Get your patient on Ecoza - Econazole Nitrate aerosol, Foam (Econazole Nitrate)
Ecoza - Econazole Nitrate aerosol, Foam prescribing information
INDICATIONS AND USAGE
Ecoza (econazole nitrate) topical foam, 1%, is indicated for the treatment of interdigital tinea pedis caused by Trichophyton rubrum, Trichophyton mentagrophytes, and Epidermophyton floccosum in patients 12 years of age and older.
DOSAGE AND ADMINISTRATION
Ecoza topical foam, 1% is for topical use only. Ecoza topical foam, 1% is not for oral, ophthalmic, or intravaginal use.
Ecoza topical foam, 1% should be applied to cover affected areas once daily for 4 weeks.
DOSAGE FORMS AND STRENGTHS
Foam, 1%. Each gram of Ecoza topical foam, 1%, contains 10 mg of econazole nitrate in a white to off-white foam.
USE IN SPECIFIC POPULATIONS
Pregnancy
Pregnancy Category C
There are no adequate and well-controlled trials with Ecoza topical foam in pregnant women. Ecoza topical foam should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Econazole nitrate has not been shown to be teratogenic when administered orally to mice, rabbits or rats. Fetotoxic or embryotoxic effects were observed in Segment I oral studies with rats receiving 10 to 40 times the human dermal dose. Similar effects were observed in Segment II or Segment III studies with mice, rabbits and/or rats receiving oral doses 80 or 40 times the human dermal dose.
Nursing Mothers
It is not known whether econazole nitrate is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when econazole nitrate is administered to a nursing woman. Following oral administration of econazole nitrate to lactating rats, econazole and/or metabolites were excreted in milk and were found in nursing pups.
Pediatric Use
Of the 173 subjects treated with Ecoza topical foam, 1% in the clinical trials, 2 subjects were 12 to 17 years old. In a pediatric maximal use trial, Ecoza topical foam, 1% was applied once daily to eighteen subjects aged 12 to 17 years with interdigital tinea pedis for 28 days [ see Clinical Pharmacology (12.3) ]. The safety findings for subjects 12 to 17 years were similar to those in adult population.
Geriatric Use
Of the 173 subjects treated with Ecoza topical foam, 1% in the adult clinical trials, 6 subjects were 65 years or older. No overall differences in safety or effectiveness were observed between these subjects and younger subjects.
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
Contents are flammable. Instruct the patient to avoid heat, flame, and/or smoking during and immediately following application. (5.1 )
Flammability
Ecoza topical foam is flammable. Avoid heat, flame, and smoking during and immediately following application. Contents under pressure. Do not puncture and/or incinerate the containers. Do not expose containers to heat and/or store at temperatures above 120°F (49°C) even when empty. Do not store in direct sunlight.
ADVERSE REACTIONS
During clinical trials with Ecoza topical foam, the most common adverse reactions were application site reactions which occurred in less than 1% of subjects in both the Ecoza and vehicle arms. (6.1 )
To report SUSPECTED ADVERSE REACTIONS, contact Resilia Pharmaceuticals at 1-888-998-0770 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In two double-blind, vehicle-controlled clinical trials, 495 subjects were exposed to Ecoza topical foam or vehicle (246 subjects were exposed to Ecoza topical foam, 1% and 249 were exposed to vehicle). Subjects with interdigital tinea pedis applied foam or vehicle once daily for approximately 28 days. During clinical trials with Ecoza topical foam, the most common adverse reactions were application site reactions which occurred in less than 1% of subjects in both the Ecoza and vehicle arms.
DRUG INTERACTIONS
Warfarin
Concomitant administration of econazole and warfarin has resulted in enhancement of anticoagulant effect. Most cases reported product application with use under occlusion, genital application, or application to a large body surface area which may increase the systemic absorption of econazole nitrate. Monitoring of International Normalized Ratio (INR) and/or prothrombin time may be indicated especially for patients who apply econazole to large body surface areas, in the genital area, or under occlusion.
