Get your patient on Dyrenium - Triamterene capsule (Triamterene)
Dyrenium - Triamterene capsule prescribing information
Warnings
Abnormal elevation of serum potassium levels (greater than or equal to 5.5 mEq/liter) can occur with all potassium-sparing agents, including Dyrenium. Hyperkalemia is more likely to occur in patients with renal impairment and diabetes (even without evidence of renal impairment), and in the elderly or severely ill. Since uncorrected hyperkalemia may be fatal, serum potassium levels must be monitored at frequent intervals especially in patients receiving Dyrenium, when dosages are changed or with any illness that may influence renal function.
INDICATIONS AND USAGE
Dyrenium (triamterene) is indicated in the treatment of edema associated with congestive heart failure, cirrhosis of the liver and the nephrotic syndrome; steroid-induced edema, idiopathic edema and edema due to secondary hyperaldosteronism.
Dyrenium may be used alone or with other diuretics, either for its added diuretic effect or its potassium-sparing potential. It also promotes increased diuresis when patients prove resistant or only partially responsive to thiazides or other diuretics because of secondary hyperaldosteronism.
Usage in Pregnancy . The routine use of diuretics in an otherwise healthy woman is inappropriate and exposes mother and fetus to unnecessary hazard. Diuretics do not prevent development of toxemia of pregnancy, and there is no satisfactory evidence that they are useful in the treatment of developed toxemia.
Edema during pregnancy may arise from pathological causes or from the physiologic and mechanical consequences of pregnancy. Diuretics are indicated in pregnancy (however, see PRECAUTIONS below) when edema is due to pathologic causes, just as they are in the absence of pregnancy. Dependent edema in pregnancy, resulting from restriction of venous return by the expanded uterus, is properly treated through elevation of the lower extremities and use of support hose; use of diuretics to lower intravascular volume in this case is illogical and unnecessary. There is hypervolemia during normal pregnancy which is harmful to neither the fetus nor the mother (in the absence of cardiovascular disease), but which is associated with edema, including generalized edema, in the majority of pregnant women. If this edema produces discomfort, increased recumbency will often provide relief. In rare instances, this edema may cause extreme discomfort which is not relieved by rest. In these cases, a short course of diuretics may provide relief and may be appropriate.
DOSAGE AND ADMINISTRATION
Adult Dosage
Dosage should be titrated to the needs of the individual patient. When used alone, the usual starting dose is 100 mg twice daily after meals. When combined with another diuretic or antihypertensive agent, the total daily dosage of each agent should usually be lowered initially and then adjusted to the patient’s needs. The total daily dosage should not exceed 300 mg. Please refer to PRECAUTIONS−General.
When Dyrenium (triamterene) is added to other diuretic therapy or when patients are switched to Dyrenium from other diuretics, all potassium supplementation should be discontinued.
CONTRAINDICATIONS
Anuria. Severe or progressive kidney disease or dysfunction, with the possible exception of nephrosis. Severe hepatic disease. Hypersensitivity to the drug or any of its components.
Dyrenium (triamterene) should not be used in patients with pre-existing elevated serum potassium, as is sometimes seen in patients with impaired renal function or azotemia, or in patients who develop hyperkalemia while on the drug. Patients should not be placed on dietary potassium supplements, potassium salts or potassium-containing salt substitutes in conjunction with Dyrenium.
Dyrenium should not be given to patients receiving other potassium-sparing agents, such as spironolactone, amiloride hydrochloride, or other formulations containing triamterene. Two deaths have been reported in patients receiving concomitant spironolactone and Dyrenium or Dyazide ® . Although dosage recommendations were exceeded in one case and in the other serum electrolytes were not properly monitored, these two drugs should not be given concomitantly.
