Eurax prescribing information
INDICATIONS AND USAGE
For eradication of scabies ( Sarcoptes scabiei ) and for symptomatic treatment of pruritic skin.
DOSAGE AND ADMINISTRATION
In Scabies: Thoroughly massage into the skin of the whole body from the chin down, paying particular attention to all folds and creases. A second application is advisable 24 hours later. Change clothing and bed linen the next morning. Take a cleansing bath 48 hours after the last application.
In Pruritus: Massage gently into affected areas until medication is completely absorbed. Repeat as needed.
DIRECTIONS FOR PATIENTS WITH SCABIES:
- Take a routine bath or shower. Thoroughly massage Eurax cream into the skin from the chin to the toes including folds and creases.
- Put Eurax cream under fingernails after trimming the fingernails short, because scabies are very likely to remain there. A toothbrush can be used to apply the Eurax cream under the fingernails. Immediately after use, wrap the toothbrush in paper and throw it away. Use of the same brush in the mouth could lead to poisoning.
- A second application is advisable 24 hours later.
- A 60 gram tube is sufficient for two applications.
- Change clothing and bed linen the next day. Dry clean contaminated clothing and bed linen, or wash in the hot cycle of the washing machine.
- Take a cleansing bath 48 hours after the last application.
CONTRAINDICATIONS
Do not apply Eurax topically to patients who develop a sensitivity or are allergic to it or who manifest a primary irritation response to topical medications.
ADVERSE REACTIONS
Primary irritation reactions, such as dermatitis, pruritus, and rash, and allergic sensitivity reactions have been reported in a few patients.
Drug Interactions
None known.
DESCRIPTION
Eurax (crotamiton, USP) is a scabicidal and antipruritic agent available as a cream for topical use only. Eurax provides 10% (w/w) of the synthetic, crotamiton, USP, in a vanishing-cream base containing: carbomer homopolymer type B, cetyl alcohol, diazolidinyl urea, dimethicone, glyceryl monostearate, laureth-23, magnesium aluminum silicate, benzyl alcohol, petrolatum, propylene glycol, sodium hydroxide, steareth-2, and water. Crotamiton is N-ethyl-N-( o -methylphenyl)-2-butenamide and its structural formula is:
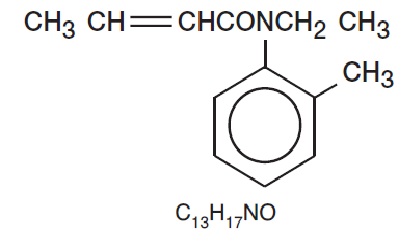
Crotamiton, USP is a colorless to slightly yellowish oil, having a faint amine-like odor. It is soluble with alcohol and with methanol. Crotamiton is a mixture of the cis and trans isomers. Its molecular weight is 203.28.
CLINICAL PHARMACOLOGY
Eurax has scabicidal and antipruritic actions. The mechanisms of these actions are not known. The pharmacokinetics of crotamiton and its degree of systemic absorption following topical application have not been determined.
HOW SUPPLIED
Eurax ® (crotamiton, USP) Cream, 10% is a white to yellowish-white soft cream and supplied as: 60 g tube NDC 69489-311-60
Store at room temperature, 20˚C - 25˚C (68˚F - 77˚F); Excursions permitted: 15˚C - 30˚C (59˚F - 86˚F) [See USP Controlled Room Temperature].
Keep out of reach of children.
To report SUSPECTED ADVERSE REACTIONS, contact Journey Medical Corporation at 1-855-531-1859 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .

Manufactured for: Journey Medical Corporation Scottsdale, AZ 85258 www.JMCderm.com
EUR-P01-R00 Revised 12/2025
DIRECTIONS FOR PATIENTS WITH SCABIES:
- Take a routine bath or shower. Thoroughly massage Eurax cream into the skin from the chin to the toes including folds and creases.
- Put Eurax cream under fingernails after trimming the fingernails short, because scabies are very likely to remain there. A toothbrush can be used to apply the Eurax cream under the fingernails. Immediately after use, wrap the toothbrush in paper and throw it away. Use of the same brush in the mouth could lead to poisoning.
- A second application is advisable 24 hours later.
- A 60 gram tube is sufficient for two applications.
- Change clothing and bed linen the next day. Dry clean contaminated clothing and bed linen, or wash in the hot cycle of the washing machine.
- Take a cleansing bath 48 hours after the last application.