Get your patient on Diazoxide - Diazoxide suspension (Diazoxide)
Diazoxide - Diazoxide suspension prescribing information
INDICATIONS AND USAGE
Diazoxide Oral Suspension is indicated for the management of hypoglycemia due to hyperinsulinism associated with the following conditions:
- Adults: Inoperable islet cell adenoma or carcinoma, or extrapancreatic malignancy.
- Infants and Children: Leucine sensitivity, islet cell hyperplasia, nesidioblastosis, extrapancreatic malignancy, islet cell adenoma, or adenomatosis. Diazoxide Oral Suspension may be used preoperatively as a temporary measure, and postoperatively, if hypoglycemia persists.
Diazoxide should be used only after a diagnosis of hypoglycemia due to one of the above conditions has been definitely established. When other specific medical therapy or surgical management either has been unsuccessful or is not feasible, treatment with diazoxide should be considered.
DOSAGE AND ADMINISTRATION
Patients should be under close clinical observation when treatment with diazoxide is initiated. Carefully monitor the clinical response and blood glucose until the patient’s condition has stabilized satisfactory; in most instances, this may be accomplished in several days. If administration of diazoxide is not effective after 2 or 3 weeks, discontinue diazoxide.
Individualize the dosage of diazoxide based on the severity of the hypoglycemic condition and the blood glucose level and clinical response of the patient. Adjust the dosage until the desired clinical and laboratory effects are produced with the least amount of diazoxide. Take special care to ensure the accuracy of the dosage in infants and young children.
Adults and children:
The recommended starting dosage is 3 mg/kg/day, administered orally, divided into 3 equal doses every 8 hours or 2 equal doses every 12 hours. The dosage may be titrated to a maximum of 8 mg/kg/day. Patients with refractory hypoglycemia may require higher dosages.
Infants and newborns:
The recommended starting dosage is 8 mg/kg/day, administered orally, divided into 3 equal doses every 8 hours or 2 equal doses every 12 hours. The dosage may be titrated to a maximum of 15 mg/kg/day.
CONTRAINDICATIONS
Diazoxide is contraindicated in patients with:
- Functional hypoglycemia
- Hypersensitivity to diazoxide, any of the excipients in diazoxide, or other thiazides
ADVERSE REACTIONS:
Frequent and Serious:
Sodium and fluid retention is most common in young infants and in adults and may precipitate congestive heart failure in patients with compromised cardiac reserve (see DRUG INTERACTIONS ).
Infrequent but Serious:
Diabetic ketoacidosis and hyperosmolar nonketotic coma may develop very rapidly. Monitor patients for up to 7 days due to the long half-life of diazoxide (see OVERDOSAGE ).
Other frequent adverse reactions:
Hirsutism of the lanugo type, mainly on the forehead, back and limbs, occurs most commonly in children and women and may be cosmetically unacceptable. It subsides on discontinuation of the drug.
Hyperglycemia or glycosuria may require reduction in dosage in order to avoid progression to ketoacidosis or hyperosmolar coma.
Gastrointestinal intolerance may include anorexia, nausea, vomiting, abdominal pain, ileus, diarrhea, transient loss of taste.
Tachycardia, palpitations, increased levels of serum uric acid are common.
Thrombocytopenia with or without purpura may require discontinuation of the drug. Neutropenia is transient, is not associated with increased susceptibility to infection, and ordinarily does not require discontinuation of the drug. Skin rash, headache, weakness, and malaise may also occur.
Other adverse reactions:
Cardiovascular: Hypotension occurs occasionally, which may be augmented by thiazide diuretics given concurrently. A few cases of transient hypertension, for which no explanation is apparent, have been noted. Chest pain has been reported rarely. Pulmonary hypertension has been reported in neonates and young infants (see WARNINGS ).
There have been postmarketing reports of pericardial effusion in patients without structural heart disease; the majority of cases occurred in pediatric patients and infants.
Gastrointestinal: There have been postmarketing reports of necrotizing enterocolitis; the majority of cases occurred in infants with underlying co-morbid conditions.
Hematologic: eosinophilia; decreased hemoglobin / hematocrit; excessive bleeding, decreased IgG.
Hepato-renal: increased AST, alkaline phosphatase; azotemia, decreased creatinine clearance, reversible nephrotic syndrome, decreased urinary output, hematuria, albuminuria. Neurologic: anxiety, dizziness, insomnia, polyneuritis, paresthesia, pruritus, extrapyramidal signs.
