Dasatinib - Dasatinib tablet prescribing information
INDICATIONS AND USAGE
Dasatinib tablets are indicated for the treatment of adult patients with
- Newly diagnosed Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML) in chronic phase.
- Chronic, accelerated or myeloid or lymphoid blast phase Ph+ CML with resistance or intolerance to prior therapy including imatinib.
- Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ ALL) with resistance or intolerance to prior therapy.
Pediatric use information is approved for Bristol-Myers Squibb Company's Sprycel (dasatinib) tablets. However, due to Bristol-Myers Squibb Company's marketing exclusivity rights, this drug product is not labeled with that pediatric information.
DOSAGE AND ADMINISTRATION
Dosage of Dasatinib in Adult Patients
The recommended starting dosage of dasatinib for chronic phase CML in adults is 100 mg administered orally once daily. The recommended starting dosage of dasatinib for accelerated phase CML, myeloid or lymphoid blast phase CML or Ph+ ALL in adults is 140 mg administered orally once daily. Tablets should not be crushed, cut or chewed; they should be swallowed whole. Dasatinib can be taken with or without a meal, either in the morning or in the evening.
Pediatric use information is approved for Bristol-Myers Squibb Company's Sprycel (dasatinib) tablets. However, due to Bristol-Myers Squibb Company's marketing exclusivity rights, this drug product is not labeled with that pediatric information.
Dose Modification
Strong CYP3A4 Inducers
Avoid the use of concomitant strong CYP3A4 inducers and St. John's wort. If patients must be coadministered a strong CYP3A4 inducer, consider a dasatinib dose increase. If the dose of dasatinib is increased, monitor the patient carefully for toxicity [see Drug Interactions (7.1 )] .
Strong CYP3A4 Inhibitors
Avoid the use of concomitant strong CYP3A4 inhibitors and grapefruit juice. Recommend selecting an alternate concomitant medication with no or minimal enzyme inhibition potential, if possible. If dasatinib must be administered with a strong CYP3A4 inhibitor, consider a dose decrease to:
- 40 mg daily for patients taking dasatinib 140 mg daily.
- 20 mg daily for patients taking dasatinib 100 mg daily.
- 20 mg daily for patients taking dasatinib 70 mg daily.
For patients taking dasatinib 60 mg or 40 mg daily, consider interrupting dasatinib until the inhibitor is discontinued. Allow a washout period of approximately 1 week after the inhibitor is stopped before reinitiating dasatinib.
These reduced doses of dasatinib are predicted to adjust the area under the curve (AUC) to the range observed without CYP3A4 inhibitors; however, clinical data are not available with these dose adjustments in patients receiving strong CYP3A4 inhibitors. If dasatinib is not tolerated after dose reduction, either discontinue the strong CYP3A4 inhibitor or interrupt dasatinib until the inhibitor is discontinued. Allow a washout period of approximately 1 week after the inhibitor is stopped before the dasatinib dose is increased [see Drug Interactions (7.1 )] .
Pediatric use information is approved for Bristol-Myers Squibb Company's Sprycel (dasatinib) tablets. However, due to Bristol-Myers Squibb Company's marketing exclusivity rights, this drug product is not labeled with that pediatric information.
Dose Escalation in Adults with CML and Ph+ ALL
For adult patients with CML and Ph+ ALL, consider dose escalation to 140 mg once daily (chronic phase CML) or 180 mg once daily (advanced phase CML and Ph+ ALL) in patients who do not achieve a hematologic or cytogenetic response at the recommended starting dosage.
Pediatric use information is approved for Bristol-Myers Squibb Company's Sprycel (dasatinib) tablets. However, due to Bristol-Myers Squibb Company's marketing exclusivity rights, this drug product is not labeled with that pediatric information.
Dose Adjustment for Adverse Reactions
Myelosuppression
In clinical studies, myelosuppression was managed by dose interruption, dose reduction or discontinuation of study therapy. Hematopoietic growth factor has been used in patients with resistant myelosuppression. Guidelines for dose modifications for adult patients are summarized in Table 3.