DESCRIPTION
Ecoza (econazole nitrate) topical foam, 1% contains the azole antifungal agent, econazole nitrate in an oil-in-water emulsion base consisting of the following inactive ingredients: dimethicone, glycerin, polysorbate 20, povidone, propylene glycol, stearic acid, trolamine, purified water and butane as a propellant. Each gram of Ecoza topical foam, 1% contains 10 mg of econazole nitrate, USP, in a white to off-white foam. Ecoza topical foam, 1% is alcohol (ethanol)-free and for topical use only.
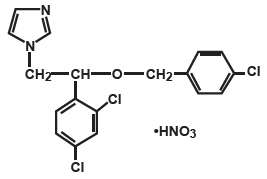
Chemically, econazole nitrate is 1-[2-{(4-chloro-phenyl)methoxy}-2-(2,4-dichlorophenyl) ethyl]-1H-imidazole mononitrate. Econazole nitrate has the molecular formula C 18 H 15 Cl 3 N 2 O. HNO 3 and a molecular weight of 444.70. Its molecular structure is as follows:
CLINICAL PHARMACOLOGY
Mechanism of Action
Ecoza topical foam is an azole antifungal [ see Clinical Pharmacology (12.4) ] .
Pharmacodynamics
The pharmacodynamics of Ecoza topical foam, 1% have not been established .
Pharmacokinetics
The systemic absorption of Ecoza topical foam, 1% following topical application was studied in one clinical trial in adults and one clinical study in pediatric subjects.
In the adult trial, 19 subjects (male and female) with tinea pedis applied Ecoza topical foam, 1% once daily for 29 days. Subjects applied a mean daily amount of 2.4 g of Ecoza topical foam, 1% to soles, toes, interdigital spaces and tops of both feet up to the ankles. Blood samples were obtained on Day 29 at pre-dose and 1, 2, 4, 6, 8, and 12 hours after application. Results (mean ± SD) showed the time to reach peak plasma concentrations (T max ) was 6.8 ± 5.1 h with maximum concentration (C max ) of 417 ± 218 pg/ml. The area under the concentration time curve for the first 12 hours post application on Day 29 (AUC (0-12) ) was 3440 ± 1920 pg-h/ml.
In the pediatric trial, 18 subjects (male and female ages 12 - 17) with interdigital tinea pedis and positive fungal cultures were treated with Ecoza topical foam, 1% once daily for 4 weeks. Subjects applied a mean daily amount of 3.2 g of Ecoza topical foam, 1% to soles, toes, interdigital spaces and tops of both feet up to the ankles. Blood samples were obtained on Day 28 at pre-dose and 7 h and 11 h post-dose. The mean ± SD econazole plasma concentration was 397 ± 289, 534 ± 745 and 575 ± 638 pg/mL at pre-dose and 7 h and 11 h post-dose, respectively.
Microbiology
Mechanism of Action
Econazole nitrate, an azole antifungal agent, inhibits fungal cytochrome P-450-mediated 14 alpha-lanosterol demethylase enzyme. This enzyme functions to convert lanosterol to ergosterol. The accumulation of 14 alpha-methyl sterols correlates with the subsequent loss of ergosterol in the fungal cell wall and may be responsible for the fungistatic activity of econazole. Mammalian cell demethylation is less sensitive to econazole inhibition.
Activity in vitro and in clinical infections
Econazole nitrate has been shown to be active against most strains of the following microorganisms, both in vitro and in clinical infections [ see Indications and Usage (1) ].
Trichophyton rubrum
Epidermophyton floccosum
Trichophyton mentagrophytes
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies to determine the carcinogenic potential of Ecoza topical foam have not been performed.
Oral administration of econazole nitrate in rats has been reported to produce prolonged gestation.
CLINICAL STUDIES
In two multi-center, randomized, double-blind, vehicle-controlled clinical trials a total of 505 subjects with interdigital tinea pedis were randomized 1:1 to Ecoza topical foam or vehicle; subjects applied the assigned medication once daily for 4 weeks. The severity of erythema, scaling, fissuring, maceration, vesiculation, and pruritus were graded using a 4-point scale (none, mild, moderate, severe). Subjects had KOH examination and fungal cultures taken to confirm eligibility. A total of 339 subjects with positive fungal cultures were evaluated for efficacy. Efficacy was evaluated on Day 43, 2 weeks post-treatment with treatment success being defined as complete cure (negative KOH and fungal culture and no evidence of clinical disease). The study population ranged in age from 12 to 71 years with 5 subjects less than 18 years of age at baseline. The subjects were 71% male and 51% Caucasian. Table 1 presents the efficacy results for each trial.
