Get your patient on Entacapone - Entacapone tablet, Film Coated (Entacapone)
Entacapone - Entacapone tablet, Film Coated prescribing information
INDICATIONS
Entacapone tablets are indicated as an adjunct to levodopa and carbidopa to treat end-of-dose “wearing-off” in patients with Parkinson’s disease (see CLINICAL PHARMACOLOGY, Clinical Studies).
Entacapone tablets effectiveness has not been systematically evaluated in patients with Parkinson’s disease who do not experience end-of-dose “wearing-off”.
DOSAGE AND ADMINISTRATION
The recommended dose of entacapone is one 200 mg tablet administered concomitantly with each levodopa and carbidopa dose to a maximum of 8 times daily (200 mg x 8 = 1,600 mg per day).
Clinical experience with daily doses above 1,600 mg is limited.
Entacapone tablets should always be administered in association with levodopa and carbidopa. Entacapone has no antiparkinsonian effect of its own.
In clinical studies, the majority of patients required a decrease in daily levodopa dose if their daily dose of levodopa had been greater than or equal to 800 mg or if patients had moderate or severe dyskinesia before beginning treatment.
To optimize an individual patient’s response, reductions in daily levodopa dose or extending the interval between doses may be necessary. In clinical studies, the average reduction in daily levodopa dose was about 25% in those patients requiring a levodopa dose reduction. (More than 58% of patients with levodopa doses above 800 mg daily required such a reduction.)
Entacapone tablets can be combined with both the immediate and sustained-release formulations of levodopa and carbidopa.
Entacapone tablets may be taken with or without food (see CLINICAL PHARMACOLOGY).
Patients With Impaired Hepatic Function : Patients with hepatic impairment should be treated with caution. The AUC and C max of entacapone approximately doubled in patients with documented liver disease, compared to controls. However, these studies were conducted with single-dose entacapone without levodopa and dopa decarboxylase inhibitor coadministration, and therefore the effects of liver disease on the kinetics of chronically administered entacapone have not been evaluated (see CLINICAL PHARMACOLOGY, Pharmacokinetics of Entacapone).
Withdrawing Patients from Entacapone Tablets : Rapid withdrawal or abrupt reduction in the entacapone tablets dose could lead to emergence of signs and symptoms of Parkinson’s disease (see CLINICAL PHARMACOLOGY, Clinical Studies), and may lead to hyperpyrexia and confusion, a symptom complex resembling NMS (see PRECAUTIONS, Other Events Reported With DopaminergicTherapy). This syndrome should be considered in the differential diagnosis for any patient who develops a high fever or severe rigidity. If a decision is made to discontinue treatment with entacapone tablets, patients should be monitored closely and other dopaminergic treatments should be adjusted as needed. Although tapering entacapone has not been systematically evaluated, it seems prudent to withdraw patients slowly if the decision to discontinue treatment is made.
CONTRAINDICATIONS
Entacapone tablets are contraindicated in patients who have demonstrated hypersensitivity to the drug or its ingredients.
ADVERSE REACTIONS
Because clinical studies are conducted under widely varying conditions, the incidence of adverse reactions (number of unique patients experiencing an adverse reaction associated with treatment per total number of patients treated) observed in the clinical studies of a drug cannot be directly compared to the incidence of adverse reactions in the clinical studies of another drug and may not reflect the incidence of adverse reactions observed in practice.
A total of 1,450 patients with Parkinson’s disease were treated with entacapone in premarketing clinical studies. Included were patients with fluctuating symptoms, as well as those with stable responses to levodopa therapy. All patients received concomitant treatment with levodopa preparations, however, and were similar in other clinical aspects.
The most commonly observed adverse reactions (incidence at least 3% greater than placebo) in double- blind, placebo-controlled studies (N=1,003) associated with the use of entacapone were: dyskinesia, urine discoloration, diarrhea, nausea, hyperkinesia, abdominal pain, vomiting, and dry mouth.
