Get your patient on Flexprofen (Carprofen)
Flexprofen prescribing information
DOSAGE AND ADMINISTRATION: Always provide Client Information Sheet with prescription. Carefully consider the potential benefits and risk of Flexprofen Tablets and other treatment options before deciding to use Flexprofen Tablets. Use the lowest effective dose for the shortest duration consistent with individual response. The recommended dosage for oral administration to dogs is 2 mg/lb (4.4 mg/kg) of body weight daily. The total daily dose may be administered as 2 mg/lb of body weight once daily or divided and administered as 1 mg/lb (2.2 mg/kg) twice daily. For the control of postoperative pain, administer approximately 2 hours before the procedure. Tablets are scored and dosage should be calculated in half-tablet increments.
CONTRAINDICATIONS: Flexprofen Tablets should not be used in dogs exhibiting previous hypersensitivity to carprofen.
ADVERSE REACTIONS: During investigational studies of osteoarthritis with twice daily administration of 1 mg/lb, no clinically significant adverse reactions were reported. Some clinical signs were observed during field studies (n=297) which were similar for carprofen- and placebo-treated dogs. Incidences of the following were observed in both groups: vomiting (4%), diarrhea (4%), changes in appetite (3%), lethargy (1.4%), behavioral changes (1%), and constipation (0.3%). The product vehicle served as control.
There were no serious adverse events reported during clinical field studies of osteoarthritis with once daily administration of 2 mg/lb. The following categories of abnormal health observations were reported. The product vehicle served as control.
| Observation | Carprofen tablets (n=129) | Placebo (n=132) |
|---|---|---|
| Inappetence | 1.6 | 1.5 |
| Vomiting | 3.1 | 3.8 |
| Diarrhea/Soft stool | 3.1 | 4.5 |
| Behavior change | 0.8 | 0.8 |
| Dermatitis | 0.8 | 0.8 |
| PU/PD | 0.8 | -- |
| SAP increase | 7.8 | 8.3 |
| ALT increase | 5.4 | 4.5 |
| AST increase | 2.3 | 0.8 |
| BUN increase | 3.1 | 1.5 |
| Bilirubinuria | 16.3 | 12.1 |
| Ketonuria | 14.7 | 9.1 |
Clinical pathology parameters listed represent reports of increases from pre-treatment values; medical judgment is necessary to determine clinical relevance.
During investigational studies of surgical pain for the tablet formulation, no clinically significant adverse reactions were reported. The product vehicle served as control.
| Observation A single dog may have experienced more than one occurrence of an event. | Carprofen tablets (n=148) | Placebo (n=149) |
|---|---|---|
| Vomiting | 10.1 | 13.4 |
| Diarrhea/soft stool | 6.1 | 6.0 |
| Ocular disease | 2.7 | 0 |
| Inappetence | 1.4 | 0 |
| Dermatitis/skin lesion | 2.0 | 1.3 |
| Dysrhythmia | 0.7 | 0 |
| Apnea | 1.4 | 0 |
| Oral/periodontal disease | 1.4 | 0 |
| Pyrexia | 0.7 | 1.3 |
| Urinary tract disease | 1.4 | 1.3 |
| Wound drainage | 1.4 | 0 |
Post-Approval Experience:
Although not all adverse reactions are reported, the following adverse reactions are based on voluntary post-approval adverse drug experience reporting. The categories of adverse reactions are listed in decreasing order of frequency by body system.
Gastrointestinal: Vomiting, diarrhea, constipation, inappetence, melena, hematemesis, gastrointestinal ulceration, gastrointestinal bleeding, pancreatitis.
Hepatic: Inappetence, vomiting, jaundice, acute hepatic toxicity, hepatic enzyme elevation, abnormal liver function test(s), hyperbilirubinemia, bilirubinuria, hypoalbuminemia. Approximately one-fourth of hepatic reports were in Labrador Retrievers.
Neurologic: Ataxia, paresis, paralysis, seizures, vestibular signs, disorientation.
Urinary: Hematuria, polyuria, polydipsia, urinary incontinence, urinary tract infection, azotemia, acute renal failure, tubular abnormalities including acute tubular necrosis, renal tubular acidosis, glucosuria.
Behavioral: Sedation, lethargy, hyperactivity, restlessness, aggressiveness.
Hematologic: Immune-mediated hemolytic anemia, immune-mediated thrombocytopenia, blood loss anemia, epistaxis.
Dermatologic: Pruritus, increased shedding, alopecia, pyotraumatic moist dermatitis (hot spots), necrotizing panniculitis/vasculitis, ventral ecchymosis.