•ANC: absolute neutrophil count | ||
| Chronic Phase CML (starting dose 100 mg once daily) | ANC • < 0.5 × 10 9 /L or Platelets < 50 × 10 9 /L | 1. Stop dasatinib until ANC ≥ 1 × 10 9 /L and platelets ≥ 50 × 10 9 /L. |
| 2. Resume treatment with dasatinib at the original starting dose if recovery occurs in ≤ 7 days. | ||
| 3. If platelets < 25 × 10 9 /L or recurrence of ANC < 0.5 × 10 9 /L for > 7 days, repeat Step 1 and resume dasatinib at a reduced dose of 80 mg once daily for second episode. For third episode, further reduce dose to 50 mg once daily (for newly diagnosed patients) or discontinue dasatinib (for patients resistant or intolerant to prior therapy including imatinib). | ||
| Accelerated Phase CML, Blast Phase CML and Ph+ ALL (starting dose 140 mg once daily) | ANC • < 0.5 × 10 9 /L or Platelets < 10 × 10 9 /L | 1. Check if cytopenia is related to leukemia (marrow aspirate or biopsy). 2. If cytopenia is unrelated to leukemia, stop dasatinib until ANC ≥ 1 × 10 9 /L and platelets ≥ 20 × 10 9 /L and resume at the original starting dose. 3. If recurrence of cytopenia, repeat Step 1 and resume dasatinib at a reduced dose of 100 mg once daily (second episode) or 80 mg once daily (third episode). 4. If cytopenia is related to leukemia, consider dose escalation to 180 mg once daily. |
Non-Hematologic Adverse Reactions
For adults with Ph+ CML and ALL if a severe nonhematologic adverse reaction develops with dasatinib use, treatment must be withheld until the adverse reaction has resolved or improved. Thereafter, treatment can be resumed as appropriate at a reduced dose depending on the severity and recurrence [see Warnings and Precautions (5 )] .
Pediatric use information is approved for Bristol-Myers Squibb Company's Sprycel (dasatinib) tablets. However, due to Bristol-Myers Squibb Company's marketing exclusivity rights, this drug product is not labeled with that pediatric information.
Duration of Treatment
In clinical studies, treatment with dasatinib in adults with chronic phase CML was continued until disease progression or until no longer tolerated by the patient. The effect of stopping treatment on long-term disease outcome after the achievement of a cytogenetic response (including complete cytogenetic response [CCyR]) or major molecular response (MMR and MR4.5) has not been established.
Dasatinib is a hazardous product. Follow applicable special handling and disposal procedures. 1
Pediatric use information is approved for Bristol-Myers Squibb Company's Sprycel (dasatinib) tablets. However, due to Bristol-Myers Squibb Company's marketing exclusivity rights, this drug product is not labeled with that pediatric information.
DOSAGE FORMS AND STRENGTHS
Dasatinib Tablets, 20 mg are white to off-white, round, film-coated tablets, debossed with '1741' on one side and plain on the other side
Dasatinib Tablets, 50 mg are white to off-white, oval, film-coated tablets, debossed with '1742' on one side and plain on the other side.
Dasatinib Tablets, 70 mg are white to off-white, round, film-coated tablets, debossed with '1743' on one side and plain on the other side.
Dasatinib Tablets, 80 mg are white to off-white, oblong, film-coated tablets, debossed with '1744' on one side and plain on the other side
Dasatinib Tablets, 100 mg are white to off-white, oval, film-coated tablets, debossed with '1745' on one side and plain on the other side
Dasatinib Tablets, 140 mg are white to off-white, round, film-coated tablets, debossed with '1746' on one side and plain on the other side.
USE IN SPECIFIC POPULATIONS
- Lactation: Advise women not to breastfeed. (8.2 )
Pediatric use information is approved for Bristol-Myers Squibb Company's Sprycel (dasatinib) tablets. However, due to Bristol-Myers Squibb Company's marketing exclusivity rights, this drug product is not labeled with that pediatric information.