Approximately 14% of the 603 patients given entacapone in the double-blind, placebo-controlled studies discontinued treatment due to adverse reactions, compared to 9% of the 400 patients who received placebo. The most frequent causes of discontinuation in decreasing order were: psychiatric disorders (2% vs. 1%), diarrhea (2% vs. 0%), dyskinesia and hyperkinesia (2% vs. 1%), nausea (2% vs. 1%), and abdominal pain (1% vs. 0%).
Adverse Event Incidence in Controlled Clinical Studies
Table 4 lists treatment-emergent adverse events that occurred in at least 1% of patients treated with entacapone participating in the double-blind, placebo-controlled studies and that were numerically more common in the entacapone group, compared to placebo. In these studies, either entacapone or placebo was added to levodopa and carbidopa (or levodopa and benserazide).
Table 4: Summary of Patients with Adverse Events after Start of Trial Drug Administration At least 1% in Entacapone Group and Greater Than Placebo
| SYSTEM ORGAN CLASS Preferred term | Entacapone (n = 603) % of patients | Placebo (n = 400) % of patients | ||
| SKIN AND APPENDAGES DISORDERS | ||||
| Sweating increased | 2 | 1 | ||
| MUSCULOSKELETAL SYSTEM DISORDERS | ||||
| Back pain | 2 | 1 | ||
| CENTRAL AND PERIPHERAL NERVOUS SYSTEM DISORDERS | ||||
| Dyskinesia | 25 | 15 | ||
| Hyperkinesia | 10 | 5 | ||
| Hypokinesia | 9 | 8 | ||
| Dizziness | 8 | 6 | ||
| SPECIAL SENSES, OTHER DISORDERS | ||||
| Taste perversion | 1 | 0 | ||
| PSYCHIATRIC DISORDERS | ||||
| Anxiety | 2 | 1 | ||
| Somnolence | 2 | 0 | ||
| Agitation | 1 | 0 | ||
| GASTROINTESTINAL SYSTEM DISORDERS | ||||
| Nausea | 14 | 8 | ||
| Diarrhea | 10 | 4 | ||
| Abdominal pain | 8 | 4 | ||
| Constipation | 6 | 4 | ||
| Vomiting | 4 | 1 | ||
| Mouth dry | 3 | 0 | ||
| Dyspepsia | 2 | 1 | ||
| Flatulence | 2 | 0 | ||
| Gastritis | 1 | 0 | ||
| Gastrointestinal disorders | 1 | 0 | ||
| RESPIRATORY SYSTEM DISORDERS | ||||
| Dyspnea | 3 | 1 | ||
| PLATELET, BLEEDING AND CLOTTING DISORDERS | ||||
| Purpura | 2 | 1 | ||
| URINARY SYSTEM DISORDERS | ||||
| Urine discoloration | 10 | 0 | ||
| BODY AS A WHOLE - GENERAL DISORDERS | ||||
| Back pain | 4 | 2 | ||
| Fatigue | 6 | 4 | ||
| Asthenia | 2 | 1 | ||
| RESISTANCE MECHANISM DISORDERS | ||||
| Infection bacterial | 1 | 0 | ||
Effects of Gender and Age on Adverse Reactions No differences were noted in the rate of adverse events attributable to entacapone by age or gender. Postmarketing Reports
The following spontaneous reports of adverse events temporally associated with entacapone have been identified since market introduction and are not listed in Table 4. Because these reactions are reported voluntarily from a population of unknown size, it is not always possible to reliably estimate their frequency or establish causal relationship to entacapone exposure.
Hepatitis with mainly cholestatic features has been reported.
Drug Interactions
In vitro studies of human CYP enzymes showed that entacapone inhibited the CYP enzymes 1A2, 2A6, 2C9, 2C19, 2D6, 2E1 and 3A only at very high concentrations (IC50 from 200 microM to over 1,000 microM; an oral 200 mg dose achieves a highest level of approximately 5 microM in people); these enzymes would therefore not be expected to be inhibited in clinical use.
