Get your patient on Flucytosine - Flucytosine capsule (Flucytosine)
Flucytosine - Flucytosine capsule prescribing information
WARNING
Use with extreme caution in patients with impaired renal function. Close monitoring of hematologic, renal and hepatic status of all patients is essential. These instructions should be thoroughly reviewed before administration of Flucytosine Capsules, 250 mg and 500 mg.
INDICATIONS AND USAGE
Flucytosine capsules are indicated only in the treatment of serious infections caused by susceptible strains of Candida and/or Cryptococcus .
Candida: Septicemia, endocarditis and urinary system infections have been effectively treated with flucytosine. Limited trials in pulmonary infections justify the use of flucytosine.
Cryptococcus: Meningitis and pulmonary infections have been treated effectively. Studies in septicemias and urinary tract infections are limited, but good responses have been reported.
Flucytosine capsules should be used in combination with amphotericin B for the treatment of systemic candidiasis and cryptococcosis because of the emergence of resistance to Flucytosine capsules (see MICROBIOLOGY ).
DOSAGE AND ADMINISTRATION
The usual dosage of flucytosine capsules are 50 to 150 mg/kg/day administered in divided doses at 6-hour intervals. Nausea or vomiting may be reduced or avoided if the capsules are given a few at a time over a 15-minute period. If the BUN or the serum creatinine is elevated, or if there are other signs of renal impairment, the initial dose should be at the lower level (see WARNINGS ).
Flucytosine capsules should be used in combination with amphotericin B for the treatment of systemic candidiasis and cryptococcosis because of the emergence of resistance to flucytosine capsules (see MICROBIOLOGY ).
CONTRAINDICATIONS
Flucytosine capsule is contraindicated in patients with a known hypersensitivity to the drug.
Flucytosine capsule is contraindicated in patients with known complete dihydropyrimidine dehydrogenase (DPD) enzyme deficiency (see WARNINGS ).
ADVERSE REACTIONS
The adverse reactions which have occurred during treatment with Flucytosine capsules are grouped according to organ system affected.
Cardiovascular: Cardiac arrest, myocardial toxicity, ventricular dysfunction.
Respiratory: Respiratory arrest, chest pain, dyspnea.
Dermatologic: Rash, pruritus, urticaria, photosensitivity.
Gastrointestinal: Nausea, emesis, abdominal pain, diarrhea, anorexia, dry mouth, duodenal ulcer, gastrointestinal hemorrhage, acute hepatic injury including hepatic necrosis with possible fatal outcome in debilitated patients, hepatic dysfunction, jaundice, ulcerative colitis, enterocolitis, bilirubin elevation, increased hepatic enzymes.
Genitourinary: Azotemia, creatinine and BUN elevation, crystalluria, renal failure.
Hematologic: Anemia, agranulocytosis, aplastic anemia, eosinophilia, leukopenia, pancytopenia, thrombocytopenia, and fatal cases of bone marrow aplasia.
Neurologic: Ataxia, hearing loss, headache, paresthesia, parkinsonism, peripheral neuropathy, pyrexia, vertigo, sedation, convulsions.
Psychiatric: Confusion, hallucinations, psychosis.
Miscellaneous: Fatigue, hypoglycemia, hypokalemia, weakness, allergic reactions, Lyell's syndrome.
You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Avet Pharmaceuticals Inc. at 1-866-901-DRUG (3784) or go to www.avetpharma.com.
DESCRIPTION
Flucytosine capsules USP, an antifungal agent, is available as 250 mg and 500 mg capsules for oral administration. In addition to the active ingredient of flucytosine, each capsule contains, corn starch, lactose monohydrate, and talc. The 250 mg capsule shell contains black iron oxide, D&C Yellow No. 10, FD&C Blue No. 1, FD&C Yellow No. 6, gelatin and titanium dioxide. The 500 mg capsule shell contains black iron oxide, gelatin and titanium dioxide. The imprinting ink Tekprint™ SW-9008 Black contains, black iron oxide, potassium hydroxide, propylene glycol and shellac.
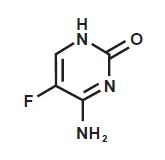
Chemically, flucytosine is 5-fluorocytosine, a fluorinated pyrimidine which is related to fluorouracil and floxuridine. It is a white to off-white powder with a molecular weight of 129.09 and the following structural formula:
FDA approved dissolution specification differs from the USP dissolution specification.
