Fosfomycin Tromethamine prescribing information
INDICATIONS AND USAGE
Fosfomycin Tromethamine is indicated only for the treatment of uncomplicated urinary tract infections (acute cystitis) in women due to susceptible strains of Escherichia coli and Enterococcus faecalis . Fosfomycin Tromethamine Granules for Oral Solution is not indicated for the treatment of pyelonephritis or perinephric abscess.
If persistence or reappearance of bacteriuria occurs after treatment with Fosfomycin Tromethamine, other therapeutic agents should be selected. (See PRECAUTIONS and CLINICAL STUDIES sections.)
DOSAGE AND ADMINISTRATION
The recommended dosage for women 18 years of age and older for uncomplicated urinary tract infection (acute cystitis) is one sachet of Fosfomycin Tromethamine Granules for Oral Solution. Fosfomycin Tromethamine Granules for Oral Solution may be taken with or without food. Fosfomycin Tromethamine Granules for Oral Solution should not be taken in its dry form. Always mix Fosfomycin Tromethamine Granules for Oral Solution with water before ingesting. (See PREPARATION section.)
CONTRAINDICATIONS
Fosfomycin Tromethamine Granules for Oral Solution is contraindicated in patients with known hypersensitivity to the drug.
ADVERSE REACTIONS
Clinical Trials
In clinical studies, drug related adverse events which were reported in greater than 1% of the fosfomycin-treated study population are listed below:
| Adverse Events | Fosfomycin N = 1233 | Nitrofurantoin N = 374 | Trimethoprim/Sulfamethoxazole N = 428 | Ciprofloxacin N = 455 |
|---|---|---|---|---|
| Diarrhea | 9.0 | 6.4 | 2.3 | 3.1 |
| Vaginitis | 5.5 | 5.3 | 4.7 | 6.3 |
| Nausea | 4.1 | 7.2 | 8.6 | 3.4 |
| Headache | 3.9 | 5.9 | 5.4 | 3.4 |
| Dizziness | 1.3 | 1.9 | 2.3 | 2.2 |
| Asthenia | 1.1 | 0.3 | 0.5 | 0.0 |
| Dyspepsia | 1.1 | 2.1 | 0.7 | 1.1 |
In clinical trials, the most frequently reported adverse events occurring in > 1% of the study population regardless of drug relationship were: diarrhea 10.4%, headache 10.3%, vaginitis 7.6%, nausea 5.2%, rhinitis 4.5%, back pain 3.0%, dysmenorrheal 2.6%, pharyngitis 2.5%, dizziness 2.3%, abdominal pain 2.2%, pain 2.2%, dyspepsia 1.8%, asthenia 1.7%, and rash 1.4%.
The following adverse events occurred in clinical trials at a rate of less than 1%, regardless of drug relationship: abnormal stools, anorexia, constipation, dry mouth, dysuria, ear disorder, fever, flatulence, flu syndrome, hematuria, infection, insomnia, lymphadenopathy, menstrual disorder, migraine, myalgia, nervousness, paresthesia, pruritus, SGPT increased, skin disorder, somnolence, and vomiting.
One patient developed unilateral optic neuritis, an event considered possibly related to Fosfomycin Tromethamine Granules for Oral Solution therapy.
Post-marketing Experience
Serious adverse events from the marketing experience with Fosfomycin Tromethamine Granules for Oral Solution outside of the United States have been rarely reported and include angioedema, aplastic anemia, asthma (exacerbation), cholestatic jaundice, hepatic necrosis, and toxic megacolon. Although causality has not been established, during post-marketing surveillance, the following events have occurred in patients prescribed Fosfomycin Tromethamine: anaphylaxis and hearing loss.
Laboratory Changes
Significant laboratory changes reported in U.S. clinical trials of Fosfomycin Tromethamine Granules for Oral Solution without regard to drug relationship include: increased eosinophil count, increased or decreased WBC count, increased bilirubin, increased SGPT, increased SGOT, increased alkaline phosphatase, decreased hematocrit, decreased hemoglobin, increased and decreased platelet count. The changes were generally transient and were not clinically significant.
To report SUSPECTED ADVERSE REACTIONS, contact Precision Dose, Inc. at 1-800-397-9228 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Metoclopramide
When coadministered with Fosfomycin Tromethamine Granules for Oral Solution, metoclopramide, a drug which increases gastrointestinal motility, lowers the serum concentration and urinary excretion of fosfomycin. Other drugs that increase gastrointestinal motility may produce similar effects.
Cimetidine
Cimetidine does not affect the pharmacokinetics of fosfomycin when coadministered with Fosfomycin Tromethamine Granules for Oral Solution.
