Get your patient on Fludrocortisone Acetate - Fludrocortisone Acetate tablet (Fludrocortisone Acetate)
Fludrocortisone Acetate - Fludrocortisone Acetate tablet prescribing information
INDICATIONS & USAGE
Fludrocortisone acetate tablets, 0.1 mg are indicated as partial replacement therapy for primary and secondary adrenocortical insufficiency in Addison’s disease and for the treatment of salt-losing adrenogenital syndrome.
DOSAGE & ADMINISTRATION
Dosage depends on the severity of the disease and the response of the patient. Patients should be continually monitored for signs that indicate dosage adjustment is necessary, such as remission or exacerbations of the disease and stress (surgery, infection, trauma) (see WARNINGS and PRECAUTIONS, General ).
Addison's Disease
In Addison’s disease, the combination of fludrocortisone acetate tablets with a glucocorticoid such as hydrocortisone or cortisone provides substitution therapy approximating normal adrenal activity with minimal risks of unwanted effects.
The usual dose is 0.1 mg of fludrocortisone acetate tablets daily, although dosage ranging from 0.1 mg three times a week to 0.2 mg daily has been employed. In the event transient hypertension develops as a consequence of therapy, the dose should be reduced to 0.05 mg daily. Fludrocortisone acetate tablets are preferably administered in conjunction with cortisone (10 mg to 37.5 mg daily in divided doses) or hydrocortisone (10 mg to 30 mg daily in divided doses).
Salt-Losing Adrenogenital Syndrome
The recommended dosage for treating the salt-losing adrenogenital syndrome is 0.1 mg to 0.2 mg of fludrocortisone acetate tablets daily.
CONTRAINDICATIONS
Corticosteroids are contraindicated in patients with systemic fungal infections and in those with a history of possible or known hypersensitivity to these agents.
ADVERSE REACTIONS
Most adverse reactions are caused by the drug’s mineralocorticoid activity (retention of sodium and water) and include hypertension, edema, cardiac enlargement, congestive heart failure, potassium loss, and hypokalemic alkalosis.
When fludrocortisone is used in the small dosages recommended, the glucocorticoid side effects often seen with cortisone and its derivatives are not usually a problem; however, the following untoward effects should be kept in mind, particularly when fludrocortisone is used over a prolonged period of time or in conjunction with cortisone or a similar glucocorticoid.
Musculoskeletal —muscle weakness, steroid myopathy, loss of muscle mass, osteoporosis, vertebral compression fractures, aseptic necrosis of femoral and humeral heads, pathologic fracture of long bones, and spontaneous fractures.
Gastrointestinal —peptic ulcer with possible perforation and hemorrhage, pancreatitis, abdominal distention, and ulcerative esophagitis.
Dermatologic —impaired wound healing, thin fragile skin, bruising, petechiae and ecchymoses, facial erythema, increased sweating, subcutaneous fat atrophy, purpura, striae, hyperpigmentation of the skin and nails, hirsutism, acneiform eruptions and hives; reactions to skin tests may be suppressed.
Neurological —convulsions, increased intracranial pressure with papilledema (psuedo-tumor cerebri) usually after treatment, vertigo, headache, and severe mental disturbances.
Endocrine —menstrual irregularities; development of the cushingoid state; suppression of growth in children; secondary adrenocortical and pituitary unresponsiveness, particularly in times of stress (e.g., trauma, surgery, or illness); decreased carbohydrate tolerance; manifestations of latent diabetes mellitus; and increased requirements for insulin or oral hypoglycemic agents in diabetics.
Ophthalmic —posterior subcapsular cataracts, increased intraocular pressure, glaucoma, and exophthalmos.
Metabolic —hyperglycemia, glycosuria, and negative nitrogen balance due to protein catabolism.
Allergic Reactions —allergic skin rash, maculopapular rash, and urticaria.
Other adverse reactions that may occur following the administration of a corticosteroid are necrotizing angiitis, thrombophlebitis, aggravation or masking of infections, insomnia, syncopal episodes, and anaphylactoid reactions.
To report SUSPECTED ADVERSE REACTIONS, contact Novitium Pharma, LLC at 1-855-204-1431 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
When administered concurrently, the following drugs may interact with adrenal corticosteroids.
Amphotericin B or potassium-depleting diuretics (benzothiadiazines and related drugs, ethacrynic acid and furosemide)—enhanced hypokalemia. Check serum potassium levels at frequent intervals; use potassium supplements if necessary (see WARNINGS ).
Digitalis glycosides — enhanced possibility of arrhythmias or digitalis toxicity associated with hypokalemia. Monitor serum potassium levels; use potassium supplements if necessary.