CLINICAL PHARMACOLOGY
Flucytosine is rapidly and virtually completely absorbed following oral administration. Flucytosine capsules are not metabolized significantly when given orally to man. Bioavailability estimated by comparing the area under the curve of serum concentrations after oral and intravenous administration showed 78% to 89% absorption of the oral dose. Peak serum concentrations of 30 to 40 mcg/mL were reached within 2 hours of administration of a 2 g oral dose to normal subjects. Other studies revealed mean serum concentrations of approximately 70 to 80 mcg/mL 1 to 2 hours after a dose in patients with normal renal function receiving a 6-week regimen of flucytosine (150 mg/kg/day given in divided doses every 6 hours) in combination with amphotericin B. The half-life in the majority of healthy subjects ranged between 2.4 and 4.8 hours. Flucytosine is excreted via the kidneys by means of glomerular filtration without significant tubular reabsorption. More than 90% of the total radioactivity after oral administration was recovered in the urine as intact drug. Flucytosine is deaminated (probably by gut bacteria) to 5-fluorouracil. The area under the curve (AUC) ratio of 5-fluorouracil to flucytosine is 4%. Approximately 1% of the dose is present in the urine as the α-fluoro-β-ureido-propionic acid metabolite. A small portion of the dose is excreted in the feces.
The half-life of flucytosine is prolonged in patients with renal insufficiency; the average half-life in nephrectomized or anuric patients was 85 hours (range: 29.9 to 250 hours). A linear correlation was found between the elimination rate constant of flucytosine and creatinine clearance.
In vitro studies have shown that 2.9% to 4% of flucytosine is protein-bound over the range of therapeutic concentrations found in the blood. Flucytosine readily penetrates the blood-brain barrier, achieving clinically significant concentrations in cerebrospinal fluid.
Pharmacokinetics in Pediatric Patients
Limited data are available regarding the pharmacokinetics of flucytosine capsules administered to neonatal patients being treated for systemic candidiasis. After five days of continuous therapy, median peak levels in infants were 19.6 mcg/mL, 27.7 mcg/mL, and 83.9 mcg/mL at doses of 25 mg/kg (N=3), 50 mg/kg (N=4), and 100 mg/kg (N=3), respectively. Mean time to peak serum levels was of 2.5 ± 1.3 hours, similar to that observed in adult patients. A good deal of interindividual variability was noted, which did not correlate with gestational age. Some patients had serum levels > 100 mcg/mL, suggesting a need for drug level monitoring during therapy. In another study, serum concentrations were determined during flucytosine therapy in two patients (total assays performed =10). Median serum flucytosine concentrations at steady state were calculated to be 57 ± 10 mcg/mL (doses of 50 to 125 mg/kg/day, normalized to 25 mg/kg per dose for comparison). In three infants receiving flucytosine 25 mg/kg/day (four divided doses), a median flucytosine half-life of 7.4 hours was observed, approximately double that seen in adult patients. The concentration of flucytosine in the cerebrospinal fluid of one infant was 43 mcg/mL 3 hours after a 25 mg oral dose, and ranged from 20 to 67 mg/L in another neonate receiving oral doses of 120 to 150 mg/kg/day.
HOW SUPPLIED
Flucytosine capsules, USP are supplied as capsules containing 250 mg and 500 mg flucytosine as follows:
| NDC | Strength | Quantity | Description |
| 23155-860-03 | 250 mg | Bottles of 30 | Green opaque cap imprinted in black with "FLU" and grey opaque body imprinted in black with "250" |
| 23155-860-01 | 250 mg | Bottles of 100 | Green opaque cap imprinted in black with "FLU" and grey opaque body imprinted in black with "250" |
| 23155-861-03 | 500 mg | Bottles of 30 | Grey opaque cap imprinted in black with "FLU" and white opaque body imprinted in black with "500" |
| 23155-861-01 | 500 mg | Bottles of 100 | Grey opaque cap imprinted in black with "FLU" and white opaque body imprinted in black with "500" |
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F).
Manufactured by:
Strides Pharma Science Limited
Bengaluru - 562106, India
Manufactured for:
Avet Pharmaceuticals Inc.
East Brunswick, NJ 08816
1.866.901.DRUG (3784)

Revised: 04/2023
Flucytosine - Flucytosine capsule PubMed™ news
- Journal Article • 2026 MayEvaluation of Sensititre YeastOne for antifungal susceptibility testing of Candida (Candidozyma) auris: misclassification of FKS1 mutants and overestimation of caspofungin resistance in a global collection of isolates.
- Journal Article • 2026 MayPhenotypic and Molecular Characterization of Naganishia uzbekistanensis: Diagnostic Challenges and Antifungal Resistance Profile.
- Journal Article • 2026 AprEpidemiology, clinical and microbiologic aspects of Rhodotorula fungemia: A review of reported cases over the past 16 years.
- Journal Article • 2026 AprCandida urinary tract infections, antifungal susceptibility and factors associated with candiduria among pregnant women seeking antenatal care at Murang'a County Referral Hospital, Kenya.
- Journal Article • 2026 AprRefractory Candidozyma (Candida) auris-associated central nervous system infection in a postoperative neurosurgical patient.