Pregnancy
Risk Summary
Based on limited human data, dasatinib can cause fetal harm when administered to a pregnant woman. Adverse pharmacologic effects including hydrops fetalis, fetal leukopenia and fetal thrombocytopenia have been reported with maternal exposure to dasatinib. Animal reproduction studies in rats have demonstrated extensive mortality during organogenesis, the fetal period and in neonates. Skeletal malformations were observed in a limited number of surviving rat and rabbit conceptuses. These findings occurred at dasatinib plasma concentrations below those in humans receiving therapeutic doses of dasatinib [see Data] . Advise a pregnant woman of the potential risk to a fetus.
The estimated background risk in the U.S. general population of major birth defects is 2% to 4% and of miscarriage is 15% to 20% of clinically recognized pregnancies.
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Transplacental transfer of dasatinib has been reported. Dasatinib has been measured in fetal plasma and amniotic fluid at concentrations comparable to those in maternal plasma. Hydrops fetalis, fetal leukopenia and fetal thrombocytopenia have been reported with maternal exposure to dasatinib. These adverse pharmacologic effects on the fetus are similar to adverse reactions observed in adult patients and may result in fetal harm or neonatal death [see Warnings and Precautions (5.1 , 5.3 )] .
Data
Human Data
Based on human experience, dasatinib is suspected to cause congenital malformations, including neural tube defects and harmful pharmacological effects on the fetus when administered during pregnancy.
Animal Data
In nonclinical studies at plasma concentrations below those observed in humans receiving therapeutic doses of dasatinib, embryo-fetal toxicities were observed in rats and rabbits. Fetal death was observed in rats. In both rats and rabbits, the lowest doses of dasatinib tested (rat: 2.5 mg/kg/day [15 mg/m 2 /day] and rabbit: 0.5 mg/kg/day [6 mg/m 2 /day]) resulted in embryo-fetal toxicities. These doses produced maternal AUCs of 105 ng•h/mL and 44 ng•h/mL (0.1-fold the human AUC) in rats and rabbits, respectively. Embryo-fetal toxicities included skeletal malformations at multiple sites (scapula, humerus, femur, radius, ribs and clavicle), reduced ossification (sternum; thoracic, lumbar and sacral vertebrae; forepaw phalanges; pelvis and hyoid body), edema and microhepatia. In a pre- and postnatal development study in rats, administration of dasatinib from gestation day (GD) 16 through lactation day (LD) 20, GD 21 through LD 20 or LD 4 through LD 20 resulted in extensive pup mortality at maternal exposures that were below the exposures in patients treated with dasatinib at the recommended labeling dose.
Lactation
Risk Summary
No data are available regarding the presence of dasatinib in human milk, the effects of the drug on the breastfed child or the effects of the drug on milk production. However, dasatinib is present in the milk of lactating rats. Because of the potential for serious adverse reactions in nursing children from dasatinib, breastfeeding is not recommended during treatment with dasatinib and for 2 weeks after the last dose.
Females and Males of Reproductive Potential
Dasatinib can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1 )] .
Contraception
Advise females of reproductive potential and males with female partners of reproductive potential to use effective contraception during treatment with dasatinib and for 30 days after the last dose.
Infertility
Based on animal data, dasatinib may result in damage to female and male reproductive tissues [see Nonclinical Toxicology (13.1 )] .
Pediatric Use
Monitor bone growth and development in pediatric patients [see Warnings and Precautions (5.10 )].
Pediatric use information is approved for Bristol-Myers Squibb Company's Sprycel (dasatinib) tablets. However, due to Bristol-Myers Squibb Company's marketing exclusivity rights, this drug product is not labeled with that pediatric information.
Geriatric Use
Of the 2,712 patients in clinical studies of dasatinib, 617 (23%) were 65 years of age and older and 123 (5%) were 75 years of age and older. No differences in confirmed Complete Cytogenetic Response (cCCyR) and MMR were observed between older and younger patients. While the safety profile of dasatinib in the geriatric population was similar to that in the younger population, patients aged 65 years and older are more likely to experience the commonly reported adverse reactions of fatigue, pleural effusion, diarrhea, dyspnea, cough, lower gastrointestinal hemorrhage and appetite disturbance and more likely to experience the less frequently reported adverse reactions of abdominal distention, dizziness, pericardial effusion, congestive heart failure, hypertension, pulmonary edema and weight decrease and should be monitored closely.