In an interaction study in healthy volunteers, entacapone did not significantly change the plasma levels of S-warfarin while the AUC for R-warfarin increased on average by 18% [Cl90 11% to 26%], and the INR values increased on average by 13% [Cl90 6% to 19%]. Nevertheless, cases of significantly increased INR in patients concomitantly using warfarin have been reported during the postapproval use of entacapone. Therefore, monitoring of INR is recommended when entacapone treatment is initiated or when the dose is increased for patients receiving warfarin.
Protein Binding
Entacapone is highly protein bound (98%). In vitro studies have shown no binding displacement between entacapone and other highly bound drugs, such as warfarin, salicylic acid, phenylbutazone, and diazepam.
Drugs Metabolized by Catechol- O -Methyltransferase (COMT)
See WARNINGS.
Hormone Levels
Levodopa is known to depress prolactin secretion and increase growth hormone levels. Treatment with entacapone coadministered with levodopa and dopa decarboxylase inhibitor does not change these effects.
Effect of Entacapone on the Metabolism of Other Drugs
See WARNINGS regarding concomitant use of entacapone and non-selective MAO inhibitors.
No interaction was noted with the MAO-B inhibitor selegiline in two multiple-dose interaction studies when entacapone was coadministered with a levodopa and dopa decarboxylase inhibitor (n=29). More than 600 patients with Parkinson’s disease in clinical studies have used selegiline in combination with entacapone and levodopa and dopa decarboxylase inhibitor.
As most entacapone excretion is via the bile, caution should be exercised when drugs known to interfere with biliary excretion, glucuronidation, and intestinal beta-glucuronidase are given concurrently with entacapone. These include probenecid, cholestyramine, and some antibiotics (e.g., erythromycin, rifampicin, ampicillin, and chloramphenicol).
No interaction with the tricyclic antidepressant imipramine was shown in a single-dose study with entacapone without coadministered levodopa and dopa-decarboxylase inhibitor.
DESCRIPTION
Entacapone is available as tablets containing 200 mg entacapone, USP.
Entacapone is an inhibitor of catechol-O-methyltransferase (COMT), used in the treatment of Parkinson’s disease as an adjunct to levodopa and carbidopa therapy. It is a nitrocatechol-structured compound with a relative molecular mass of 305.29. The chemical name of entacapone is (E)-2-cyano3-(3,4-dihydroxy-5-nitrophenyl)-N,N-diethyl-2-propenamide. Its empirical formula is C 14 H 15 N 3 O 5 and its structural formula is: 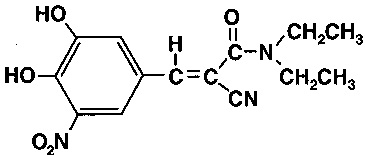
The inactive ingredients of the entacapone tablets, USP are microcrystalline cellulose, mannitol, croscarmellose sodium, colloidal silicon dioxide, magnesium stearate, hydrogenated vegetable oil, hypromellose, glycerin, titanium dioxide, iron oxide yellow, sucrose, polysorbate 80 and iron oxide red.
FDA approved dissolution testing specifications differ from USP.
CLINICAL PHARMACOLOGY
Mechanism of Action
Entacapone is a selective and reversible inhibitor of COMT. In mammals, COMT is distributed throughout various organs with the highest activities in the liver and kidney. COMT also occurs in the heart, lung, smooth and skeletal muscles, intestinal tract, reproductive organs, various glands, adipose tissue, skin, blood cells, and neuronal tissues, especially in glial cells. COMT catalyzes the transfer of the methyl group of S-adenosyl-L-methionine to the phenolic group of substrates that contain a catechol structure. Physiological substrates of COMT include dopa, catecholamines (dopamine, norepinephrine, and epinephrine) and their hydroxylated metabolites. The function of COMT is the elimination of biologically active catechols and some other hydroxylated metabolites. In the presence of a decarboxylase inhibitor, COMT becomes the major metabolizing enzyme for levodopa, catalyzing the metabolism to 3-methoxy-4-hydroxy-L phenylalanine (3-OMD) in the brain and periphery.
