Get your patient on Glucagon - Glucagon (Glucagon)
Glucagon - Glucagon prescribing information
INDICATIONS AND USAGE
Glucagon for Injection is an antihypoglycemic agent and a gastrointestinal motility inhibitor indicated:
Severe Hypoglycemia
Glucagon for Injection is indicated for the treatment of severe hypoglycemia in pediatric and adult patients with diabetes mellitus.
Diagnostic Aid
Glucagon for Injection is indicated as a diagnostic aid for use during radiologic examinations to temporarily inhibit movement of the gastrointestinal tract in adult patients.
DOSAGE AND ADMINISTRATION
Dosage in adult and pediatric patients to treat severe hypoglycemia (2.2 )
- Adults and Pediatric Patients Weighing 20 kg or More:
- The recommended dosage is 1 mg (1 mL) injected subcutaneously or intramuscularly into the upper arm, thigh, or buttocks, or intravenously.
- If there has been no response after 15 minutes, an additional 1 mg dose (1 mL) may be administered while waiting for emergency assistance.
- Pediatric Patients Weighing Less Than 20 kg:
- The recommended dosage is 0.5 mg (0.5 mL) or dose equivalent to 20 mcg/kg to 30 mcg/kg injected subcutaneously or intramuscularly into the upper arm, thigh, or buttocks, or intravenously.
- If there has been no response after 15 minutes, an additional 0.5 mg dose (0.5 mL) may be administered while waiting for emergency assistance.
Important Administration Instructions for Using Glucagon for Injection to Treat Severe Hypoglycemia (2.1 )
- Glucagon for Injection is for subcutaneous, intramuscular, or intravenous injection. Administer intravenously ONLY under medical supervision.
- See the Full Prescribing Information for administration instructions
Dosage in Adults for Using Glucagon for Injection as a Diagnostic Aid (2.4 )
- Doses required for relaxation of the stomach, duodenum, and small bowel, depend on the onset and duration of effect required for the examination.
- The dose for relaxation of the duodenum and small bowel is 0.25 mg to 0.5 mg given intravenously or 1 mg given intramuscularly.
- For the stomach 0.5 mg intravenous or 2 mg intramuscular doses are recommended.
- For the colon, it is recommended that a 2 mg dose be administered intramuscularly approximately 10 minutes prior to the procedure.
- See the Full Prescribing Information for administration instructions (2.3 )
Important Administration Instructions for Using Glucagon for Injection to Treat Severe Hypoglycemia
Glucagon for Injection is for subcutaneous, intramuscular, or intravenous injection. Administer intravenously ONLY under medical supervision.
Instruct patients and their caregivers on the signs and symptoms of severe hypoglycemia. Because severe hypoglycemia requires the help of others to recover, instruct the patient to inform those around them about Glucagon for Injection and its Instructions for Use. Administer Glucagon for Injection as soon as possible when severe hypoglycemia is recognized.
Instruct the patient or caregiver to read the Instructions for Use at the time they receive a prescription for Glucagon for Injection. Emphasize the following instructions to the patient or caregiver:
- Using the supplied prefilled syringe, carefully insert the needle through the rubber stopper of the vial containing Glucagon for Injection powder and inject all the liquid from the syringe into the vial.
- Swirl the vial gently until the powder is completely dissolved and no particles remain in the fluid. The reconstituted solution should be clear and of a water-like consistency at time of use. Inspect visually for particulate matter and discoloration. If the resulting solution is cloudy or contains particulate matter do not use.
- The reconstituted solution is 1 mg per mL glucagon. Use immediately after reconstitution.
- Immediately after reconstitution, use the same syringe to withdraw the correct dose of Glucagon for Injection.
- Inject the solution subcutaneously or intramuscularly in the upper arm, thigh, or buttocks. In addition, healthcare providers may administer intravenously.
- Call for emergency assistance immediately after administering the dose.
- If there has been no response after 15 minutes, an additional dose of Glucagon for Injection may be administered while waiting for emergency assistance.
- When the patient has responded to the treatment and is able to swallow, give oral carbohydrates to restore the liver glycogen and prevent recurrence of hypoglycemia.
- Discard any unused portion.