Immunologic or hypersensitivity: Facial swelling, hives, erythema.
In rare situations, death has been associated with some of the adverse reactions listed above.
DESCRIPTION: Carprofen is a non-steroidal anti-inflammatory drug (NSAID) of the propionic acid class that includes ibuprofen, naproxen, and ketoprofen. Carprofen is the nonproprietary designation for a substituted carbazole, 6-chloro-α-methyl-9Hcarbazole-2-acetic acid. The empirical formula is C 15 H 12 CINO 2 and the molecular weight 273.72. The chemical structure of carprofen is:
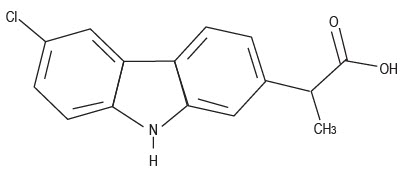
Carprofen is a white, crystalline compound. It is freely soluble in ethanol, but practically insoluble in water at 25°C.
CLINICAL PHARMACOLOGY: Carprofen is a non-narcotic, non-steroidal antiinflammatory agent with characteristic analgesic and antipyretic activity approximately equipotent to indomethacin in animal models. 1
The mechanism of action of carprofen, like that of other NSAIDs, is believed to be associated with the inhibition of cyclooxygenase activity. Two unique cyclooxygenases have been described in mammals. 2 The constitutive cyclooxygenase, COX-1, synthesizes prostaglandins necessary for normal gastrointestinal and renal function. The inducible cyclooxygenase, COX-2, generates prostaglandins involved in inflammation. Inhibition of COX-1 is thought to be associated with gastrointestinal and renal toxicity while inhibition of COX-2 provides anti-inflammatory activity. The specificity of a particular NSAID for COX-2 versus COX-1 may vary from species to species. 3 In an in vitro study using canine cell cultures, carprofen demonstrated selective inhibition of COX-2 versus COX-1. 4 Clinical relevance of these data has not been shown. Carprofen has also been shown to inhibit the release of several prostaglandins in two inflammatory cell systems: rat polymorphonuclear leukocytes (PMN) and human rheumatoid synovial cells, indicating inhibition of acute (PMN system) and chronic (synovial cell system) inflammatory reactions. 1
Several studies have demonstrated that carprofen has modulatory effects on both humoral and cellular immune responses. 5–9 Data also indicate that carprofen inhibits the production of osteoclast-activating factor (OAF), PGE 1 , and PGE 2 by its inhibitory effects on prostaglandin biosynthesis. 1
Based upon comparison with data obtained from intravenous administration, carprofen is rapidly and nearly completely absorbed (more than 90% bioavailable) when administered orally. 10 Peak blood plasma concentrations are achieved in 1–3 hours after oral administration of 1, 5, and 25 mg/kg to dogs. The mean terminal half-life of carprofen is approximately 8 hours (range 4.5–9.8 hours) after single oral doses varying from 1–35 mg/kg of body weight. After a 100 mg single intravenous bolus dose, the mean elimination half-life was approximately 11.7 hours in the dog. Carprofen tablets are more than 99% bound to plasma protein and exhibit a very small volume of distribution.
Carprofen is eliminated in the dog primarily by biotransformation in the liver followed by rapid excretion of the resulting metabolites (the ester glucuronide of carprofen and the ether glucuronides of 2 phenolic metabolites, 7-hydroxy carprofen and 8-hydroxy carprofen) in the feces (70–80%) and urine (10–20%). Some enterohepatic circulation of the drug is observed.
HOW SUPPLIED: Flexprofen (carprofen) Tablets are scored, and contain 25 mg, 75 mg, or 100 mg of carprofen per tablet. Each tablet size is packaged in bottles containing 30, 60, or 180 tablets.
| NDC Number | Tablets Size | Tablets/Bottle |
|---|---|---|
| XXXXX-XXX-XX | 25 mg | 30 Flavored tablets |
| 17030-245-60 | 25 mg | 60 Flavored tablets |
| 17030-245-80 | 25 mg | 180 Flavored tablets |
| XXXXX-XXX-XX | 75 mg | 30 Flavored tablets |
| 17030-246-60 | 75 mg | 60 Flavored tablets |
| 17030-246-80 | 75 mg | 180 Flavored tablets |
| XXXXX-XXX-XX | 100 mg | 30 Flavored tablets |
| 17030-247-60 | 100 mg | 60 Flavored tablets |
| 17030-247-80 | 100 mg | 180 Flavored tablets |