WARNINGS
Abnormal elevation of serum potassium levels (greater than or equal to 5.5 mEq/liter) can occur with all potassium-sparing agents, including Dyrenium. Hyperkalemia is more likely to occur in patients with renal impairment and diabetes (even without evidence of renal impairment), and in the elderly or severely ill. Since uncorrected hyperkalemia may be fatal, serum potassium levels must be monitored at frequent intervals especially in patients receiving Dyrenium, when dosages are changed or with any illness that may influence renal function.
There have been isolated reports of hypersensitivity reactions; therefore, patients should be observed regularly for the possible occurrence of blood dyscrasias, liver damage or other idiosyncratic reactions.
Periodic BUN and serum potassium determinations should be made to check kidney function, especially in patients with suspected or confirmed renal insufficiency. It is particularly important to make serum potassium determinations in elderly or diabetic patients receiving the drug; these patients should be observed carefully for possible serum potassium increases.
If hyperkalemia is present or suspected, an electrocardiogram should be obtained. If the ECG shows no widening of the QRS or arrhythmia in the presence of hyperkalemia, it is usually sufficient to discontinue Dyrenium (triamterene) and any potassium supplementation, and substitute a thiazide alone. Sodium polystyrene sulfonate may be administered to enhance the excretion of excess potassium. The presence of a widened QRS complex or arrhythmia in association with hyperkalemia requires prompt additional therapy. For tachyarrhythmia, infuse 44 mEq of sodium bicarbonate or 10 mL of 10% calcium gluconate or calcium chloride over several minutes. For asystole, bradycardia or A-V block transvenous pacing is also recommended.
The effect of calcium and sodium bicarbonate is transient and repeated administration may be required. When indicated by the clinical situation, excess K+ may be removed by dialysis or oral or rectal administration of Sodium polystyrene sulfonate. Infusion of glucose and insulin has also been used to treat hyperkalemia.
ADVERSE REACTIONS
Adverse effects are listed in decreasing order of frequency; however, the most serious adverse effects are listed first, regardless of frequency. All adverse effects occur rarely (that is, 1 in 1000, or less).
Hypersensitivity: anaphylaxis, rash, photosensitivity.
Metabolic: hyperkalemia, hypokalemia.
Renal: azotemia, elevated BUN and creatinine, renal stones, acute interstitial nephritis (rare), acute renal failure (one case of irreversible renal failure has been reported).
Gastrointestinal: jaundice and/or liver enzyme abnormalities, nausea and vomiting, diarrhea.
Hematologic: thrombocytopenia, megaloblastic anemia.
Central Nervous System: weakness, fatigue, dizziness, headache, dry mouth.
| To report SUSPECTED ADVERSE REACTIONS, contact Advanz Pharma (US) Corp. at 1-877-370-1142 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. |
Drug Interactions
Caution should be used when lithium and diuretics are used concomitantly because diuretic-induced sodium loss may reduce the renal clearance of lithium and increase serum lithium levels with risk of lithium toxicity. Patients receiving such combined therapy should have serum lithium levels monitored closely and the lithium dosage adjusted if necessary.
A possible interaction resulting in acute renal failure has been reported in a few subjects when indomethacin, a nonsteroidal anti-inflammatory agent, was given with triamterene. Caution is advised in administering nonsteroidal anti-inflammatory agents with triamterene.
The effects of the following drugs may be potentiated when given together with triamterene: antihypertensive medication, other diuretics, preanesthetic and anesthetic agents, skeletal muscle relaxants (non-depolarizing).
Potassium-sparing agents should be used with caution in conjunction with angiotensin-converting enzyme (ACE) inhibitors due to an increased risk of hyperkalemia.
The following agents, given together with triamterene, may promote serum potassium accumulation and possibly result in hyperkalemia because of the potassium-sparing nature of triamterene, especially in patients with renal insufficiency: blood from blood bank (may contain up to 30 mEq of potassium per liter of plasma or up to 65 mEq per liter of whole blood when stored for more than 10 days); low-salt milk (may contain up to 60 mEq of potassium per liter); potassium-containing medications (such as parenteral penicillin G potassium); salt substitutes (most contain substantial amounts of potassium).