The mechanism of action of entacapone is believed to be through its ability to inhibit COMT and alter the plasma pharmacokinetics of levodopa. When entacapone is given in conjunction with levodopa and an aromatic amino acid decarboxylase inhibitor, such as carbidopa, plasma levels of levodopa are greater and more sustained than after administration of levodopa and an aromatic amino acid decarboxylase inhibitor alone. It is believed that at a given frequency of levodopa administration, these more sustained plasma levels of levodopa result in more constant dopaminergic stimulation in the brain, leading to greater effects on the signs and symptoms of Parkinson’s disease. The higher levodopa levels also lead to increased levodopa adverse effects, sometimes requiring a decrease in the dose of levodopa.
In animals, while entacapone enters the central nervous system (CNS) to a minimal extent, it has been shown to inhibit central COMT activity. In humans, entacapone inhibits the COMT enzyme in peripheral tissues. The effects of entacapone on central COMT activity in humans have not been studied.
Pharmacodynamics COMT Activity in Erythrocytes: Studies in healthy volunteers have shown that entacapone reversibly inhibits human erythrocyte COMT activity after oral administration. There was a linear correlation between entacapone dose and erythrocyte COMT inhibition, the maximum inhibition being 82% following an 800 mg single dose. With a 200 mg single dose of entacapone, maximum inhibition of erythrocyte COMT activity is on average 65% with a return to baseline level within 8 hours. Effect on the Pharmacokinetics of Levodopa and its Metabolites When 200 mg entacapone is administered together with levodopa and carbidopa, it increases the area under the curve (AUC) of levodopa by approximately 35% and the elimination half-life of levodopa is prolonged from 1.3 hours to 2.4 hours. In general, the average peak levodopa plasma concentration and the time of its occurrence (T max of 1 hour) are unaffected. The onset of effect occurs after the first administration and is maintained during long-term treatment. Studies in Parkinson’s disease patients suggest that the maximal effect occurs with 200 mg entacapone. Plasma levels of 3-OMD are markedly and dose-dependently decreased by entacapone when given with levodopa and carbidopa. Pharmacokinetics of Entacapone Entacapone pharmacokinetics are linear over the dose range of 5 mg to 800 mg, and are independent of levodopa and carbidopa coadministration. The elimination of entacapone is biphasic, with an elimination half-life of 0.4 hour to 0.7 hour based on the β-phase and 2.4 hours based on the γ-phase. The γ-phase accounts for approximately 10% of the total AUC. The total body clearance after intravenous administration is 850 mL per min. After a single 200 mg dose of entacapone, the C max is approximately 1.2 mcg per mL.
Absorption : Entacapone is rapidly absorbed, with a T max of approximately 1 hour. The absolute bioavailability following oral administration is 35%. Food does not affect the pharmacokinetics of entacapone.
Distribution : The volume of distribution of entacapone at steady state after intravenous injection is small (20 L). Entacapone does not distribute widely into tissues due to its high plasma protein binding. Based on in vitro studies, the plasma protein binding of entacapone is 98% over the concentration range of 0.4 mcg per mL to 50 mcg per mL. Entacapone binds mainly to serum albumin.
Metabolism and Elimination : Entacapone is almost completely metabolized prior to excretion, with only a very small amount (0.2% of dose) found unchanged in urine. The main metabolic pathway is isomerization to the cis -isomer, followed by direct glucuronidation of the parent and cis -isomer; the glucuronide conjugate is inactive. After oral administration of a 14 C-labeled dose of entacapone, 10% of labeled parent and metabolite is excreted in urine and 90% in feces.
Special Populations : Entacapone pharmacokinetics are independent of age. No formal gender studies have been conducted. Racial representation in clinical studies was largely limited to Caucasians; therefore, no conclusions can be reached about the effect of entacapone on groups other than Caucasian.
Hepatic Impairment : A single 200 mg dose of entacapone, without levodopa and dopa decarboxylase inhibitor coadministration, showed approximately 2-fold higher AUC and C max values in patients with a history of alcoholism and hepatic impairment (n=10) compared to normal subjects (n=10). All patients had biopsy-proven liver cirrhosis caused by alcohol. According to Child-Pugh grading seven patients with liver disease had mild hepatic impairment and three patients had moderate hepatic impairment. As only about 10% of the entacapone dose is excreted in urine as parent compound and conjugated glucuronide, biliary excretion appears to be the major route of excretion of this drug. Consequently, entacapone should be administered with care to patients with biliary obstruction.
