients/caregivers to avoid exposure of the patient to hot or very warm environmental temperatures.
5.4 Operating Machinery or an Automobile
Glycopyrrolate oral solution may produce drowsiness or blurred vision. As appropriate for a given age, warn the patient not to engage in activities requiring mental alertness such as operating a motor vehicle or other machinery, or performing hazardous work while taking glycopyrrolate oral solution.
5.5 Anticholinergic Drug Effects
Use glycopyrrolate oral solution with caution in patients with conditions that are exacerbated by anticholinergic drug effects including:
- Autonomic neuropathy
- Renal disease
- Ulcerative colitis – Large doses may suppress intestinal motility to the point of producing a paralytic ileus and for this reason may precipitate or aggravate “toxic megacolon”, a serious complication of the disease.
- Hyperthyroidism
- Coronary heart disease, congestive heart failure, cardiac tachyarrhythmias, tachycardia, and hypertension
- Hiatal hernia associated with reflux esophagitis, since anticholinergic drugs may aggravate this condition
6 ADVERSE REACTIONS
The following serious adverse reactions are described elsewhere in the labeling:
- Constipation or intestinal pseudo-obstruction [see Warnings and Precautions (5.1)]
- Incomplete mechanical intestinal obstruction [see Warnings and Precautions (5.2)]
The most common adverse reactions reported with glycopyrrolate oral solution are dry mouth, vomiting, constipation, flushing, and nasal congestion
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to glycopyrrolate oral solution in 151 subjects, including 20 subjects who participated in an 8-week placebo-controlled study (Study 1) and 137 subjects who participated in a 24-week open-label study (six subjects who received glycopyrrolate oral solution in the placebo-controlled study and 131 new subjects).
Table 2 presents adverse reactions reported by ≥ 15% of glycopyrrolate oral solution -treated subjects from the placebo-controlled clinical trial.
Glycopyrrolate oral solution (N=20) n (%) | Placebo (N=18) n (%) | |
Dry Mouth | 8 (40%) | 2 (11%) |
Vomiting | 8 (40%) | 2 (11%) |
Constipation | 7 (35%) | 4 (22%) |
Flushing | 6 (30%) | 3 (17%) |
Nasal Congestion | 6 (30%) | 2 (11%) |
Headache | 3 (15%) | 1 (6%) |
Sinusitis | 3 (15%) | 1 (6%) |
Upper Respiratory Tract Infection | 3 (15%) | 0 |
Urinary Retention | 3 (15%) | 0 |
The following adverse reactions occurred at a rate of <2% of patients receiving glycopyrrolate oral solution in the open-label study.
Gastrointestinal: Abdominal distention, abdominal pain, stomach discomfort, chapped lips, flatulence, retching, dry tongue General Disorders: Irritability, pain
Infections: Pneumonia, sinusitis, tracheostomy infection, upper respiratory tract infection, urinary tract infection Investigations: Heart rate increase
Metabolism and Nutrition: Dehydration
Nervous System: Headache, convulsion, dysgeusia, nystagmus
Psychiatric: Agitation, restlessness, abnormal behavior, aggression, crying, impulse control disorder, moaning, mood altered Respiratory: Increased viscosity of bronchial secretion, nasal congestion, nasal dryness
Skin: Dry skin, pruritus, rash
Vascular: Pallor
6.2 Post Marketing Experience
The following adverse reactions have been identified during postapproval use of other formulations of glycopyrrolate for other indications. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Additional adverse reactions identified during postapproval use of glycopyrrolate tablets include: loss of taste and suppression of lactation.
7 DRUG INTERACTIONS
Drugs Affected by Reduced GI Transit Time
Glycopyrrolate reduces GI transit time, which may result in altered release of certain drugs when formulated in delayed- or controlled-release dosage forms.
- The passage of potassium chloride tablets through the GI tract may be arrested or delayed with coadministration of glycopyrrolate. Solid dosage forms of potassium chloride are contraindicated [see Contraindications (4)].
- Digoxin administered as slow dissolution oral tablets may have increased serum levels and enhanced action when administered with glycopyrrolate. Monitor patients receiving slow dissolution digoxin for increased action if glycopyrrolate oral solution is coadministered regularly. Consider the use of other oral dosage forms of digoxin (e.g., elixir or capsules).
Amantadine
The anticholinergic effects of glycopyrrolate may be increased with concomitant administration of amantadine. Consider decreasing the dose of glycopyrrolate during coadministration of amantadine.
Drugs Whose Plasma Levels May be Increased by Glycopyrrolate
Coadministration of glycopyrrolate may result in increased levels of certain drugs.
- Atenolol’s bioavailability may be increased with coadministration of glycopyrrolate. A reduction in the atenolol dose may be needed.
- Metformin plasma levels may be elevated with coadministration of glycopyrrolate, increasing metformin’s pharmacologic and toxic effects. Monitor clinical response to metformin with concomitant glycopyrrolate administration; consider a dose reduction of metformin if warranted.
Drugs Whose Plasma Levels May be Decreased by Glycopyrrolate
Coadministration of glycopyrrolate may result in decreased levels of certain drugs.
- Haloperidol’s serum level may be decreased when coadministered with glycopyrrolate, resulting in worsening of schizophrenic symptoms, and development of tardive dyskinesia. Closely monitor patients if coadministration cannot be avoided.
- Levodopa’s therapeutic effect may be reduced with glycopyrrolate administration. Consider increasing the dose of levodopa.