Dosage for Treatment of Severe Hypoglycemia
Adults and Pediatric Patients Weighing 20 kg or More
- The recommended dosage is 1 mg (1 mL) injected subcutaneously or intramuscularly into the upper arm, thigh, or buttocks. Alternatively, healthcare providers may administer the dose intravenously.
- If there has been no response after 15 minutes, an additional 1 mg dose (1 mL) of Glucagon for Injection may be administered while waiting for emergency assistance.
Pediatric Patients Weighing Less Than 20 kg
- The recommended dosage is 0.5 mg (0.5 mL) or dose equivalent to 20 mcg/kg to 30 mcg/kg injected subcutaneously or intramuscularly into the upper arm, thigh, or buttocks. Alternatively, healthcare providers may administer the dose intravenously.
- If there has been no response after 15 minutes, an additional 0.5 mg dose (0.5 mL) of Glucagon for Injection may be administered while waiting for emergency assistance.
Important Administration Instructions for Using Glucagon for Injection as a Diagnostic Aid
- Reconstitute Glucagon for Injection with 1 mL of diluent.
- Swirl the vial gently until the powder is completely dissolved and no particles remain in the fluid. The reconstituted solution should be clear and of a water-like consistency at time of use. Inspect visually for particulate matter and discoloration. If the resulting solution is cloudy or contains particulate matter do not use.
- Withdraw the correct dose of Glucagon for Injection.
- The reconstituted solution is 1 mg per mL glucagon.
- Immediately after reconstitution, inject the solution intravenously or intramuscularly into upper arm, thigh, or buttocks.
- Discard unused portion.
- After the end of the diagnostic procedure, give oral carbohydrates to patients who have been fasting, if this is compatible with the diagnostic procedure.
Dosage in Adults for Use as a Diagnostic Aid
- Doses required for relaxation of the stomach, duodenum, and small bowel, depend on the onset and duration of effect required for the examination [see Pharmacodynamics (12.2) ] .
- The usual dose for relaxation of the stomach, duodenum and small bowel is 0.25 mg to 0.5 mg given intravenously or 1 mg given intramuscularly, but up to 2 mg intravenously or intramuscularly may be used if required (2 mg doses produce a higher rate of nausea and vomiting than lower doses) [see Adverse Reactions (6) ] .
- For the stomach because it is less sensitive to the effect of glucagon, 0.5 mg intravenous or 2 mg intramuscular doses are recommended.
- For the examination of the colon, it is recommended that a 2 mg dose be administered intramuscularly approximately 10 minutes prior to the procedure.
DOSAGE FORMS AND STRENGTHS
Glucagon for Injection, USP is a white to off white lyophilized powder or plug supplied as follows:
- Single-dose vial containing 1 mg Glucagon for Injection, USP with a pre-filled syringe containing 1 mL of diluent for Glucagon.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
Available data from case reports and a small number of observational studies with glucagon use in pregnant women over decades of use have not identified a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. Multiple small studies have demonstrated a lack of transfer of pancreatic glucagon across the human placental barrier during early gestation. In a rat reproduction study, no embryofetal toxicity was observed with glucagon administered by injection during the period of organogenesis at doses representing up to 40 times the human dose, based on body surface area (mg/m 2 ) (see Data ).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
In pregnant rats given animal sourced glucagon twice-daily by injection at doses up to 2 mg/kg (up to 40 times the human dose based on body surface area extrapolation, mg/m 2 ) during the period of organogenesis, there was no evidence of increased malformations or embryofetal lethality.
Lactation
Risk Summary
There is no information available on the presence of glucagon in human or animal milk, the effects of glucagon on the breastfed child or the effects of glucagon on milk production. However, glucagon is a peptide and would be expected to be broken down to its constituent amino acids in the infant's digestive tract and is therefore, unlikely to cause harm to an exposed infant.
Pediatric Use
The safety and effectiveness of Glucagon for Injection for the treatment of severe hypoglycemia in pediatric patients with diabetes have been established.
Safety and effectiveness for use as a diagnostic aid during radiologic examinations to temporarily inhibit movement of the gastrointestinal tract in pediatric patients have not been established.
