Doxycycline Hyclate - Doxycycline Hyclate tablet prescribing information
INDICATIONS AND USAGE
Doxycycline hyclate tablets, USP are indicated for use as an adjunct to scaling and root planing to promote attachment level gain and to reduce pocket depth in patients with adult periodontitis.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of doxycycline hyclate tablets and other antibacterial drugs, doxycycline hyclate tablets should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture susceptibility information is available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
DOSAGE AND ADMINISTRATION
THE DOSAGE OF DOXYCYCLINE HYCLATE TABLETS DIFFERS FROM THAT OF DOXYCYCLINE USED TO TREAT INFECTIONS. EXCEEDING THE RECOMMENDED DOSAGE MAY RESULT IN AN INCREASED INCIDENCE OF SIDE EFFECTS INCLUDING THE DEVELOPMENT OF RESISTANT MICROORGANISMS.
Doxycycline hyclate tablets 20 mg twice daily as an adjunct following scaling and root planing may be administered for up to 9 months. Doxycycline hyclate tablets should be taken twice daily at 12 hour intervals, usually in the morning and evening. It is recommended that if doxycycline hyclate tablets is taken close to meal times, allow at least one hour prior to or two hours after meals. Safety beyond 12 months and efficacy beyond 9 months have not been established.
Administration of adequate amounts of fluid along with the tablets is recommended to wash down the drug and reduce the risk of esophageal irritation and ulceration. (See ADVERSE REACTIONS Section).
CONTRAINDICATIONS
This drug is contraindicated in persons who have shown hypersensitivity to doxycycline or any of the other tetracyclines.
ADVERSE REACTIONS
Adverse Reactions in Clinical Trials of a bioequivalent form of doxycycline hyclate capsules : In clinical trials of adult patients with periodontal disease 213 patients received 20 mg BID over a 9 to 12 month period. The most frequent adverse reactions occurring in studies involving treatment with a bioequivalent form of doxycycline hyclate capsules or placebo are listed below:
Incidence (%) of Adverse Reactions in Clinical Trials of Doxycycline Hyclate Capsules, 20 mg (Bioequivalent to Doxycycline Hyclate Tablets, 20 mg) vs. Placebo
Adverse Reactions | Doxycycline Hyclate Capsules 20 mg BID (n=213) | Placebo (n=215) |
Headache | 55 (26%) | 56 (26%) |
Common Cold | 47 (22%) | 46 (21%) |
Flu Symptoms | 24 (11%) | 40 (19%) |
Tooth Ache | 14 (7%) | 28 (13%) |
Periodontal Abscess | 8 (4%) | 21 (10%) |
Tooth Disorder | 13 (6%) | 19 (9%) |
Nausea | 17 (8%) | 12 (6%) |
Sinusitis | 7 (3%) | 18 (8%) |
Injury | 11 (5%) | 18 (8%) |
Dyspepsia | 13 (6%) | 5 (2%) |
Sore Throat | 11 (5%) | 13 (6%) |
Joint Pain | 12 (6%) | 8 (4%) |
Diarrhea | 12 (6%) | 8 (4%) |
Sinus Congestion | 11 (5%) | 11 (5%) |
Coughing | 9 (4%) | 11 (5%) |
Sinus Headache | 8 (4%) | 8 (4%) |
Rash | 8 (4%) | 6 (3%) |
Back Pain | 7 (3%) | 8 (4%) |
Back Ache | 4 (2%) | 9 (4%) |
Menstrual Cramp | 9 (4%) | 5 (2%) |
Acid Indigestion | 8 (4%) | 7 (3%) |
Pain | 8 (4%) | 5 (2%) |
Infection | 4 (2%) | 6 (3%) |
Gum Pain | 1 (<1%) | 6 (3%) |
Bronchitis | 7 (3%) | 5 (2%) |
Muscle Pain | 2 (1%) | 6 (3%) |
Note: Percentages are based on total number of study participants in each treatment group.