11 DESCRIPTION
Glycopyrrolate oral solution is an anticholinergic drug available as an oral solution containing 1 mg glycopyrrolate per 5 mL. The chemical name for glycopyrrolate is pyrrolidinium, 3-[(cyclopentylhydroxyphenylacetyl) oxy]-1,1-dimethyl-bromide. The chemical structure is:
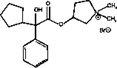
The empirical formula for glycopyrrolate oral solution is C 19 H 28 BrNO 3 and the molecular weight is 398.33. The inactive ingredients in glycopyrrolate oral solution are: citric acid, glycerin, sour cherry flavor, ethyl paraben, propylene glycol, propyl paraben, saccharin sodium, sodium citrate, sorbitol solution, and purified water
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Glycopyrrolate is a competitive inhibitor of acetylcholine receptors that are located on certain peripheral tissues, including salivary glands. Glycopyrrolate indirectly reduces the rate of salivation by preventing the stimulation of these receptors.
12.2 Pharmacodynamics
Glycopyrrolate inhibits the action of acetylcholine on salivary glands thereby reducing the extent of salivation.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
When glycopyrrolate was administered via oral gavage to mice for up to 24 months at dosages of 2.5, 7, and 20 mg/kg/day in both genders, resulting in systemic exposures (estimated AUC 0-inf values) approximately 0.1, 0.3, and 0.8 times, respectively, the estimated systemic exposure in humans at the MRHD (9 mg per day, administered in three divided doses), no significant changes in tumor incidence were observed when compared to control.
When glycopyrrolate was administered via oral gavage to rats for up to 24 months at dosages of 5, 15, and 40 mg/kg/day in both genders, resulting in systemic exposures approximately 0.2, 0.8, and 2 times, respectively, the estimated systemic exposure in humans at the MRHD, no significant changes in tumor incidence were observed when compared to control.
Glycopyrrolate did not elicit any genotoxic effects in the Ames mutagenicity assay, the human lymphocyte chromosome aberration assay, or the micronucleus assay.
Glycopyrrolate was assessed for effects on fertility or general reproductive function in rats. Rats of both genders received glycopyrrolate at dosages up to 100 mg/kg/day via oral gavage, resulting in systemic exposures (estimated AUC0-inf values) in males and females up to approximately 11 and 15 times, respectively, the estimated systemic exposure in humans at the MRHD. No treatment-related effects on fertility or reproductive parameters were observed in either gender in this study.
14 CLINICAL STUDIES
Glycopyrrolate oral solution was evaluated in a multi-center, randomized, double-blind, placebo-controlled, parallel, eight-week study for the control of pathologic drooling in children (Study 1). The study enrolled 38 subjects aged 3-23 years; thirty-six subjects were aged 3-16 years and two patients were greater than 16 years. The subjects were male or female, weighed at least 13 kg (27 lbs), and had cerebral palsy, mental retardation, or another neurologic condition associated with problem drooling defined as drooling in the absence of treatment so that clothing became damp on most days (approximately five to seven days per week). Subjects were randomized in a 1:1 fashion to receive glycopyrrolate oral solution or placebo. Doses of study medication were titrated over a 4-week period to optimal response beginning at 0.02 mg/kg three times a day increasing doses in increments of approximately 0.02 mg/kg three times per day every 5-7 days, not to exceed the lesser of approximately 0.1 mg/kg three times per day or 3 mg three times per day.
Subjects were evaluated on the 9-point modified Teacher’s Drooling Scale (mTDS), which is presented below. The mTDS evaluations were recorded by parents/caregivers 3 times daily approximately two hours post-dose on evaluation days during pre-treatment baseline and at Weeks 2, 4, 6 and 8 of therapy.
Modified Teacher’s Drooling Scale
1= Dry: never drools
2= Mild: only the lips are wet; occasionally
3= Mild: only the lips are wet; frequently
4= Moderate: wet on the lips and chin; occasionally 5= Moderate: wet on the lips and chin; frequently
6= Severe: drools to the extent that clothing becomes damp; occasionally
7= Severe: drools to the extent that clothing becomes damp; frequently
8= Profuse: clothing, hands, tray, and objects become wet; occasionally
9= Profuse: clothing, hands, tray, and objects become wet; frequently
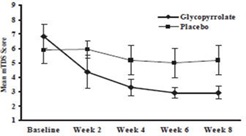
Responders were defined as subjects with at least a 3-point reduction in mean daily m TDS scores from baseline to Week 8. Table 4 presents the proportion of responders at Week 8 and Figure 1 presents the mean m TDS values from baseline through Week 8.
Glycopyrrolate Oral Solution Group (N=20) | Placebo Group (N=18) |
15/20 (75%) | 2/18 (11%) |
Figure 1. Mean (± 2 Standard Errors) mDTS Scores
16 HOW SUPPLIED/STORAGE AND HANDLING
NDC 68022-0156-1; 1 mg/5mL clear, cherry-flavored solution; 16 oz. bottle.
Store at room temperature 20° - 25°C (68° - 77°F); excursions permitted to 15° - 30°C (59° - 86°F) [See USP Controlled Room Temperature].
12.1 Mechanism of Action
Glycopyrrolate is a competitive inhibitor of acetylcholine receptors that are located on certain peripheral tissues, including salivary glands. Glycopyrrolate indirectly reduces the rate of salivation by preventing the stimulation of these receptors.