Geriatric Use
Clinical studies of glucagon did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient when used as a diagnostic aid should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
CONTRAINDICATIONS
Glucagon for Injection is contraindicated in patients with:
- Pheochromocytoma because of the risk of substantial increase in blood pressure [see Warning and Precautions (5.1) ]
- Insulinoma because of the risk of hypoglycemia [see Warning and Precautions (5.2) ]
- Known hypersensitivity to glucagon or any of the excipients in Glucagon for Injection. Allergic reactions have been reported with glucagon and include anaphylactic shock with breathing difficulties and hypotension [see Warning and Precautions (5.3) ]
- Glucagonoma when used as a diagnostic aid because of the risk of hypoglycemia [see Warnings and Precautions (5.8) ]
WARNINGS AND PRECAUTIONS
- Substantial Increase in Blood Pressure in Patients with Pheochromocytoma : Contraindicated in patients with pheochromocytoma because Glucagon for Injection may stimulate the release of catecholamines from the tumor. (4 , 5.1 )
- Hypoglycemia in Patients with Insulinoma : In patients with insulinoma, glucagon administration may produce an initial increase in blood glucose; however, Glucagon for Injection may stimulate exaggerated insulin release from an insulinoma and cause hypoglycemia. If a patient develops symptoms of hypoglycemia after a dose of Glucagon for Injection, give glucose orally or intravenously. (4 , 5.2 )
- Hypersensitivity and Allergic Reactions : Allergic reactions, which have been reported with glucagon, may occur and include generalized rash, and in some cases anaphylactic shock with breathing difficulties, and hypotension. (4 , 5.3 )
- Lack of Efficacy in Patients with Decreased Glycogen: Glucagon for Injection is effective in treating hypoglycemia only if sufficient hepatic glycogen is present. Patients in states of starvation, with adrenal insufficiency or chronic hypoglycemia may not have adequate levels of hepatic glycogen for Glucagon for Injection administration to be effective. Patients with these conditions should be treated with glucose. (5.4 )
- Necrolytic Migratory Erythema (NME): Necrolytic migratory erythema (NME), a skin rash, have been reported post marketing following continuous glucagon infusion and resolved with discontinuation of the glucagon. Should NME occur, consider whether the benefits of continuous glucagon infusion outweigh the risks. (5.5 )
- Hyperglycemia in Patients with Diabetes Mellitus when Used as a Diagnostic Aid : Treatment with Glucagon for Injection in patients with diabetes mellitus may cause hyperglycemia. Monitor diabetic patients for changes in blood glucose levels during treatment and treat if indicated. (5.6 )
- Blood Pressure and Heart Rate Increase in Patients with Cardiac Disease when Used as a Diagnostic Aid : Glucagon for Injection may increase myocardial oxygen demand, blood pressure, and pulse rate. Cardiac monitoring is recommended in patients with cardiac disease during use of Glucagon for Injection as a diagnostic aid, and an increase in blood pressure and pulse rate may require therapy. (5.7 )
- Hypoglycemia in Patients with Glucagonoma: Glucagon administered to patients with glucagonoma may cause secondary hypoglycemia. Test patients suspected of having glucagonoma for blood levels of glucagon prior to treatment. (4 , 5.8 )
Substantial Increase in Blood Pressure in Patients with Pheochromocytoma
Glucagon for Injection is contraindicated in patients with pheochromocytoma because glucagon may stimulate the release of catecholamines from the tumor [see Contraindications (4) ]. If the patient develops a substantial increase in blood pressure and a previously undiagnosed pheochromocytoma is suspected, 5 to 10 mg of phentolamine mesylate, administered intravenously, has been shown to be effective in lowering blood pressure.
Hypoglycemia in Patients with Insulinoma
In patients with insulinoma, administration of glucagon may produce an initial increase in blood glucose; however, Glucagon for Injection administration may directly or indirectly (through an initial rise in blood glucose) stimulate exaggerated insulin release from an insulinoma and cause hypoglycemia. Glucagon for Injection is contraindicated in patients with insulinoma [see Contraindications (4) ]. If a patient develops symptoms of hypoglycemia after a dose of Glucagon for Injection, give glucose orally or intravenously.
Hypersensitivity and Allergic Reactions
Allergic reactions have been reported with glucagon, these include generalized rash, and in some cases anaphylactic shock with breathing difficulties and hypotension. Glucagon for Injection is contraindicated in patients with a prior hypersensitivity reaction [see Contraindications (4) ] .