Table 1: Efficacy Results at Two Weeks Post-treatment (Day 43) Complete Cure, Effective Treatment and Mycological Cure | ||||
| Trial 1 | Trial 2 | |||
Ecoza topical foam 1% N = 82 n(%) | Foam Vehicle N = 83 n(%) | Ecoza topical foam, 1% N = 91 n(%) | Foam Vehicle N = 83 n(%) | |
| Complete cure a | 19 (23.2%) | 2 (2.4%) | 23 (25.3%) | 4 (4.8%) |
| Effective treatment b | 40 (48.8%) | 9 (10.8%) | 44 (48.4%) | 9 (10.8%) |
| Mycological cure c | 56 (68.3%) | 13 (15.7%) | 61 (67.0%) | 15 (18.1%) |
a Mycological cure and an absence of clinical signs and symptoms (erythema, scaling, fissuring, maceration, vesiculation, or pruritus). b Mycological cure and no or mild erythema and/or scaling with all other signs and symptoms absent. c Negative KOH and fungal culture.
HOW SUPPLIED/ STORAGE AND HANDLING
Ecoza topical foam, 1% is white to off-white foam supplied in 10g (NDC 81811-100-10) and 70g (NDC 81811-100-70) aluminum pressurized canister.
Store at controlled room temperature 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C and 30°C (59°F and 86°F). Do not refrigerate or freeze.
Ecoza topical foam is flammable. Avoid heat, flame, and smoking during and immediately following application.
Contents under pressure. Do not puncture and/or incinerate the containers.
Do not expose containers to heat and/or store at temperatures above 120°F (49°C) even when empty.
Do not store in direct sunlight.
Instructions for Use ECOZA ® (ee-ko-zah) (econazole nitrate) topical foam, 1%
| Important information: Ecoza topical foam is for use on skin only. Do not use Ecoza topical foam in your eyes or vagina. |
Parts of Ecoza topical foam Canister. (See Figure A )
 |
| Figure A |
How to apply Ecoza topical foam:
| Step 1: | Before you apply Ecoza topical foam, shake the Ecoza topical foam canister for about 5 seconds. |
| Step 2: | Remove the cap and turn the Ecoza topical foam canister upside down over the palm of your hand. |
| Step 3: | Press down firmly on the actuator until there is a small amount of foam about the size of a golf ball in the palm of your hand. (See Figures B and C ) |
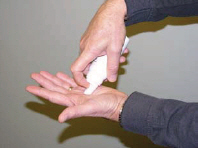 |  |
| Figure B | Figure C |
| Step 4: | Use your finger-tips to scoop up small amounts of Ecoza topical foam and apply to the affected skin areas on your feet. Gently rub the foam into the skin. (See Figure D ) |
| |
| Figure D | |
| Step 5: | You should apply Ecoza topical foam to your toes, to the spaces between your toes, and to the surrounding areas 1 time a day for 4 weeks or as prescribed by your doctor. |
| Step 6: | Replace the cap. Wash your hands after applying Ecoza topical foam. |
How should I store Ecoza topical foam?
- Store Ecoza topical foam at room temperature, between 68°F to 77°F (20°C to 25°C).
- Do not refrigerate or freeze Ecoza topical foam.
- Do not store Ecoza topical foam in direct sunlight.
- Ecoza topical foam is flammable. Keep the Ecoza topical foam canister away from heat and temperatures above 120°F (49°C), even if the canister is empty.
- Do not puncture or burn the Ecoza topical foam canister.
Keep Ecoza topical foam and all medicines out of the reach of children.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured for Resilia Pharmaceuticals, Inc., Atlanta, GA 30328
Issued: 12/2023
Mechanism of Action
Ecoza topical foam is an azole antifungal [ see Clinical Pharmacology (12.4) ] .