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
- Myelosuppression and Bleeding Events: Severe thrombocytopenia, neutropenia and anemia may occur. Use caution if used concomitantly with medications that inhibit platelet function or anticoagulants. Monitor complete blood counts regularly. Transfuse and interrupt dasatinib when indicated. (2.5 , 5.1 , 5.2 )
- Fluid Retention: Fluid retention, sometimes severe, including pleural effusions. Manage with supportive care measures and/or dose modification. (2.5 , 5.3 )
- Cardiovascular Toxicity: Monitor patients for signs or symptoms and treat appropriately. (5.4 )
- Pulmonary Arterial Hypertension (PAH): Dasatinib may increase the risk of developing PAH which may be reversible on discontinuation. Consider baseline risk and evaluate patients for signs and symptoms of PAH during treatment. Stop dasatinib if PAH is confirmed. (5.5 )
- QT Prolongation: Use dasatinib with caution in patients who have or may develop prolongation of the QT interval. (5.6 )
- Severe Dermatologic Reactions: Individual cases of severe mucocutaneous dermatologic reactions have been reported. (5.7 )
- Tumor Lysis Syndrome: Tumor lysis syndrome has been reported. Maintain adequate hydration and correct uric acid levels prior to initiating therapy with dasatinib. (5.8 )
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise patients of reproductive potential of potential risk to fetus and to use effective contraception. (5.9 , 8.1 , 8.3 )
- Effects on Growth and Development in Pediatric Patients: epiphyses delayed fusion, osteopenia, growth retardation and gynecomastia have been reported. Monitor bone growth and development in pediatric patients. (5.10 )
- Hepatotoxicity: Assess liver function before initiation of treatment and monthly thereafter or as clinically indicated. Monitor liver function when combined with chemotherapy known to be associated with liver dysfunction. (5.11 )
Myelosuppression
Treatment with dasatinib is associated with severe (NCI CTCAE Grade 3 or 4) thrombocytopenia, neutropenia and anemia, which occur earlier and more frequently in patients with advanced phase CML or Ph+ ALL than in patients with chronic phase CML [see Adverse Reactions (6.1 )] .
In patients with chronic phase CML, perform complete blood counts (CBCs) every 2 weeks for 12 weeks, then every 3 months thereafter or as clinically indicated. In patients with advanced phase CML or Ph+ ALL, perform CBCs weekly for the first 2 months and then monthly thereafter or as clinically indicated.
Myelosuppression is generally reversible and usually managed by withholding dasatinib temporarily and/or dose reduction [see Dosage and Administration (2.5 )] .
Pediatric use information is approved for Bristol-Myers Squibb Company's Sprycel (dasatinib) tablets. However, due to Bristol-Myers Squibb Company's marketing exclusivity rights, this drug product is not labeled with that pediatric information.
Bleeding-Related Events
Dasatinib can cause serious and fatal bleeding. In all CML or Ph+ ALL clinical studies, Grade ≥ 3 central nervous system (CNS) hemorrhages, including fatalities, occurred in < 1% of patients receiving dasatinib. The incidence of Grade 3/4 hemorrhage occurred in 5.8% of adult patients and generally required treatment interruptions and transfusions. The incidence of Grade 5 hemorrhage occurred in 0.4% of adult patients. The most frequent site of hemorrhage was gastrointestinal [see Adverse Reactions (6.1 )] . Most bleeding events in clinical studies were associated with severe thrombocytopenia. In addition to causing thrombocytopenia in human subjects, dasatinib caused platelet dysfunction in vitro .
Concomitant medications that inhibit platelet function or anticoagulants may increase the risk of hemorrhage.
