Dicyclomine Hydrochloride - Dicyclomine Hydrochloride tablet prescribing information
INDICATIONS AND USAGE
Dicyclomine hydrochloride tablets are indicated for the treatment of patients with functional bowel/irritable bowel syndrome.
DOSAGE AND ADMINISTRATION
Dosage must be adjusted to individual patient needs.
Oral Dosage and Administration in Adults
The recommended initial dose is 20 mg four times a day. After one week treatment with the initial dose, the dose may be increased to 40 mg four times a day unless side effects limit dosage escalation. If efficacy is not achieved within 2 weeks or side effects require doses below 80 mg per day, the drug should be discontinued. Documented safety data are not available for doses above 80 mg daily for periods longer than 2 weeks.
DOSAGE FORMS AND STRENGTHS
Dicyclomine hydrochloride tablets USP, 20 mg are blue, round, unscored, flat-faced, beveled-edge tablets, debossed C and 170 on one side and plain on the other side and free from physical defects.
CONTRAINDICATIONS
Dicyclomine hydrochloride is contraindicated in infants less than 6 months of age [see Use in Specific Populations (8.4 )], nursing mothers [see Use in Specific Populations (8.3 )] , and in patients with:
• unstable cardiovascular status in acute hemorrhage • myasthenia gravis [see Warnings and Precautions (5.4 )] • glaucoma [see Adverse Reactions (6.3 ) and Drug Interactions (7.1 )] • obstructive uropathy [see Warnings and Precautions (5.8 )] • obstructive disease of the gastrointestinal tract [see Warnings and Precautions (5.5 )] • severe ulcerative colitis [see Warnings and Precautions (5.7 )] • reflux esophagitis
WARNINGS AND PRECAUTIONS
• Cardiovascular conditions : worsening of conditions (5.2 ) • Peripheral and central nervous system : heat prostration can occur with drug use (fever and heat stroke due to decreased sweating); drug should be discontinued and supportive measures instituted (5.3 ) • Psychosis and delirium have been reported in patients sensitive to anticholinergic drugs (such as elderly patients and/or in patients with mental illness): signs and symptoms resolve within 12 to 24 hours after discontinuation of dicyclomine hydrochloride (5.3 ) • Myasthenia Gravis : overdose may lead to muscular weakness and paralysis. Dicyclomine hydrochloride should be given to patients with myasthenia gravis only to reduce adverse muscarinic effects of an anticholinesterase (5.4 ) • Incomplete intestinal obstruction : diarrhea may be an early symptom especially in patients with ileostomy or colostomy. Treatment with dicyclomine hydrochloride would be inappropriate and possibly fatal (5.5 ) • Salmonella dysenteric patients : due to risk of toxic megacolon (5.6 ) • Ulcerative colitis: Dicyclomine hydrochloride should be used with caution in these patients; large doses may suppress intestinal motility or aggravate the serious complications of toxic megacolon (5.7 ) • Prostatic hypertrophy : Dicyclomine hydrochloride should be used with caution in these patients; may lead to urinary retention (5.8 ) • Hepatic and renal disease : should be used with caution (5.9 ) • Geriatric : use with caution in elderly who may be more susceptible to dicyclomine hydrochloride's adverse events (5.10 )
Cardiovascular Conditions
Dicyclomine hydrochloride needs to be used with caution in conditions characterized by tachyarrhythmia such as thyrotoxicosis, congestive heart failure and in cardiac surgery, where they may further accelerate the heart rate. Investigate any tachycardia before administration of dicyclomine hydrochloride. Care is required in patients with coronary heart disease, as ischemia and infarction may be worsened, and in patients with hypertension [see Adverse Reactions (6.3 )].
Peripheral and Central Nervous System
The peripheral effects of dicyclomine hydrochloride are a consequence of their inhibitory effect on muscarinic receptors of the autonomic nervous system. They include dryness of the mouth with difficulty in swallowing and talking, thirst, reduced bronchial secretions, dilatation of the pupils (mydriasis) with loss of accommodation (cycloplegia) and photophobia, flushing and dryness of the skin, transient bradycardia followed by tachycardia, with palpitations and arrhythmias, and difficulty in micturition, as well as reduction in the tone and motility of the gastrointestinal tract leading to constipation [see Adverse Reactions (6 )] .
In the presence of high environmental temperature heat prostration can occur with drug use (fever and heat stroke due to decreased sweating). It should also be used cautiously in patients with fever. If symptoms occur, the drug should be discontinued and supportive measures instituted. Because of the inhibitory effect on muscarinic receptors within the autonomic nervous system, caution should be taken in patients with autonomic neuropathy. Central nervous system (CNS) signs and symptoms include confusional state, disorientation, amnesia, hallucinations, dysarthria, ataxia, coma, euphoria, fatigue, insomnia, agitation and mannerisms, and inappropriate affect.