Adverse Reactions for Tetracyclines: The following adverse reactions have been observed in patients receiving tetracyclines:
Gastrointestinal: anorexia, nausea, vomiting, diarrhea, glossitis, dysphagia, enterocolitis, and inflammatory lesions (with vaginal candidiasis) in the anogenital region. Hepatotoxicity has been reported rarely. Rare instances of esophagitis and esophageal ulcerations have been reported in patients receiving the capsule forms of the drugs in the tetracycline class. Most of these patients took medications immediately before going to bed. (See DOSAGE AND ADMINISTRATION Section).
Skin: maculopapular and erythematous rashes, Stevens-Johnson syndrome, toxic epidermal necrolysis, erythema multiforme, and fixed drug eruption have been reported. Exfoliative dermatitis has been reported but is uncommon. Photosensitivity is discussed above. (See WARNINGS Section).
Renal toxicity: Rise in BUN has been reported and is apparently dose related. (See WARNINGS Section).
Hypersensitivity reactions: urticaria, angioneurotic edema, anaphylaxis, anaphylactoid purpura, serum sickness, pericarditis, and exacerbation of systemic lupus erythematosus.
Blood: Hemolytic anemia, thrombocytopenia, neutropenia, and eosinophilia have been reported.
Psychiatric: Depression, anxiety, suicidal ideation, insomnia, abnormal dreams, hallucination.
Drug Interactions
Because tetracyclines have been shown to depress plasma prothrombin activity, patients who are on anticoagulant therapy may require downward adjustment of their anticoagulant dosage.
Since bacterial antibiotics, such as the tetracycline class of antibiotics, may interfere with the bactericidal action of members of the β-lactam (e.g. penicillin) class of antibiotics, it is not advisable to administer these antibiotics concomitantly.
Absorption of tetracyclines is impaired by antacids containing aluminum, calcium, or magnesium, and iron-containing preparations, and by bismuth subsalicylate.
Barbiturates, carbamazepine, and phenytoin decrease the half-life of doxycycline.
The concurrent use of tetracycline and methoxyflurane has been reported to result in fatal renal toxicity.
Concurrent use of tetracyclines may render oral contraceptives less effective.
DESCRIPTION
Doxycycline hyclate tablets, USP are available as a 20 mg formulation of doxycycline for oral administration.
The structural formula of doxycycline hyclate is:
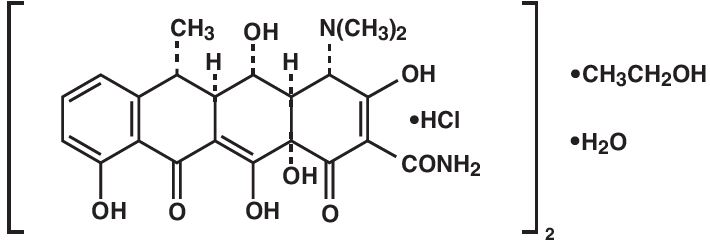
with a structural formula of (C 22 H 24 N 2 O 8 •HCl) 2 •C 2 H 6 O•H 2 O and a molecular weight of 1025.89. The chemical designation for doxycycline is 4-(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,5,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-2-naphthacenecarboxamide monohydrochloride, compound with ethyl alcohol (2:1), monohydrate.
Doxycycline hyclate is a yellow to light-yellow crystalline powder which is soluble in water.
Each tablet contains the following inactive ingredients: hypromellose, lactose anhydrous, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80 and titanium dioxide.
Meet USP Dissolution Test 5.
CLINICAL PHARMACOLOGY
After oral administration, doxycycline hyclate is rapidly and nearly completely absorbed from the gastrointestinal tract. Doxycycline is eliminated with a half-life of approximately 18 hours by renal and fecal excretion of unchanged drug.
HOW SUPPLIED
Doxycycline hyclate tablets USP, 20 mg are round, white unscored tablet, debossed “Є” above “362” on one side and plain on the other side. They are supplied as follows:
NDC 42806-362-01 Bottles of 100 tablets
NDC 42806-362-10 Bottles of 1000 tablets
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Dispense in tight, light-resistant containers as defined in the USP.
KEEP THIS AND ALL DRUGS OUT OF THE REACH OF CHILDREN.