Ophthalmologic: transient cataracts, subconjunctival hemorrhage, ring scotoma, blurred vision, diplopia, lacrimation. Skeletal, integumentary; monilial dermatitis, herpes, advance in bone age; loss of scalp hair. Systemic: fever, lymphadenopathy. Other; gout acute pancreatitis/pancreatic necrosis, galactorrhea, enlargement of lump in breast.
Drug Interactions:
Since diazoxide is highly bound to serum proteins, it may displace other substances which are also bound to protein, such as bilirubin or coumarin and its derivatives, resulting in higher blood levels of these substances. Concomitant administration of oral diazoxide and diphenylhydantoin may result in a loss of seizure control. Consider these potential interactions when administering diazoxide.
The concomitant administration of thiazides or other diuretics may potentiate the hyperglycemic and hyperuricemic effects of diazoxide.
DESCRIPTION
Diazoxide Oral Suspension, USP is a nondiuretic benzothiadiazine derivative taken orally for the management of symptomatic hypoglycemia. Diazoxide Oral Suspension, USP contains 50 mg of diazoxide in each milliliter and has a chocolate-mint flavor; alcohol content is approximately 7.25%. Other ingredients include sorbitol solution, chocolate mint flavor, propylene glycol, magnesium aluminum silicate, carboxymethylcellulose sodium, sodium benzoate, methylparaben, poloxamer 188, propylparaben, and purified water. Hydrochloric acid or sodium hydroxide may be added to adjust pH.
Diazoxide has the following structural formula:
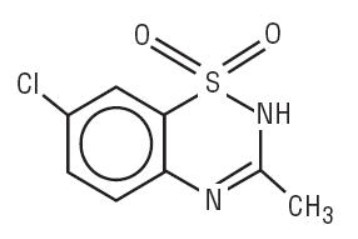
Diazoxide is 7-chloro-3-methyl-2 H -1,2,4-benzothiadiazine 1,1-dioxide with the empirical formula C 8 H 7 ClN 2 O 2 S and the molecular weight 230.7. It is a white powder practically insoluble to sparingly soluble in water.
CLINICAL PHARMACOLOGY
Diazoxide produces a dose-related increase in blood glucose, due primarily to an inhibition of insulin release from the pancreas, and also to an extrapancreatic effect.
The hyperglycemic effect of diazoxide begins within an hour and generally lasts no more than eight hours in the presence of normal renal function.
Diazoxide decreases the excretion of sodium and water, resulting in fluid retention which may be clinically significant.
The hypotensive effect of diazoxide is usually not marked (see ADVERSE REACTIONS ).
Other pharmacologic actions of diazoxide include increased pulse rate; increased serum uric acid levels due to decreased excretion; increased serum levels of free fatty acids' decreased chloride excretion; decreased para-aminohippuric acid; (PAH) clearance with no appreciable effect on glomerular filtration rate.
The concomitant administration of a benzothiazide diuretic may intensify the hyperglycemic and hyperuricemic effects of diazoxide. In the presence of hypokalemia, hyperglycemic effects are also potentiated.
Diazoxide-induced hyperglycemia is reversed by the administration of insulin or tolbutamide. The inhibition of insulin release by diazoxide is antagonized by alpha-adrenergic blocking agents.
Diazoxide is extensively bound (more than 90%) to serum proteins, and is excreted in the kidneys. The plasma half-life following intravenous administration is 28 ± 8.3 hours. Limited data on oral administration revealed a half-life of 24 and 36 hours in two adults. In four children aged four months to six years, the plasma half-life varied from 9.5 to 24 hours on long-term oral administration. The half-life may be prolonged following overdosage, and in patients with impaired renal function.
HOW SUPPLIED
Diazoxide Oral Suspension, USP 50 mg/mL, a chocolate-mint flavored suspension; bottle of 30 mL (NDC 13517-100-30), with dropper calibrated to deliver 10, 20, 30, 40 and 50 mg diazoxide.
Shake well before each use. Protect from light. Store in carton until contents are used. Store in light resistant container as defined in the USP. Store Diazoxide Oral Suspension at 25°C (77°F) excursions permitted 15°-30°C (59-86°F). [See USP Controlled Room Temperature].
Rx only
Manufactured for: e5 Pharma, LLC Boca Raton, FL 33432
1000001287 Rev. 01/2025