Psychosis and delirium have been reported in sensitive individuals (such as elderly patients and/or in patients with mental illness) given anticholinergic drugs. These CNS signs and symptoms usually resolve within 12 to 24 hours after discontinuation of the drug.
Dicyclomine hydrochloride may produce drowsiness, dizziness or blurred vision. The patient should be warned not to engage in activities requiring mental alertness, such as operating a motor vehicle or other machinery or performing hazardous work while taking dicyclomine hydrochloride.
Myasthenia Gravis
With overdosage, a curare-like action may occur (i.e., neuromuscular blockade leading to muscular weakness and possible paralysis). It should not be given to patients with myasthenia gravis except to reduce adverse muscarinic effects of an anticholinesterase [see Contraindications (4 )] .
Intestinal Obstruction
Diarrhea may be an early symptom of incomplete intestinal obstruction, especially in patients with ileostomy or colostomy. In this instance, treatment with this drug would be inappropriate and possibly harmful [see Contraindications (4 )] .
Rarely development of Ogilvie’s syndrome (colonic pseudo-obstruction) has been reported. Ogilvie’s syndrome is a clinical disorder with signs, symptoms, and radiographic appearance of an acute large bowel obstruction but with no evidence of distal colonic obstruction
Toxic Dilatation of Intestine megacolon
Toxic dilatation of intestine and intestinal perforation is possible when anticholinergic agents are administered in patients with Salmonella dysentery.
Ulcerative Colitis
Caution should be taken in patients with ulcerative colitis. Large doses may suppress intestinal motility to the point of producing a paralytic ileus and the use of this drug may precipitate or aggravate the serious complication of toxic megacolon [see Adverse Reactions (6.3 )] . Dicyclomine hydrochloride is contraindicated in patients with severe ulcerative colitis [see Contraindications (4 )] .
Prostatic Hypertrophy
Dicyclomine hydrochloride should be used with caution in patients with known or suspected prostatic enlargement, in whom prostatic enlargement may lead to urinary retention [see Adverse Reactions (6.3 )] .
Hepatic and Renal Disease
Dicyclomine hydrochloride should be used with caution in patients with known hepatic and renal impairment.
Geriatric Population
Dicyclomine hydrochloride should be used with caution in elderly who may be more susceptible to its adverse effects.
ADVERSE REACTIONS
The pattern of adverse effects seen with dicylomine is mostly related to its pharmacological actions at muscarinic receptors [see Clinical Pharmacology (12 )] . They are a consequence of the inhibitory effect on muscarinic receptors within the autonomic nervous system. These effects are dose-related and are usually reversible when treatment is discontinued.
The most serious adverse reactions reported with dicyclomine hydrochloride include cardiovascular and central nervous system symptoms [see Warnings and Precautions (5.2 , 5.3 )].
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. The data described below reflect exposure in controlled clinical trials involving over 100 patients treated for functional bowel/irritable bowel syndrome with dicyclomine hydrochloride at initial doses of 160 mg daily (40 mg four times a day)
In these trials most of the side effects were typically anticholinergic in nature and were reported by 61% of the patients. Table 1 presents adverse reactions ( MedDRA 13.0 preferred terms) by decreasing order of frequency in a side-by-side comparison with placebo. Table 1: Adverse reactions experienced in controlled clinical trials with decreasing order of frequency
| MedDRA Preferred Term | Dicyclomine Hydrochloride (40 mg four times a day) % | Placebo % |
| Dry Mouth | 33 | 5 |
| Dizziness | 40 | 5 |
| Vision blurred | 27 | 2 |
| Nausea | 14 | 6 |
| Somnolence | 9 | 1 |
| Asthenia | 7 | 1 |
| Nervousness | 6 | 2 |
Nine percent (9%) of patients were discontinued from dicyclomine hydrochloride because of one or more of these side effects (compared with 2% in the placebo group). In 41% of the patients with side effects, side effects disappeared or were tolerated at the 160 mg daily dose without reduction. A dose reduction from 160 mg daily to an average daily dose of 90 mg was required in 46% of the patients with side effects who then continued to experience a favorable clinical response; their side effects either disappeared or were tolerated.