Lack of Efficacy in Patients with Decreased Hepatic Glycogen
Glucagon for Injection is effective in treating hypoglycemia only if sufficient hepatic glycogen is present. Patients in states of starvation, with adrenal insufficiency or chronic hypoglycemia may not have adequate levels of hepatic glycogen for Glucagon for Injection administration to be effective. Patients with these conditions should be treated with glucose.
Necrolytic Migratory Erythema
Necrolytic Migratory Erythema (NME), a skin rash commonly associated with glucagonomas (glucagon-producing tumors) and characterized by scaly, pruritic erythematous plaques, bullae, and erosions, has been reported post marketing following continuous glucagon infusion. NME lesions may affect the face, groin, perineum and legs or be more widespread. In the reported cases, NME resolved with discontinuation of the glucagon, and treatment with corticosteroids was not effective. Should NME occur, consider whether the benefits of continuous glucagon infusion outweigh the risks.
Hyperglycemia in Patients with Diabetes Mellitus when Used as a Diagnostic Aid
Treatment with Glucagon for Injection in patients with diabetes mellitus may cause hyperglycemia. Monitor diabetic patients for changes in blood glucose levels during treatment and treat if indicated.
Blood Pressure and Heart Rate Increase in Patients with Cardiac Disease when Used as a Diagnostic Aid
Glucagon for Injection may increase myocardial oxygen demand, blood pressure, and pulse rate which may be life-threatening in patients with cardiac disease. Cardiac monitoring is recommended in patients with cardiac disease during use of Glucagon for Injection as a diagnostic aid, and an increase in blood pressure and pulse rate may require therapy.
Hypoglycemia in Patients with Glucagonoma
Glucagon administered to patients with glucagonoma may cause secondary hypoglycemia. Test patients suspected of having glucagonoma for blood levels of glucagon prior to use as a diagnostic aid as Glucagon for Injection is contraindicated in this setting [see Contraindications (4) ].
ADVERSE REACTIONS
The following important adverse reactions are described below and elsewhere in the labeling:
- Hypersensitivity and Allergic Reactions [see Warnings and Precautions (5.3) ]
- Necrolytic Migratory Erythema [see Warnings and Precautions (5.5) ]
- Hyperglycemia in Patients with Diabetes Mellitus when Used as a Diagnostic Aid [see Warnings and Precautions (5.6) ]
- Blood Pressure and Heart Rate Increase in Patients with Cardiac Disease when used as a Diagnostic Aid [see Warnings and Precautions (5.7) ]
The following adverse reactions have been identified during post-approval use of glucagon. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Injection site reactions including erythema and swelling
- Nausea
- Vomiting
- Headache
- Dizziness
- Asthenia
- Pallor
- Diarrhea
- Somnolence
- Generalized allergic reactions including anaphylactic shock with breathing difficulties and hypotension
- Hypertension and tachycardia
- Decreased blood pressure. Hypotension has been reported up to 2 hours after administration in patients receiving glucagon as premedication for upper gastrointestinal endoscopy procedures.
- Hypoglycemia and hypoglycemic coma. Patients taking indomethacin may be more likely to experience hypoglycemia following glucagon administration [see Drug Interactions (7) ] .
- Necrolytic Migratory Erythema (NME) cases have been reported post marketing in patients receiving continuous infusion of glucagon.
DRUG INTERACTIONS
Beta-Blockers | |
Clinical Impact: | Patients taking beta-blockers may have a transient increase in pulse and blood pressure when given Glucagon for Injection. |
Intervention: | The increase in blood pressure and heart rate may require therapy in patients with coronary artery disease. |
Indomethacin | |
Clinical Impact: | In patients taking indomethacin, Glucagon for Injection may lose its ability to raise blood glucose or may even produce hypoglycemia. |
Intervention: | Monitor blood glucose levels during glucagon treatment of patients taking indomethacin. |
Anticholinergic Drugs | |
Clinical Impact: | The concomitant use of anticholinergic drugs and Glucagon for Injection increase the risk of gastrointestinal adverse reactions due to additive effects on inhibition of gastrointestinal motility. |
Intervention: | Concomitant use of anticholinergic drugs with Glucagon for Injection for use as a diagnostic aid is not recommended. |
Warfarin | |
Clinical Impact: | Glucagon for Injection may increase the anticoagulant effect of warfarin. |
Intervention: | Monitor patients for unusual bruising or bleeding, as adjustments in warfarin dosage may be required. |
Insulin | |
Clinical Impact: | Insulin acts antagonistically to glucagon. |
Intervention: | Monitor blood glucose when Glucagon for Injection is used as a diagnostic aid in patients receiving insulin. |
DESCRIPTION
Glucagon is an antihypoglycemic agent and a gastrointestinal motility inhibitor. Glucagon is produced by solid state peptide synthesis. The chemical structure of the glucagon is identical to human glucagon. Glucagon is a single-chain polypeptide that contains 29 amino acid residues and has a molecular weight of 3482.8 Daltons.