Oral anticoagulants — decreased prothrombin time response. Monitor prothrombin levels and adjust anticoagulant dosage accordingly.
Antidiabetic drugs (oral agents and insulin)—diminished antidiabetic effect. Monitor for symptoms of hyperglycemia; adjust dosage of antidiabetic drug upward if necessary.
Aspirin — increased ulcerogenic effect; decreased pharmacologic effect of aspirin. Rarely salicylate toxicity may occur in patients who discontinue steroids after concurrent high-dose aspirin therapy. Monitor salicylate levels or the therapeutic effect for which aspirin is given; adjust salicylate dosage accordingly if effect is altered (see PRECAUTIONS, General ).
Barbiturates, phenytoin, or rifampin —increased metabolic clearance of fludrocortisone acetate because of the induction of hepatic enzymes. Observe the patient for possible diminished effect of steroid and increase the steroid dosage accordingly.
Anabolic steroids (particularly C-17 alkylated androgens such as oxymetholone, methandrostenolone, norethandrolone, and similar compounds)—enhanced tendency toward edema. Use caution when giving these drugs together, especially in patients with hepatic or cardiac disease.
Vaccines —neurological complications and lack of antibody response (see WARNINGS ).
Estrogen —increased levels of corticosteroid-binding globulin thereby increasing the bound (inactive) fraction; this effect is at least balanced by decreased metabolism of corticosteroids. When estrogen therapy is initiated, a reduction in corticosteroid dosage may be required, and increased amounts may be required when estrogen is terminated.
DESCRIPTION
Fludrocortisone acetate tablets USP, 0.1 mg contain fludrocortisone acetate, a synthetic adrenocortical steroid possessing very potent mineralocorticoid properties and high glucocorticoid activity; it is used only for its mineralocorticoid effects. The chemical name for fludrocortisone acetate is 9-fluoro-11β, 17, 21-trihydroxypregn-4-ene-3, 20-dione 21-acetate; its structural formula is:
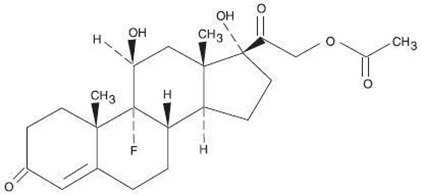
C 23 H 31 FO 6 MW 422.49
Fludrocortisone acetate tablets USP, 0.1 mg are available for oral administration as scored tablets providing 0.1 mg fludrocortisone acetate per tablet. Inactive ingredients: lactose anhydrous, lactose monohydrate, microcrystalline cellulose, croscarmellose sodium, magnesium stearate.
FDA approved dissolution test specifications differ from USP.
CLINICAL PHARMACOLOGY
Corticosteroids are thought to act at least in part, by controlling the rate of synthesis of proteins. Although there are a number of instances in which the synthesis of specific proteins is known to be induced by corticosteroids, the links between the initial actions of the hormones and the final metabolic effects have not been completely elucidated.
The physiologic action of fludrocortisone acetate is similar to that of hydrocortisone. However, the effects of fludrocortisone acetate, particularly on electrolyte balance, but also on carbohydrate metabolism, are considerably heightened and prolonged. Mineralocorticoids act on the distal tubules of the kidney to enhance the reabsorption of sodium ions from the tubular fluid into the plasma; they increase the urinary excretion of both potassium and hydrogen ions. The consequence of these three primary effects together with similar actions on cation transport in other tissues appear to account for the entire spectrum of physiological activities that are characteristic of mineralocorticoids. In small oral doses, fludrocortisone acetate produces marked sodium retention and increased urinary potassium excretion. It also causes a rise in blood pressure, apparently because of these effects on electrolyte levels.
In larger doses, fludrocortisone acetate inhibits endogenous adrenal cortical secretion, thymic activity, and pituitary corticotropin excretion; promotes the deposition of liver glycogen; and, unless protein intake is adequate, induces negative nitrogen balance.
The approximate plasma half-life of fludrocortisone (fluorohydrocortisone) is 3.5 hours or more and the biological half-life is 18 to 36 hours.
HOW SUPPLIED
Fludrocortisone Acetate Tablets USP, 0.1 mg — White to off white, round bisect tablets, debossed with “N” above the bisect and “252” below the bisect and plain on other side.
They are available as follows:
Bottles of 30: NDC 70954-252-10
Bottles of 100: NDC 70954-252-20
Bottles of 500: NDC 70954-252-30
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Avoid excessive heat.
Dispense in a tightly-closed, light-resistant container (USP).
Manufactured by:
Novitium Pharma LLC
70 Lake Drive, East Windsor
New Jersey 08520
Issued: 03/2022
LB4456-00