Postmarketing Experience
The following adverse reactions, presented by system organ class in alphabetical order, have been identified during post approval use of dicyclomine hydrochloride. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. • Cardiac disorders: palpitations, tachyarrhythmias • Eye disorders: cycloplegia, mydriasis, vision blurred • Gastrointestinal disorders: abdominal distension, abdominal pain, constipation, dry mouth, dyspepsia, nausea, vomiting • General disorders and administration site conditions: fatigue, malaise • Immune System Disorders: drug hypersensitivity including face oedema, angioedema, anaphylactic shock • Nervous system disorders: dizziness, headache, somnolence, syncope • Psychiatric disorders: As with the other anti-cholinergic drugs, cases of delirium or symptoms of delirium such as amnesia (or transient global amnesia), agitation, confusional state, delusion, disorientation, hallucination (including visual hallucination) as well as mania, mood altered and pseudodementia, have been reported with the use of Dicyclomine. Nervousness and insomnia have also been reported. • Reproductive system and breast disorders: suppressed lactation • Respiratory, thoracic and mediastinal disorders: dyspnoea, nasal congestion • Skin and subcutaneous tissue disorder: dermatitis allergic, erythema, rash
Adverse Reactions Reported with Similar Drugs with Anticholinergic/Antispasmodic Action
Gastrointestinal: anorexia, Central Nervous System: tingling, numbness, dyskinesia, speech disturbance, insomnia Peripheral Nervous System: With overdosage, a curare-like action may occur (i.e., neuromuscular blockade leading to muscular weakness and possible paralysis) Ophthalmologic: diplopia, increased ocular tension Dermatologic/Allergic: urticaria, itching, and other dermal manifestations Genitourinary: urinary hesitancy, urinary retention in patients with prostatic hypertrophy Cardiovascular: hypertension Respiratory: apnea Other: decreased sweating, sneezing, throat congestion, impotence.
DRUG INTERACTIONS
• Antiglaucoma agents: anticholinergics antagonize antiglaucoma agents and may increase intraocular pressure (7 ) • Anticholinergic agents: may affect the gastrointestinal absorption of various drugs; may also increase certain actions or side effects of other anticholinergic drugs (7 ) • Antacids : interfere with the absorption of anticholinergic agents (7 )
Antiglaucoma Agents
Anticholinergics antagonize the effects of antiglaucoma agents. Anticholinergic drugs in the presence of increased intraocular pressure may be hazardous when taken concurrently with agents such as corticosteroids. Use of dicyclomine hydrochloride in patients with glaucoma is not recommended [see Contraindications (4 )] .
Other Drugs with Anticholinergic Activity
The following agents may increase certain actions or side effects of anticholinergic drugs including dicyclomine hydrochloride: amantadine, antiarrhythmic agents of Class I (e.g., quinidine), antihistamines, antipsychotic agents (e.g., phenothiazines), benzodiazepines, MAO inhibitors, narcotic analgesics (e.g., meperidine), nitrates and nitrites, sympathomimetic agents, tricyclic antidepressants, and other drugs having anticholinergic activity.
Other Gastrointestinal Motility Drugs
Interaction with other gastrointestinal motility drugs may antagonize the effects of drugs that alter gastrointestinal motility, such as metoclopramide.
Effect of Antacids
Because antacids may interfere with the absorption of anticholinergic agents including dicyclomine hydrochloride, simultaneous use of these drugs should be avoided.
Effect on Absorption of Other Drugs
Anticholinergic agents may affect gastrointestinal absorption of various drugs by affecting on gastrointestinal motility, such as slowly dissolving dosage forms of digoxin; increased serum digoxin concentration may result.
Effect on Gastric Acid Secretion
The inhibiting effects of anticholinergic drugs on gastric hydrochloric acid secretion are antagonized by agents used to treat achlorhydria and those used to test gastric secretion.
11 DESCRIPTION
Dicyclomine hydrochloride, USP is an antispasmodic and anticholinergic (antimuscarinic) agent available in the following dosage forms: Dicyclomine hydrochloride tablets, USP for oral use contain 20 mg dicyclomine hydrochloride, USP. Dicyclomine hydrochloride tablets USP, 20 mg also contain inactive ingredients: colloidal silicon dioxide, corn starch, hypromellose, lactose monohydrate, Lake of Brilliant Blue, sodium starch glycolate-Type A, and stearic acid. Dicyclomine hydrochloride, USP is [bicyclohexyl]-1-carboxylic acid, 2-(diethylamino) ethyl ester, hydrochloride, with a molecular formula of C 19 H 35 NO 2 •HCl and the following structural formula:
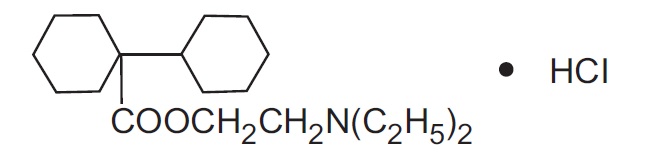
Molecular weight: 345.95 g/mol
Dicyclomine hydrochloride, USP occurs as a fine, white, crystalline, practically odorless powder with a bitter taste. It is soluble in water, freely soluble in alcohol and chloroform, and very slightly soluble in ether. FDA approved dissolution test specifications differ from the USP.