Fluid Retention
Dasatinib may cause fluid retention [see Adverse Reactions (6.1 )] . After 5 years of follow-up in the adult randomized newly diagnosed chronic phase CML study (n=258), Grade 3 or 4 fluid retention was reported in 5% of patients, including 3% of patients with Grade 3 or 4 pleural effusion. In adult patients with newly diagnosed or imatinib-resistant or -intolerant chronic phase CML, Grade 3 or 4 fluid retention occurred in 6% of patients treated with dasatinib at the recommended dose (n=548). In adult patients with advanced phase CML or Ph+ ALL treated with dasatinib at the recommended dose (n=304), Grade 3 or 4 fluid retention was reported in 8% of patients, including Grade 3 or 4 pleural effusion reported in 7% of patients.
Evaluate patients who develop symptoms of pleural effusion or other fluid retention, such as new or worsened dyspnea on exertion or at rest, pleuritic chest pain or dry cough, promptly with a chest x-ray or additional diagnostic imaging as appropriate. Fluid retention events were typically managed by supportive care measures that may include diuretics or short courses of steroids. Severe pleural effusion may require thoracentesis and oxygen therapy. Consider dose reduction or treatment interruption [see Dosage and Administration (2.5 )] .
Pediatric use information is approved for Bristol-Myers Squibb Company's Sprycel (dasatinib) tablets. However, due to Bristol-Myers Squibb Company's marketing exclusivity rights, this drug product is not labeled with that pediatric information.
Cardiovascular Toxicity
Dasatinib can cause cardiac dysfunction [see Adverse Reactions (6.1 )] . After 5 years of follow-up in the randomized newly diagnosed chronic phase CML trial in adults (n=258), the following cardiac adverse reactions occurred: cardiac ischemic events (3.9% dasatinib vs. 1.6% imatinib), cardiac-related fluid retention (8.5% dasatinib vs. 3.9% imatinib) and conduction system abnormalities, most commonly arrhythmia and palpitations (7% dasatinib vs. 5% imatinib). Two cases (0.8%) of peripheral arterial occlusive disease occurred with imatinib and 2 (0.8%) transient ischemic attacks occurred with dasatinib. Monitor patients for signs or symptoms consistent with cardiac dysfunction and treat appropriately.
Pulmonary Arterial Hypertension
Dasatinib may increase the risk of developing pulmonary arterial hypertension (PAH) in adult patients which may occur any time after initiation, including after more than 1 year of treatment. Manifestations include dyspnea, fatigue, hypoxia and fluid retention [see Adverse Reactions (6.1 )] . PAH may be reversible on discontinuation of dasatinib. Evaluate patients for signs and symptoms of underlying cardiopulmonary disease prior to initiating dasatinib and during treatment. If PAH is confirmed, dasatinib should be permanently discontinued.
QT Prolongation
Dasatinib may increase the risk of prolongation of QTc in patients including those with hypokalemia or hypomagnesemia, patients with congenital long QT syndrome, patients taking antiarrhythmic medicines or other medicinal products that lead to QT prolongation and cumulative high-dose anthracycline therapy [see Adverse Reactions (6.1 )] . Correct hypokalemia or hypomagnesemia prior to and during dasatinib administration.
Severe Dermatologic Reactions
Cases of severe mucocutaneous dermatologic reactions, including Stevens-Johnson syndrome [see Adverse Reactions (6.2 )] and erythema multiforme, have been reported in patients treated with dasatinib. Discontinue permanently in patients who experience a severe mucocutaneous reaction during treatment if no other etiology can be identified.
Tumor Lysis Syndrome
Tumor lysis syndrome has been reported in patients with resistance to prior imatinib therapy, primarily in advanced phase disease. Due to potential for tumor lysis syndrome, maintain adequate hydration, correct uric acid levels prior to initiating therapy with dasatinib and monitor electrolyte levels. Patients with advanced stage disease and/or high tumor burden may be at increased risk and should be monitored more frequently [see Adverse Reactions (6.1 )] .
Embryo-Fetal Toxicity
Based on limited human data, dasatinib can cause fetal harm when administered to a pregnant woman. Adverse pharmacologic effects of dasatinib including hydrops fetalis, fetal leukopenia and fetal thrombocytopenia have been reported with maternal exposure to dasatinib. Advise females of reproductive potential and males with female partners of reproductive potential to use effective contraception during treatment with dasatinib and for 30 days after the last dose [see Use in Specific Populations (8.1 , 8.3 )] .