Renal Impairment : The pharmacokinetics of entacapone have been investigated after a single 200 mg entacapone dose, without levodopa and dopa decarboxylase inhibitor coadministration, in a specific renal impairment study. There were three groups: normal subjects (n=7; creatinine clearance greater than 1.12 mL per sec per 1.73 m 2 ), moderate impairment (n=10; creatinine clearance ranging from 0.60 mL per sec per 1.73 m 2 to 0.89 mL per sec per 1.73 m 2 ), and severe impairment (n=7; creatinine clearance ranging from 0.2 mL per sec per 1.73 m 2 to 0.44 mL per sec per 1.73 m 2 ). No important effects of renal function on the pharmacokinetics of entacapone were found.
Drug Interactions : See PRECAUTIONS, Drug Interactions.
Clinical Studies The effectiveness of entacapone as an adjunct to levodopa in the treatment of Parkinson’s disease was established in three 24-week multicenter, randomized, double-blind, placebo-controlled studies in patients with Parkinson’s disease. In two of these studies, patients had motor “fluctuations”, characterized by documented periods of “On” (periods of relatively good functioning) and “Off” (periods of relatively poor functioning), despite optimum levodopa therapy. There was also a withdrawal period following 6 months of treatment. In the third study, patients were not required to have motor fluctuations. Prior to the controlled part of the studies, patients were stabilized on levodopa for 2 weeks to 4 weeks. Entacapone has not been systematically evaluated in patients who have Parkinson’s disease without motor fluctuations.
In the first two studies to be described, patients were randomized to receive placebo or entacapone 200 mg administered concomitantly with each dose of levodopa and carbidopa (up to 10 times daily, but averaging 4 doses to 6 doses per day). The formal double-blind portion of both studies was 6 months long. Patients recorded the time spent in the “On” and “Off” states in home diaries periodically throughout the duration of the study. In one study, conducted in the Nordic countries, the primary outcome measure was the total mean time spent in the “On” state during an 18-hour diary recorded day (6 AM to midnight). In the other study, the primary outcome measure was the proportion of awake time spent over 24 hours in the “On” state.
In addition to the primary outcome measure: the amount of time spent in the “Off” state, subparts of the Unified Parkinson’s Disease Rating Scale (UPDRS) including mentation (Part I), activities of daily living (ADL) (Part II), motor function (Part III), complications of therapy (Part IV), and disease staging (Part V and VI) were assessed. Additional secondary endpoints included the investigator’s and patient’s global assessment of clinical condition, a 7-point subjective scale designed to assess global functioning in Parkinson’s disease; and the change in daily levodopa and carbidopa dose.
In one of the studies, 171 patients were randomized in 16 centers in Finland, Norway, Sweden, and Denmark (Nordic study), all of whom received concomitant levodopa plus dopa-decarboxylase inhibitor (either levodopa and carbidopa or levodopa and benserazide). In the second study, 205 patients were randomized in 17 centers in North America (US and Canada); all patients received concomitant levodopa and carbidopa.