CLINICAL PHARMACOLOGY
Mechanism of Action
Dicyclomine relieves smooth muscle spasm of the gastrointestinal tract. Animal studies indicate that this action is achieved via a dual mechanism: • a specific anticholinergic effect (antimuscarinic) at the acetylcholine-receptor sites with approximately 1/8 the milligram potency of atropine ( in vitro , guinea pig ileum); and • a direct effect upon smooth muscle (musculotropic) as evidenced by dicyclomine’s antagonism of bradykinin- and histamine-induced spasms of the isolated guinea pig ileum.
Atropine did not affect responses to these two agonists. In vivo studies in cats and dogs showed dicyclomine to be equally potent against acetylcholine (ACh)- or barium chloride (BaCl 2 )-induced intestinal spasm while atropine was at least 200 times more potent against effects of ACh than BaCl 2 .
Tests for mydriatic effects in mice showed that dicyclomine was approximately 1/500 as potent as atropine; antisialagogue tests in rabbits showed dicyclomine to be 1/300 as potent as atropine.
Pharmacodynamics
Dicyclomine hydrochloride can inhibit the secretion of saliva and sweat, decrease gastrointestinal secretions and motility, cause drowsiness, dilate the pupils, increase heart rate, and depress motor function
Pharmacokinetics
Absorption and Distribution In man, dicyclomine is rapidly absorbed after oral administration, reaching peak values within 60-90 minutes. Mean volume of distribution for a 20 mg oral dose is approximately 3.65 L/kg suggesting extensive distribution in tissues. Elimination The metabolism of dicyclomine was not studied. The principal route of excretion is via the urine (79.5% of the dose). Excretion also occurs in the feces, but to a lesser extent (8.4%). Mean half-life of plasma elimination in one study was determined to be approximately 1.8 hours when plasma concentrations were measured for 9 hours after a single dose. In subsequent studies, plasma concentrations were followed for up to 24 hours after a single dose, showing a secondary phase of elimination with a somewhat longer half-life.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been conducted to evaluate the carcinogenic potential of dicyclomine. In studies in rats at doses of up to 100 mg/kg/day, dicyclomine produced no deleterious effects on breeding, conception, or parturition.
CLINICAL STUDIES
In controlled clinical trials involving over 100 patients who received drug, 82% of patients treated for functional bowel/irritable bowel syndrome with dicyclomine hydrochloride at initial doses of 160 mg daily (40 mg four times daily) demonstrated a favorable clinical response compared with 55% treated with placebo (p<0.05).
HOW SUPPLIED/STORAGE AND HANDLING
Dicyclomine hydrochloride tablets USP, 20 mg are blue, round, unscored, flat-faced, beveled-edge tablets, debossed C and 170 on one side and plain on the other side and free from physical defects. They are supplied as:
Bottles of 100 tablets...................(NDC 16571-259-01) Bottles of 1000 tablets.................(NDC 16571-259-10)
To prevent fading, avoid exposure to direct sunlight.
Store at 20º to 25ºC (68º to 77ºF); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature].
Keep out of the reach of children.
Dispense in a well-closed container as defined in USP/NF.
Mechanism of Action
Dicyclomine relieves smooth muscle spasm of the gastrointestinal tract. Animal studies indicate that this action is achieved via a dual mechanism: • a specific anticholinergic effect (antimuscarinic) at the acetylcholine-receptor sites with approximately 1/8 the milligram potency of atropine ( in vitro , guinea pig ileum); and • a direct effect upon smooth muscle (musculotropic) as evidenced by dicyclomine’s antagonism of bradykinin- and histamine-induced spasms of the isolated guinea pig ileum.
Atropine did not affect responses to these two agonists. In vivo studies in cats and dogs showed dicyclomine to be equally potent against acetylcholine (ACh)- or barium chloride (BaCl 2 )-induced intestinal spasm while atropine was at least 200 times more potent against effects of ACh than BaCl 2 .
Tests for mydriatic effects in mice showed that dicyclomine was approximately 1/500 as potent as atropine; antisialagogue tests in rabbits showed dicyclomine to be 1/300 as potent as atropine.