DESCRIPTION
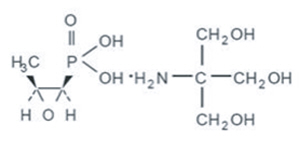
Fosfomycin Tromethamine Granules for Oral Solution contains Fosfomycin Tromethamine, a synthetic, broad spectrum, bactericidal antibiotic for oral administration. It is available as a single dose sachet which contains white granules consisting of 5.631 grams of Fosfomycin Tromethamine (equivalent to 3 grams of fosfomycin), and the following inactive ingredients: orange flavor, saccharin, and sucrose. The contents of the sachet must be dissolved in water. Fosfomycin Tromethamine, a phosphonic acid derivative, is available as (1R,2S)-(1,2-epoxypropyl)phosphonic acid, compound with 2-amino-2-(hydroxy-methyl)-1,3-propanediol (1:1). It is a white granular compound with a molecular weight of 259.2. Its empirical formula is C3H7O4P∙C4H11NO3, and its chemical structure is as follows:
CLINICAL PHARMACOLOGY
Absorption
Fosfomycin Tromethamine is rapidly absorbed following oral administration and converted to the free acid, fosfomycin. Absolute oral bioavailability under fasting conditions is 37%. After a single 3-gram dose of Fosfomycin Tromethamine Granules for Oral Solution, the mean (± 1 SD) maximum serum concentration (Cmax) achieved was 26.1 (± 9.1) mcg/mL within 2 hours. The oral bioavailability of fosfomycin is reduced to 30% under fed conditions. Following a single 3-gram oral dose of fosfomycin Tromethamine Granules for Oral Solution with a high-fat meal, the mean Cmax achieved was 17.6 (± 4.4) mcg/mL within 4 hours. Cimetidine does not affect the pharmacokinetics of Fosfomycin when co-administered with Fosfomycin Tromethamine Granules for Oral Solution. Metoclopramide lowers the serum concentrations and urinary excretion of fosfomycin when coadministered with Fosfomycin Tromethamine Granules for Oral Solution. (See PRECAUTIONS, Drug Interactions .)
Distribution
The mean apparent steady-state volume of distribution (Vss) is 136.1 (±44.1) L following oral administration of Fosfomycin Tromethamine Granules for Oral Solution. Fosfomycin is not bound to plasma proteins.
Fosfomycin is distributed to the kidneys, bladder wall, prostate, and seminal vesicles. Following a 50 mg/kg dose of fosfomycin to patients undergoing urological surgery for bladder carcinoma, the mean concentration of fosfomycin in the bladder, taken at a distance from the neoplastic site, was 18.0 mcg per gram of tissue at 3 hours after dosing. Fosfomycin has been shown to cross the placental barrier in animals and man.
Excretion
Fosfomycin is excreted unchanged in both urine and feces. Following oral administration of Fosfomycin Tromethamine Granules for Oral Solution, the mean total body clearance (CLTB) and mean renal clearance (CLR) of fosfomycin were 16.9 (± 3.5) L/hr and 6.3 (± 1.7) L/hr, respectively. Approximately 38% of a 3-gram dose of Fosfomycin Tromethamine Granules for Oral Solution is recovered from urine, and 18% is recovered from feces. Following intravenous administration, the mean CLTB and mean CLR of fosfomycin were 6.1 (±1.0) L/hr and 5.5 (± 1.2) L/hr, respectively.
A mean urine fosfomycin concentration of 706 (± 466) mcg/mL was attained within 2-4 hours after a single oral 3-gm dose of Fosfomycin Tromethamine Granules for Oral Solution under fasting conditions. The mean urinary concentration of fosfomycin was 10 mcg/mL in samples collected 72-84 hours following a single oral dose of Fosfomycin Tromethamine.
Following a 3-gram dose of Fosfomycin Tromethamine Granules for Oral Solution administered with a high fat meal, a mean urine fosfomycin concentration of 537 (± 252) mcg/mL was attained within 6-8 hours. Although the rate of urinary excretion of fosfomycin was reduced under fed conditions, the cumulative amount of fosfomycin excreted in the urine was the same, 1118 (± 201) mg (fed) vs. 1140 mg (± 238) (fasting). Further, urinary concentrations equal to or greater than 100 mcg/mL were maintained for the same duration, 26 hours, indicating that Fosfomycin Tromethamine Granules for Oral Solution can be taken without regard to food. Following oral administration of Fosfomycin Tromethamine Granules for Oral Solution the mean half-life for elimination (t1/2) is 5.7 (± 2.8) hours.
Special Populations
Geriatric
Based on limited data regarding 24-hour urinary drug concentrations, no differences in urinary excretion of fosfomycin have been observed in elderly subjects. No dosage adjustment is necessary in the elderly.
Gender
There are no gender differences in the pharmacokinetics of fosfomycin.
