Get your patient on Furosemide - Furosemide tablet (Furosemide)
Furosemide - Furosemide tablet prescribing information
INDICATIONS AND USAGE
Furosemide tablets are indicated in adults and pediatric patients for the treatment of edema associated with congestive heart failure, cirrhosis of the liver, and renal disease, including the nephrotic syndrome. Furosemide is particularly useful when an agent with greater diuretic potential is desired.
Furosemide tablets may be used in adults for the treatment of hypertension alone or in combination with other antihypertensive agents. Hypertensive patients who cannot be adequately controlled with thiazides will probably also not be adequately controlled with furosemide alone.
DOSAGE AND ADMINISTRATION
Therapy should be individualized according to patient response to gain maximal therapeutic response and to determine the minimal dose needed to maintain that response.
Adults -- The usual initial dose of furosemide tablets is 20 mg to 80 mg given as a single dose. Ordinarily a prompt diuresis ensues. If needed, the same dose can be administered 6 to 8 hours later or the dose may be increased. The dose may be raised by 20 mg or 40 mg and given not sooner than 6 to 8 hours after the previous dose until the desired diuretic effect has been obtained. The individually determined single dose should then be given once or twice daily (e.g., at 8 am and 2 pm). The dose of furosemide tablets may be carefully titrated up to 600 mg/day in patients with clinically severe edematous states.
Edema may be most efficiently and safely mobilized by giving furosemide tablets on 2 to 4 consecutive days each week.
When doses exceeding 80 mg/day are given for prolonged periods, careful clinical observation and laboratory monitoring are particularly advisable (see PRECAUTIONS: Laboratory Tests ).
Geriatric patients -- In general, dose selection for the elderly patient should be cautious, usually starting at the low end of the dosing range (see PRECAUTIONS: Geriatric Use ).
Pediatric patients -- The usual initial dose of furosemide tablets in pediatric patients is 2 mg/kg body weight, given as a single dose. If the diuretic response is not satisfactory after the initial dose, dosage may be increased by 1 or 2 mg/kg no sooner than 6 to 8 hours after the previous dose. Doses greater than 6 mg/kg body weight are not recommended. For maintenance therapy in pediatric patients, the dose should be adjusted to the minimum effective level.
Therapy should be individualized according to the patient's response to gain maximal therapeutic response and to determine the minimal dose needed to maintain the therapeutic response.
Adults -- The usual initial dose of furosemide tablets for hypertension is 80 mg, usually divided into 40 mg twice a day. Dosage should then be adjusted according to response. If response is not satisfactory, add other antihypertensive agents.
Changes in blood pressure must be carefully monitored when furosemide tablets are used with other antihypertensive drugs, especially during initial therapy. To prevent excessive drop in blood pressure, the dosage of other agents should be reduced by at least 50% when furosemide tablets are added to the regimen. As the blood pressure falls under the potentiating effect of furosemide tablets, a further reduction in dosage or even discontinuation of other antihypertensive drugs may be necessary.
Geriatric patients -- In general, dose selection and dose adjustment for the elderly patient should be cautious, usually starting at the low end of the dosing range (see PRECAUTIONS: Geriatric Use ).
CONTRAINDICATIONS
Furosemide tablets are contraindicated in patients with anuria and in patients with a history of hypersensitivity to furosemide.
ADVERSE REACTIONS
Adverse reactions are categorized below by organ system and listed by decreasing severity.
Gastrointestinal System Reactions
- hepatic encephalopathy in patients with hepatocellular insufficiency
- pancreatitis
- jaundice (intrahepatic cholestatic jaundice)
- increased liver enzymes
- anorexia
- oral and gastric irritation
- cramping
- diarrhea
- constipation
- nausea
- vomiting
Systemic Hypersensitivity Reactions
- Severe anaphylactic or anaphylactoid reactions (e.g., with shock)
- systemic vasculitis
- interstitial nephritis
- necrotizing angiitis
Central Nervous System Reactions
- tinnitus and hearing loss
- paresthesias
- vertigo
- dizziness
- headache
- blurred vision
- xanthopsia
Hematologic Reactions
- aplastic anemia
- thrombocytopenia
- agranulocytosis
- hemolytic anemia
- leukopenia
- anemia
- eosinophilia
Dermatologic-Hypersensitivity Reactions
- toxic epidermal necrolysis
- Stevens-Johnson Syndrome
- erythema multiforme
- drug rash with eosinophilia and systemic symptoms
- acute generalized exanthematous pustulosis
- exfoliative dermatitis
- bullous pemphigoid
- purpura
- photosensitivity
- rash
- pruritis
- urticaria
Cardiovascular Reaction
- Orthostatic hypotension may occur and be aggravated by alcohol, barbiturates, or narcotics.