The following tables display the results of these two studies:
Table 1. Nordic Study
| Primary Measure from Home Diary (from an 18-hour Diary Day) | |||
| Baseline | Change from Baseline at Month 6 • | p-value vs. placebo | |
| Hours of Awake Time “On” | |||
| Placebo | 9.2 | +0.1 | – |
| Entacapone | 9.3 | +1.5 | less than 0.001 |
| Duration of “On” time after first AM dose (hrs) | |||
| Placebo | 2.2 | 0 | – |
| Entacapone | 2.1 | +0.2 | less than 0.05 |
| Secondary Measures from Home Diary (from an 18-hour Diary Day) ‡‡ | |||
| Hours of Awake Time “Off” | |||
| Placebo | 5.3 | 0 | – |
| Entacapone | 5.5 | -1.3 | less than 0.001 |
| Proportion of Awake Time “On” ••• (%) | |||
| Placebo | 63.8 | +0.6 | – |
| Entacapone | 62.7 | +9.3 | less than 0.001 |
| Levodopa Total Daily Dose (mg) | |||
| Placebo | 705 | +14 | – |
| Entacapone | 701 | -87 | less than 0.001 |
| Frequency of Levodopa Daily Intakes | |||
| Placebo | 6.1 | +0.1 | – |
| Entacapone | 6.2 | -0.4 | less than 0.001 |
| Other Secondary Measures ‡‡ | |||
| Baseline | Change from Baseline at Month 6 • | p-value vs. placebo | |
| Investigator’s Global (overall) % Improved •• | |||
| Placebo | – | 28 | – |
| Entacapone | – | 56 | less than 0.01 |
| Patient’s Global (overall) % Improved •• | |||
| Placebo | – | 22 | – |
| Entacapone | – | 39 | N.S. ‡ |
| UPDRS Total | |||
| Placebo | 37.4 | -1.1 | – |
| Entacapone | 38.5 | -4.8 | less than 0.01 |
| UPDRS Motor | |||
| Placebo | 24.6 | -0.7 | – |
| Entacapone | 25.5 | -3.3 | less than 0.05 |
| UPDRS ADL | |||
| Placebo | 11 | -0.4 | – |
| Entacapone | 11.2 | -1.8 | less than 0.05 |
• Mean; the month 6 values represent the average of weeks 8, 16, and 24, by protocol-defined outcome measure, except for Investigator’s and Patient’s Global Improvement.
•• At least one category change at endpoint.
••• Not an endpoint for this study but primary endpoint in the North American Study.
‡ Not significant.
‡‡ P values for Secondary Measures and Other Secondary Measures are nominal P values without any adjustment for multiplicity.
Table 2. North American Study
| Primary Measure from Home Diary (for a 24-hour Diary Day) | |||
| Baseline | Change from Baseline at Month 6 • | p-value vs. placebo | |
| Percent of Awake Time “On” | |||
| Placebo | 60.8 | +2 | – |
| Entacapone | 60 | +6.7 | less than 0.05 |
| Secondary Measures from Home Diary (for a 24-hour Diary Day) ‡‡ | |||
| Hours of Awake Time “Off” | |||
| Placebo | 6.6 | -0.3 | – |
| Entacapone | 6.8 | -1.2 | less than 0.01 |
| Hours of Awake Time “On” | |||
| Placebo | 10.3 | +0.4 | – |
| Entacapone | 10.2 | +1 | N.S. ‡ |
| Levodopa Total Daily Dose (mg) | |||
| Placebo | 758 | +19 | – |
| Entacapone | 804 | -93 | less than 0.001 |
| Frequency of Levodopa Daily Intakes | |||
| Placebo | 6 | +0.2 | – |
| Entacapone | 6.2 | 0 | N.S. ‡ |
| Other Secondary Measures ‡‡ | |||
| Baseline | Change from Baseline at Month 6 • | p-value vs. placebo | |
| Investigator’s Global (overall) % Improved •• | |||
| Placebo | – | 21 | – |
| Entacapone | – | 34 | less than 0.05 |
| Patient’s Global (overall) % Improved •• | |||
| Placebo | – | 20 | – |
| Entacapone | – | 31 | less than 0.05 |
| UPDRS Total ••• | |||
| Placebo | 35.6 | +2.8 | – |
| Entacapone | 35.1 | -0.6 | less than 0.05 |
| UPDRS Motor ••• | |||
| Placebo | 22.6 | +1.2 | – |
| Entacapone | 22 | -0.9 | less than 0.05 |
| UPDRS ADL ••• | |||
| Placebo | 11.7 | +1.1 | – |
| Entacapone | 11.9 | 0 | less than 0.05 |
• Mean; the month 6 values represent the average of weeks 8, 16, and 24, by protocol-defined outcome measure, except for Investigator’s and Patient’s Global Improvement.