Renal Insufficiency
In 5 anuric patients undergoing hemodialysis, the t1/2 of fosfomycin during hemodialysis was 40 hours. In patients with varying degrees of renal impairment (creatinine clearances varying from 54 mL/min to 7 mL/min), the t1/2 of fosfomycin increased from 11 hours to 50 hours. The percent of fosfomycin recovered in urine decreased from 32% to 11% indicating that renal impairment significantly decreases the excretion of fosfomycin.
Microbiology
Fosfomycin (the active component of Fosfomycin Tromethamine) has in vitro activity against a broad range of gram-positive and gram-negative aerobic microorganisms which are associated with uncomplicated urinary tract infections. Fosfomycin is bactericidal in urine at therapeutic doses. The bactericidal action of fosfomycin is due to its inactivation of the enzyme enolpyruvyl transferase, thereby irreversibly blocking the condensation of uridine diphosphate-N-acetylglucosamine with penolpyruvate, one of the first steps in bacterial cell wall synthesis. It also reduces adherence of bacteria to uroepithelial cells.
There is generally no cross-resistance between fosfomycin and other classes of antibacterial agents such as beta-lactams and aminoglycosides. Fosfomycin has been shown to be active against most strains of the following microorganisms, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section:
Aerobic gram-positive microorganisms
Enterococcus faecalis
Aerobic gram-negative microorganisms
Escherichia coli
The following in vitro data are available, but their clinical significance is unknown.
Fosfomycin exhibits in vitro minimum inhibitory concentrations (MIC's) of 64 mcg/mL or less against most (≥ 90%) strains of the following microorganisms; however, the safety and effectiveness of fosfomycin in treating clinical infections due to these microorganisms has not been established in adequate and well-controlled clinical trials:
Aerobic gram-positive microorganisms
Enterococcus faecium
Aerobic gram-negative microorganisms
Citrobacter diversus Citrobacter freundii Enterobacter aerogenes Klebsiella oxytoca Klebsiella pneuomoniae Proteus mirabilis Proteus vulgaris Serratia marcescens
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
CLINICAL STUDIES
In controlled, double-blind studies of acute cystitis performed in the United States, a single dose of Fosfomycin Tromethamine Granules for Oral Solution was compared to three other oral antibiotics (See table below). The study population consisted of patients with symptoms and signs of acute cystitis of less than 4 days duration, no manifestations of upper tract infection (e.g., flank pain, chills, fever), no history of recurrent urinary tract infections (20% of patients in the clinical studies had a prior episode of acute cystitis within the preceding year), no known structural abnormalities, no clinical or laboratory evidence of hepatic dysfunction, and no known or suspected CNS disorders, such as epilepsy, or other factors which would predispose to seizures. In these studies, the following clinical success (resolution of symptoms) and microbiologic eradication rates were obtained.
| Treatment Arm | Treatment Duration | Microbiologic Eradication Rate | Clinical Success Rate | Outcome (based on difference in microbiologic eradication rates 5-11 days posttherapy | |
|---|---|---|---|---|---|
| (Days) | 5-11 days post therapy | Study day 12-21 | |||
| Fosfomycin | 1 | 630/771 (82%) | 591/771 (70%) | 542/771 (70%) | |
| Ciprofloxacin | 7 | 219/222 (98%) | 219/222 (98%) | 213/222 (96%) | Fosfomycin inferior to Ciprofloxacin |
| Trimethoprim/ Sulfamethoxazole | 10 | 194/197 (98%) | 194/197 (98%) | 186/197 (94%) | Fosfomycin inferior to trimethoprim/Sulfamethoxazole |
| Nitrofurantoin | 7 | 180/238 (76%) | 180/238 (76%) | 183/238 (77%) | Fosfomycin equivalent to nitrofurantion |
| Pathogen | Fosfomycin 3 gram single dose | Ciprofloxacin 250 mg Bid × 7 days | Trimethoprim/Sulfamethoxazole 160 mg/800 mg bid × 10 days | Nitrofurantoin 100 mg Bid × 7 days |
|---|---|---|---|---|
| E. coli | 509/644 (79%) | 184/187 (98%) | 171/174 (98%) | 146/187 (78%) |
| E. Faecalis | 10/10 (100%) | 0/0 | 4/4 (100%) | 1/2 (50%) |
HOW SUPPLIED
Fosfomycin Tromethamine Granules for Oral Solution is available as a single-dose sachet containing the equivalent of 3 grams of Fosfomycin.
| NDC | |
|---|---|
| Single-dose sachet | 68094-064-01 |
| One unit carton | 68094-064-20 |
Store at 25°C (77°F); excursions permitted to 15-30°C (59-86°F). Keep this and all drugs out of the reach of children.