The empirical formula is C 153 H 225 N 43 O 49 S. The primary sequence of glucagon is shown below.
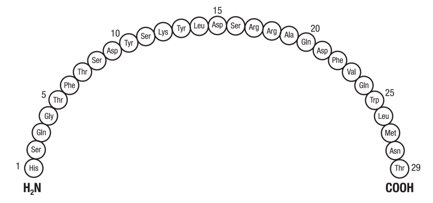
Glucagon (synthetic origin) is a white to off-white powder. It is relatively insoluble in water but is soluble at a pH of less than 3 or more than 9.5.
Glucagon for injection, USP is a sterile, white to off white lyophilized powder or plug in a vial and a syringe of sterile diluent for intravenous, intramuscular, or subcutaneous use. The vial contains 1 mg of glucagon USP, 49 mg of lactose monohydrate. Hydrochloric acid may have been added during manufacture to adjust the pH of the glucagon to 2.5 to 3.0. One International Unit of glucagon is equivalent to 1 mg of glucagon. The 1 mL diluent syringe contains 12 mg of glycerin, 1 mg hydrochloric acid, and Water for Injection, at pH 1.8 to 2.2.
CLINICAL PHARMACOLOGY
Mechanism of Action
Glucagon increases blood glucose concentration by activating hepatic glucagon receptors, thereby stimulating glycogen breakdown and release of glucose from the liver. Hepatic stores of glycogen are necessary for glucagon to produce an antihypoglycemic effect. Extrahepatic effects of glucagon include relaxation of the smooth muscle of the stomach, duodenum, small bowel, and colon.
Pharmacodynamics
Treatment of Severe Hypoglycemia
Blood glucose concentrations rise within 10 minutes of subcutaneous injection and mean maximal concentration of 136 mg/dL is attained 30 minutes after injection (see Figure 1). Similarly, following intramuscular injection, the mean peak glucose level was 138 mg/dL, which occurred at 26 minutes after injection.
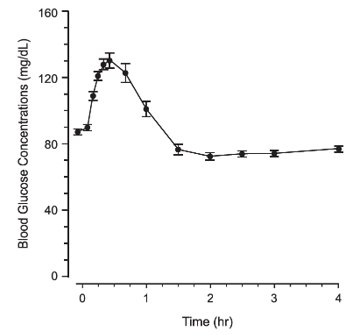
Diagnostic Aid
Table 2 displays the dose and pharmacodynamics properties of Glucagon for Injection as a diagnostic aid during radiological examination.
Dose | Route of administration | Time of onset of action | Approximate duration of effect |
0.25 to 0.5 mg | Intravenous | 1 minute | 9 to 17 minutes |
1 mg | Intramuscular | 8 to 10 minutes | 12 to 27 minutes |
2 mg •Administration of 2 mg doses produces a higher incidence of nausea and vomiting than do lower doses. | Intravenous | 1 minute | 22 to 25 minutes |
2 mg | Intramuscular | 4 to 7 minutes | 21 to 32 minutes |
Pharmacokinetics
Absorption
Glucagon injected through the intramuscular route achieved mean peak plasma levels of 6.9 ng/mL at approximately 13 minutes after dosing; and 7.9 ng/mL at approximately 20 minutes after subcutaneous dosing. Administration of the intravenous glucagon showed dose proportionality of the pharmacokinetics between 0.25 and 2 mg.