•• At least one category change at endpoint.
••• Score change at endpoint similarly to the Nordic Study.
‡ Not significant.
‡‡ P values for Secondary Measures and Other Secondary Measures are nominal P values without any adjustment for multiplicity.
Effects on “On” time did not differ by age, sex, weight, disease severity at baseline, levodopa dose and concurrent treatment with dopamine agonists or selegiline.
Withdrawal of entacapone : In the North American study, abrupt withdrawal of entacapone, without alteration of the dose of levodopa and carbidopa, resulted in a significant worsening of fluctuations, compared to placebo. In some cases, symptoms were slightly worse than at baseline, but returned to approximately baseline severity within two weeks following levodopa dose increase on average by 80 mg. In the Nordic study, similarly, a significant worsening of parkinsonian symptoms was observed after entacapone withdrawal, as assessed two weeks after drug withdrawal. At this phase, the symptoms were approximately at baseline severity following levodopa dose increase by about 50 mg.
In the third placebo-controlled study, a total of 301 patients were randomized in 32 centers in Germany and Austria. In this study, as in the other two studies, entacapone 200 mg was administered with each dose of levodopa and dopa decarboxylase inhibitor (up to 10 times daily) and UPDRS Parts II and III and total daily “On” time were the primary measures of effectiveness. The following results were observed for the primary measures, as well as for some secondary measures:
Table 3. German-Austrian Study
| Primary Measures | |||
| Baseline | Change from Baseline at Month 6 | p-value vs. placebo (LOCF) | |
| UPDRS ADL • | |||
| Placebo | 12 | +0.5 | – |
| Entacapone | 12.4 | -0.4 | less than 0.05 |
| UPDRS Motor • | |||
| Placebo | 24.1 | +0.1 | – |
| Entacapone | 24.9 | -2.5 | less than 0.05 |
| Hours of Awake Time “On” (Home diary) •• | |||
| Placebo | 10.1 | +0.5 | – |
| Entacapone | 10.2 | +1.1 | N.S. ‡ |
| Secondary Measures ‡‡ | |||
| Baseline | Change from Baseline at Month 6 | p-value vs. placebo | |
| UPDRS Total • | |||
| Placebo | 37.7 | +0.6 | – |
| Entacapone | 39 | -3.4 | less than 0.05 |
| Percent of Awake Time “On” (Home diary) •• | |||
| Placebo | 59.8 | +3.5 | – |
| Entacapone | 62 | +6.5 | N.S. ‡ |
| Hours of Awake Time “Off” (Home diary) •• | |||
| Placebo | 6.8 | -0.6 | – |
| Entacapone | 6.3 | -1.2 | 0.07 |
| Levodopa Total Daily Dose (mg) • | |||
| Placebo | 572 | +4 | – |
| Entacapone | 566 | -35 | N.S. ‡ |
| Frequency of Levodopa Daily Intake • | |||
| Placebo | 5.6 | +0.2 | – |
| Entacapone | 5.4 | 0 | less than 0.01 |
| Global (overall) % Improved ••• | |||
| Placebo | – | 34 | – |
| Entacapone | – | 38 | N.S. ‡ |
• Total population; score change at endpoint.
•• Fluctuating population, with 5 doses to 10 doses; score change at endpoint.
••• Total population; at least one category change at endpoint.
‡ Not significant.
‡‡ P values for Secondary Measures are nominal P values without any adjustment for multiplicity.
HOW SUPPLIED
Entacapone Tablets USP, 200 mg are brownish-orange colored, oval-shaped unscored film coated tablets debossed with “L 633” on one side and plain on the other side. Tablets are provided in HDPE containers as follows:
NDC 46708-478-31 bottle of 100 tablets
NDC 46708-478-91 bottle of 1,000 tablets
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature.]
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Manufactured by: Alembic Pharmaceuticals Limited (Formulation Division), Village Panelav, P. O. Tajpura, Near Baska, Taluka-Halol, Panchmahal 389350, Gujarat, India.
Revised: 09/2021