Dextrose And Sodium Chloride - Dextrose Monohydrate And Sodium Chloride injection, Solution prescribing information
INDICATIONS AND USAGE
Intravenous solutions containing dextrose and sodium chloride are indicated for parenteral replenishment of fluid, minimal carbohydrate calories, and sodium chloride as required by the clinical condition of the patient.
DOSAGE AND ADMINISTRATION
The dose is dependent upon the age, weight and clinical condition of the patient.
As reported in the literature, the dosage and constant infusion rate of intravenous dextrose must be selected with caution in pediatric patients, particularly neonates and low birth weight infants, because of the increased risk of hyperglycemia/hypoglycemia.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. See PRECAUTIONS .
Drug Interactions
Additives may be incompatible. Consult with pharmacist, if available. When introducing additives, use aseptic technique, mix thoroughly and do not store.
CONTRAINDICATIONS
None known.
ADVERSE REACTIONS
Reactions which may occur because of the solution or the technique of administration include febrile response, infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection, extravasation and hypervolemia.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures and save the remainder of the fluid for examination if deemed necessary.
DESCRIPTION
Dextrose and Sodium Chloride Injection, USP solutions are sterile and nonpyrogenic. They are large volume parenteral solutions containing various concentrations and combinations of these drugs in water for injection intended for intravenous administration.
See Table for summary of content and characteristics of these solutions.
The solutions contain no bacteriostat, antimicrobial agent or added buffer and each is intended only as a single-dose injection. When smaller doses are required the unused portion should be discarded.
The solutions are parenteral fluid, nutrient and electrolyte replenishers.
Dextrose, USP is chemically designated D-glucose monohydrate (C 6 H 12 O 6 • H 2 O), a hexose sugar freely soluble in water. It has the following structural formula:
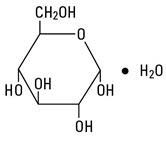
Sodium Chloride, USP is chemically designated NaCl, a white crystalline powder freely soluble in water.
Water for Injection, USP is chemically designated H 2 O.
The flexible plastic container is fabricated from a specially formulated polyvinylchloride. Water can permeate from inside the container into the overwrap but not in amounts sufficient to affect the solution significantly. Solutions in contact with the plastic container may leach out certain chemical components from the plastic in very small amounts; however, biological testing was supportive of the safety of the plastic container materials. Exposure to temperatures above 25°C/77°F during transport and storage will lead to minor losses in moisture content. Higher temperatures lead to greater losses. It is unlikely that these minor losses will lead to clinically significant changes within the expiration period.
CLINICAL PHARMACOLOGY
When administered intravenously, these solutions provide a source of water, carbohydrate and electrolytes.
Solutions which provide combinations of hypotonic or isotonic concentrations of dextrose and of sodium chloride are suitable for parenteral maintenance or replacement of water and electrolyte requirements with minimal carbohydrate calories.
Solutions containing carbohydrate in the form of dextrose restore blood glucose levels and provide calories. Carbohydrate in the form of dextrose may aid in minimizing liver glycogen depletion and exerts a protein-sparing action. Dextrose injected parenterally undergoes oxidation to carbon dioxide and water.
Sodium chloride in water dissociates to provide sodium (Na + ) and chloride (Cl¯) ions. Sodium (Na + ) is the principal cation of the extracellular fluid and plays a large part in the therapy of fluid and electrolyte disturbances. Chloride (Cl¯) has an integral role in buffering action when oxygen and carbon dioxide exchange occurs in the red blood cells. The distribution and excretion of sodium (Na + ) and chloride (Cl¯) are largely under the control of the kidney which maintains a balance between intake and output.
Water is an essential constituent of all body tissues and accounts for approximately 70% of total body weight. Average normal adult daily requirements range from two to three liters (1.0 to 1.5 liters each for insensible water loss by perspiration and urine production). Water balance is maintained by various regulatory mechanisms. Water distribution depends primarily on the concentration of electrolytes in the body compartments and sodium (Na + ) plays a major role in maintaining physiologic equilibrium.
HOW SUPPLIED
Dextrose and Sodium Chloride Injection, USP are supplied in single-dose flexible plastic containers in various sizes and concentrations as shown in the accompanying Table.
Grams/100 mL | Per 1000 mL | |||||||||
NDC No. | Product | Dextrose (hydrous) | Sodium Chloride | Sodium Na + | Chloride Cl¯ | Caloric Value | Tonicity | Osmolarity mOsmol/L (calc) | pH | Container size (mL) |
0409-7924-02 Manufactured by ICU Medical, Inc., Lake Forest, Illinois, 60045, USA 0990-7924-02 | 5% Dextrose and 0.225% Sodium Chloride Inj., USP | 5 | 0.225 | 38.5 mEq | 38.5 mEq | 170 | Hypertonic | 329 | 4.3 (3.5 to 6.5) | 250 |
0409-7924-03 Manufactured for ICU Medical, Inc., Lake Forest, Illinois, 60045, USA 0990-7924-03 , | 5% Dextrose and 0.225% Sodium Chloride Inj., USP | 5 | 0.225 | 38.5 mEq | 38.5 mEq | 170 | Hypertonic | 329 | 4.3 (3.5 to 6.5) | 500 |
0409-7924-09 , 0990-7924-09 , | 5% Dextrose and 0.225% Sodium Chloride Inj., USP | 5 | 0.225 | 38.5 mEq | 38.5 mEq | 170 | Hypertonic | 329 | 4.3 (3.5 to 6.5) | 1000 |
0409-7925-03 0990-7925-03 , | 5% Dextrose and 0.3% Sodium Chloride Inj., USP | 5 | 0.3 | 51 mEq | 51 mEq | 170 | Hypertonic | 355 | 4.3 (3.5 to 6.5) | 500 |
0409-7925-09 , 0990-7925-09 , | 5% Dextrose and 0.3% Sodium Chloride Inj., USP | 5 | 0.3 | 51 mEq | 51 mEq | 170 | Hypertonic | 355 | 4.3 (3.5 to 6.5) | 1000 |
0409-7926-02 0990-7926-02 | 5% Dextrose and 0.45% Sodium Chloride Inj., USP | 5 | 0.45 | 77 mEq | 77 mEq | 170 | Hypertonic | 406 | 4.3 (3.5 to 6.5) | 250 |
0409-7926-03 0990-7926-03 , | 5% Dextrose and 0.45% Sodium Chloride Inj., USP | 5 | 0.45 | 77 mEq | 77 mEq | 170 | Hypertonic | 406 | 4.3 (3.5 to 6.5) | 500 |
0409-7926-09 , 0990-7926-09 , | 5% Dextrose and 0.45% Sodium Chloride Inj., USP | 5 | 0.45 | 77 mEq | 77 mEq | 170 | Hypertonic | 406 | 4.3 (3.5 to 6.5) | 1000 |
0409-7941-03 0990-7941-03 , | 5% Dextrose and 0.9% Sodium Chloride Inj., USP | 5 | 0.9 | 154 mEq | 154 mEq | 170 | Hypertonic | 560 | 4.3 (3.5 to 6.5) | 500 |
0409-7941-09 , 0990-7941-09 , | 5% Dextrose and 0.9% Sodium Chloride Inj., USP | 5 | 0.9 | 154 mEq | 154 mEq | 170 | Hypertonic | 560 | 4.3 (3.5 to 6.5) | 1000 |
ICU Medical is transitioning NDC codes from "0409" to "0990" labeler code. Both NDC codes are expected to be in the market for a period of time.
Protect from freezing. Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room Temperature.]
Revised: August, 2020
IFU0000261