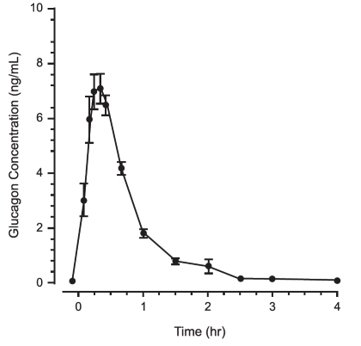
Distribution
The mean volume of distribution was 0.25 L/kg.
Elimination
The mean clearance was 13.5 mL/min/kg. The mean half-life ranged from 8 to 18 minutes.
Metabolism
Glucagon is degraded in the liver, kidney and plasma.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long term studies in animals to evaluate carcinogenic potential have not been performed. Recombinant glucagon was positive in the bacterial Ames assay. It was determined that an increase in colony counts was related to technical difficulties in running this assay with peptides. Studies in rats have shown that glucagon does not cause impaired fertility.
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Glucagon Emergency Kit for Low Blood Sugar contains 1 mg Glucagon for Injection, USP (a sterile, lyophilized white to off-white powder or plug) in a single-dose vial and 1 prefilled syringe containing 1 mL of diluent of Glucagon.
- 1 mg per vial - with 1 mL of diluting solution (PFS) (1s) NDC of Pre-filled Syringe: 0378-8066-32; NDC of Vial Label: 0378-8065-32; NDC of Carton: 0378-8067-90.
Storage and Handling
Before Reconstitution:
Store Glucagon for Injection, USP at controlled room temperature 20° to 25°C (68° to 77°F) [See USP]. Do not use Glucagon for Injection, USP if the expiration date has passed. Do not freeze. Keep in its original package and away from light.
After Reconstitution:
Use reconstituted Glucagon for Injection, USP immediately. Discard unused portion [see Dosage and Administration (2.3) ].
INSTRUCTIONS FOR USE
Glucagon (GLOO-ka-gon) for Injection, USP
Important
- Read and become familiar with this Instructions for Use before an emergency happens.
- Show your family members and others where you keep your Glucagon Emergency Kit for Low Blood Sugar and how to use it the right way.
- Call for emergency medical help right after you use Glucagon for Injection.
- Do not share your Glucagon syringes or needles with another person. You may give other people a serious infection or get a serious infection from them.
- The prefilled syringe that comes with your Glucagon Emergency Kit for Low Blood Sugar is for use with Glucagon for Injection only. Do not use Glucagon for Injection syringes to inject other medicines.
How should I store Glucagon for Injection?
Before you mix the Glucagon powder and liquid:
- Store Glucagon at room temperature between 68° to 77°F (20° to 25°C).
- Check the expiration date on the vial of Glucagon for Injection. Do not use Glucagon for Injection if the expiration date has passed.
- Do not freeze Glucagon.
- Keep Glucagon in its original package and away from light.
After you mix the Glucagon powder and liquid:
- Use Glucagon for Injection right away.
- Throw away unused Glucagon for Injection.
Supplies you will need for your Glucagon for Injection
- Glucagon Emergency Kit for Low Blood Sugar contains (see Figure A):
- 1 single-dose vial containing 1 mg Glucagon for Injection (as a sterile, white to off-white lyophilized powder or plug)
- 1 prefilled syringe containing 1 mL of diluent for Glucagon
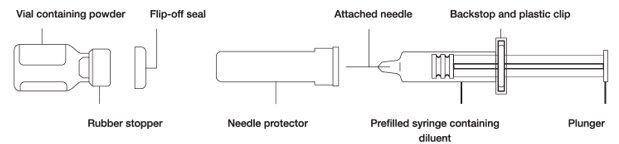
- 1 puncture resistant container for throwing away used needles and syringes. See “How should I dispose of (throw away) used Glucagon syringes?” at the end of these instructions.
- Alcohol swab (not included in kit)
Preparing to Inject Glucagon for Injection
- The Glucagon medicine comes as a dry powder. Before you use Glucagon for Injection, you must mix the dry powder with the syringe of diluent that comes in the Glucagon Emergency Kit for Low Blood Sugar. Do not use any other liquid to mix the medicine.
Step 1: Remove the flip-off seal from the vial of Glucagon. Wipe the rubber stopper on the vial with an alcohol swab (see Figure B).