- Increase in cholesterol and triglyceride serum levels
Other Reactions
- hyperglycemia
- glycosuria
- hyperuricemia
- muscle spasm
- weakness
- restlessness
- urinary bladder spasm
- thrombophlebitis
- fever
Whenever adverse reactions are moderate or severe, furosemide dosage should be reduced or therapy withdrawn.
To report SUSPECTED ADVERSE REACTIONS, contact Ipca at 1-888-472-2651 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
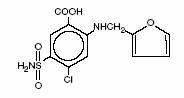
Furosemide is a diuretic which is an anthranilic acid derivative. Furosemide tablets, USP for oral administration contain furosemide, USP as the active ingredient and the following inactive ingredients: corn starch, lactose monohydrate, magnesium stearate, pregelatinized starch, and sodium starch glycolate. Chemically, it is 4-chloro-N-furfuryl-5-sulfamoylanthranilic acid. Furosemide , USP is available as white to off white round tablets for oral administration in dosage strengths of 20, 40 and 80 mg. Furosemide, USP is a white to slightly yellow odorless crystalline powder. It is practically insoluble in water, soluble in 15 parts of acetone, freely soluble in dimethylformamide and in solution of alkali hydroxides; soluble in methanol; sparingly soluble in alcohol; very slightly soluble in chloroform.
The CAS Registry Number is 54-31-9.
It has a molecular formula of C 12 H 11 ClN 2 O 5 S and a molecular weight of 330.75.
The molecular structure is as follows:
Tested by USP Dissolution Test 1
CLINICAL PHARMACOLOGY
Investigations into the mode of action of furosemide have utilized micropuncture studies in rats, stop flow experiments in dogs and various clearance studies in both humans and experimental animals. It has been demonstrated that furosemide inhibits primarily the absorption of sodium and chloride not only in the proximal and distal tubules but also in the loop of Henle. The high degree of efficacy is largely due to the unique site of action. The action on the distal tubule is independent of any inhibitory effect on carbonic anhydrase and aldosterone.
Recent evidence suggests that furosemide glucuronide is the only or at least the major biotransformation product of furosemide in man. Furosemide is extensively bound to plasma proteins, mainly to albumin. Plasma concentrations ranging from 1 mcg/mL to 400 mcg/mL are 91% to 99% bound in healthy individuals. The unbound fraction averages 2.3% to 4.1% at therapeutic concentrations.
The onset of diuresis following oral administration is within 1 hour. The peak effect occurs within the first or second hour. The duration of diuretic effect is 6 to 8 hours.
In fasted normal men, the mean bioavailability of furosemide from furosemide tablets and furosemide oral solution is 64% and 60%, respectively, of that from an intravenous injection of the drug. Although furosemide is more rapidly absorbed from the oral solution (50 minutes) than from the tablet (87 minutes), peak plasma levels and area under the plasma concentration-time curves do not differ significantly. Peak plasma concentrations increase with increasing dose but times-to-peak do not differ among doses. The terminal half-life of furosemide is approximately 2 hours.
Significantly more furosemide is excreted in urine following the IV injection than after the tablet or oral solution. There are no significant differences between the two oral formulations in the amount of unchanged drug excreted in urine.
Furosemide binding to albumin may be reduced in elderly patients. Furosemide is predominantly excreted unchanged in the urine. The renal clearance of furosemide after intravenous administration in older healthy male subjects (60 to 70 years of age) is statistically significantly smaller than in younger healthy male subjects (20 to 35 years of age). The initial diuretic effect of furosemide in older subjects is decreased relative to younger subjects (see PRECAUTIONS: Geriatric Use ).
HOW SUPPLIED
Furosemide tablets, USP 20 mg, 40 mg and 80 mg are supplied as white to off-white, round tablets. 40 mg tablets and 80 mg tablets have functional scoring.
Furosemide tablets, USP 20 mg are debossed with 'RE22' on one side and plain on the other side and are supplied as follows:
NDC 83980-002-97 Bottles of 10
NDC 83980-002-01 Bottles of 100
NDC 83980-002-10 Bottles of 1,000
Furosemide tablets, USP 40 mg are debossed with 'RE23' on one side and break-line on the other side and are supplied as follows:
NDC 83980-003-97 Bottles of 10
NDC 83980-003-01 Bottles of 100
NDC 83980-003-10 Bottles of 1,000
Furosemide tablets, USP 80 mg are debossed with 'RE24' on one side and break-line on the other side and are supplied as follows:
NDC 83980-004-97 Bottles of 10
NDC 83980-004-01 Bottles of 100
NDC 83980-004-05 Bottles of 500
NDC 83980-004-10 Bottles of 1,000
Note: Dispense in well-closed, light-resistant containers. Exposure to light might cause a slight discoloration. Discolored tablets should not be dispensed
Store at 20°C to 25°C (68°F to 77°F); [See USP Controlled Room Temperature].
Protect from light.