Dyrenium (triamterene) may raise blood glucose levels; for adult-onset diabetes, dosage adjustments of hypoglycemic agents may be necessary during and/or after therapy; concurrent use with chlorpropamide may increase the risk of severe hyponatremia.
DESCRIPTION
Each capsule for oral use, with opaque red cap and body, contains Triamterene USP, 50 or 100 mg, and is imprinted with the product name, DYRENIUM, strength (50 mg or 100 mg) and WPC 002 (for the 50-mg strength) and WPC 003 (for the 100-mg strength). Inactive ingredients consist of D&C Red No. 33, FD&C Yellow No. 6, Gelatin NF, Lactose NF, Magnesium Stearate NF, and Titanium Dioxide USP.
Triamterene is 2,4,7-triamino-6-phenyl-pteridine:
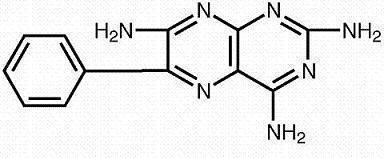
Its molecular weight is 253.27. At 50°C, triamterene is slightly soluble in water. It is soluble in dilute ammonia, dilute aqueous sodium hydroxide and dimethylformamide. It is sparingly soluble in methanol.
CLINICAL PHARMACOLOGY
Triamterene has a unique mode of action; it inhibits the reabsorption of sodium ions in exchange for potassium and hydrogen ions at that segment of the distal tubule under the control of adrenal mineralocorticoids (especially aldosterone). This activity is not directly related to aldosterone secretion or antagonism; it is a result of a direct effect on the renal tubule.
The fraction of filtered sodium reaching this distal tubular exchange site is relatively small, and the amount which is exchanged depends on the level of mineralocorticoid activity. Thus, the degree of natriuresis and diuresis produced by inhibition of the exchange mechanism is necessarily limited. Increasing the amount of available sodium and the level of mineralocorticoid activity by the use of more proximally acting diuretics will increase the degree of diuresis and potassium conservation.
Triamterene occasionally causes increases in serum potassium which can result in hyperkalemia. It does not produce alkalosis, because it does not cause excessive excretion of titratable acid and ammonium.
Triamterene has been shown to cross the placental barrier and appear in the cord blood of animals.
Pharmacokinetics
Onset of action is 2 to 4 hours after ingestion. In normal volunteers the mean peak serum levels were 30 ng/mL at 3 hours. The average percent of drug recovered in the urine (0 to 48 hours) was 21%. Triamterene is primarily metabolized to the sulfate conjugate of hydroxytriamterene. Both the plasma and urine levels of this metabolite greatly exceed triamterene levels. Triamterene is rapidly absorbed, with somewhat less than 50% of the oral dose reaching the urine. Most patients will respond to Dyrenium (triamterene) during the first day of treatment. Maximum therapeutic effect, however, may not be seen for several days. Duration of diuresis depends on several factors, especially renal function, but it generally tapers off 7 to 9 hours after administration.
HOW SUPPLIED
Capsules: 50 mg in bottles of 100, and 100 mg in bottles of 100.
Dyrenium - Triamterene capsule PubMed™ news
- Journal Article • 2025 NovCOMMD4 Drives Skin Cutaneous Melanoma Progression by Targeting PI3K-p85 to Activate PI3K-AKT.
- Journal Article • 2025 AugComputer aided greenness by design approach for resolving and quantifying Triamterene and Hydrochlorothiazide in pharmaceutical mixtures employing multiscale dynamics simulations.
- Journal Article • 2025 JunA Case of Thiazide-Induced Hyponatremia in an Elderly Patient.
- Journal Article • 2025 MayENaC inhibitors for the management of lithium related polyuria: a systematic review.
- Journal Article • 2025 MayHarnessing integrated bioinformatics to identify new diagnostic and therapeutic strategies for heart failure.