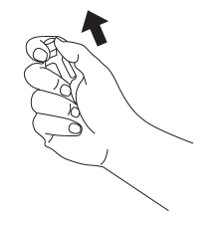
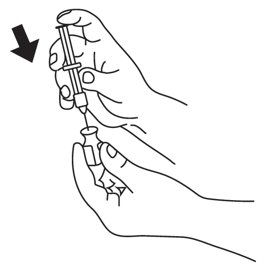
Step 2: Remove the needle protector from the syringe. Inject all of the diluent in the prefilled syringe into the vial containing Glucagon powder (see Figure C). Do not remove the plastic clip from the syringe. Remove syringe from the vial.
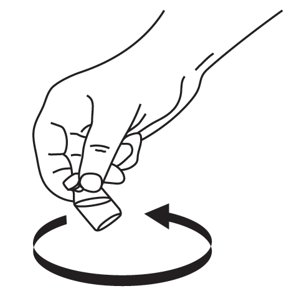
Step 3: Swirl the vial gently until the Glucagon powder dissolves completely (see Figure D). Glucagon should not be used unless the solution is clear and of a water-like consistency.
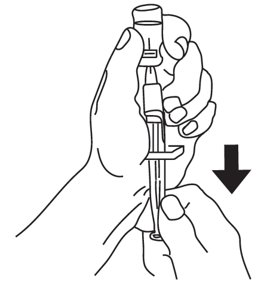
Step 4: Using the same syringe, hold the vial upside down and, making sure the needle tip remains in the solution, gently withdraw all the solution (1 mg mark on syringe) from the vial (see Figure E). The backstop and plastic clip on the syringe will prevent the rubber stopper from being pulled out of the syringe; however, if the plunger rod separates from the rubber stopper, simply reinsert the rod by turning it clockwise. The usual adult dose is 1 mg. For children weighing less than 44 lb (20 kg), give 1/2 adult dose (0.5 mg). For these children, withdraw 1/2 of the solution from the vial (0.5 mg mark on syringe).
Injecting Glucagon for Injection
Step 5: Choose your injection site and clean with an alcohol wipe. Injection sites for Glucagon for Injection are the buttocks, upper arm, or thigh.
Step 6: Insert the needle into the loose tissue under the clean injection site and push the plunger to inject all (or 1/2 for children weighing less than 44 lbs.) of the Glucagon for Injection.
After injecting Glucagon for Injection
Step 7: Apply light pressure at the injection site and withdraw the needle. Press an alcohol swab against the injection site.
Throw away your used syringe with the needle attached and any Glucagon that you did not use in an FDA-cleared sharps disposal container right away after use.
See “How should I dispose of (throw away) used Glucagon syringes?” at the end of these instructions.
Step 8: Turn the person on their side. When an unconscious person wakes up, they may vomit. Turning the person on their side will prevent them from choking.
Step 9: Call for emergency medical help right away.
Step 10 : Feed the person as soon as they wake up and are able to swallow . Give the person a fast-acting source of sugar (such as a regular soft drink or fruit juice) and a long-acting source of sugar (such as crackers and cheese or a meat sandwich).
Step 11: If the person does not awaken within 15 minutes, give another dose of Glucagon and inform a doctor or emergency services right away .
Step 12: Even if the Glucagon revives the person, their doctor should be notified right away. A doctor should be notified whenever severe hypoglycemia occurs.
Hypoglycemia may happen again after receiving Glucagon for Injection.
Early symptoms of hypoglycemia (low blood glucose) include:
|
|
If not treated early, the symptoms may progress to severe hypoglycemia. Signs include:
|
|
How should I dispose of (throw away) used Glucagon syringes?
- Put used syringes in an FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) loose needles and syringes in your household trash.
- If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak-resistant, and
- properly labelled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal .
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
Keep Glucagon for Injection and all medicines out of the reach of children.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured for: Mylan Pharmaceuticals Inc. Morgantown, WV 26505 U.S.A
Manufactured by: Mylan Laboratories Limited Bangalore, India
DECEMBER 2024
Mechanism of Action
Glucagon increases blood glucose concentration by activating hepatic glucagon receptors, thereby stimulating glycogen breakdown and release of glucose from the liver. Hepatic stores of glycogen are necessary for glucagon to produce an antihypoglycemic effect. Extrahepatic effects of glucagon include relaxation of the smooth muscle of the stomach, duodenum, small bowel, and colon.