Effects on Growth and Development in Pediatric Patients
In pediatric trials of dasatinib in chronic phase CML after at least 2 years of treatment, adverse reactions associated with bone growth and development were reported in 5 (5.2%) patients, one of which was severe in intensity (Growth Retardation Grade 3). These 5 cases included cases of epiphyses delayed fusion, osteopenia, growth retardation and gynecomastia [see Adverse Reactions (6.1 ) and Use in Specific Populations (8.4 )] . Of these 5 cases, 1 case of osteopenia and 1 case of gynecomastia resolved during treatment. Monitor bone growth and development in pediatric patients.
Hepatotoxicity
Dasatinib may cause hepatotoxicity as measured by elevations in bilirubin, aspartate aminotransferase (AST), alanine aminotransferase (ALT) and alkaline phosphatase [see Adverse Reactions (6.1 )] . Monitor transaminases at baseline and monthly or as clinically indicated during treatment. Reduce dose, withhold or permanently discontinue dasatinib based on severity [see Dosage and Administration (2.5 )] . When dasatinib is administered in combination with chemotherapy, liver toxicity in the form of transaminase elevation and hyperbilirubinemia has been observed. Monitor hepatic function when dasatinib is used in combination with chemotherapy.
ADVERSE REACTIONS
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
- Myelosuppression [see Dosage and Administration (2.5 ) and Warnings and Precautions (5.1 )] .
- Bleeding-related events [see Warnings and Precautions (5.2 )] .
- Fluid retention [see Warnings and Precautions (5.3 )] .
- Cardiovascular toxicity [see Warnings and Precautions (5.4 )] .
- Pulmonary arterial hypertension [see Warnings and Precautions (5.5 )] .
- QT prolongation [see Warnings and Precautions (5.6 )] .
- Severe dermatologic reactions [see Warnings and Precautions (5.7 )] .
- Tumor lysis syndrome [see Warnings and Precautions (5.8 )] .
- Effects on growth and development in pediatric patients [see Warnings and Precautions (5.10 )].
- Hepatotoxicity [see Warnings and Precautions (5.11 )] .
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to dasatinib administered as single-agent therapy at all doses tested in clinical studies (n=2,809), including 324 adult patients with newly diagnosed chronic phase CML, 2,388 adult patients with imatinib-resistant or -intolerant chronic or advanced phase CML or Ph+ ALL. The median duration of therapy in a total of 2,712 adult patients was 19.2 months (range 0 to 93.2 months). In a randomized trial in patients with newly diagnosed chronic phase CML, the median duration of therapy was approximately 60 months. The median duration of therapy in 1,618 adult patients with chronic phase CML was 29 months (range 0 to 92.9 months).
The median duration of therapy in 1,094 adult patients with advanced phase CML or Ph+ ALL was 6.2 months (range 0 to 93.2 months).
In the overall population of 2,712 adult patients, 88% of patients experienced adverse reactions at some time and 19% experienced adverse reactions leading to treatment discontinuation.
In the randomized trial in adult patients with newly diagnosed chronic phase CML, drug was discontinued for adverse reactions in 16% of patients with a minimum of 60 months of follow-up. After a minimum of 60 months of follow-up, the cumulative discontinuation rate was 39%. Among the 1,618 patients with chronic phase CML, drug-related adverse reactions leading to discontinuation were reported in 329 (20.3%) patients; among the 1,094 patients with advanced phase CML or Ph+ ALL, drug-related adverse reactions leading to discontinuation were reported in 191 (17.5%) patients.
Adverse reactions reported in ≥ 10% of adult patients and other adverse reactions of interest, in a randomized trial in patients with newly diagnosed chronic phase CML at a median follow-up of approximately 60 months are presented in Table 6.
Adverse reactions reported in ≥ 10% of adult patients treated at the recommended dose of 100 mg once daily (n=165) and other adverse reactions of interest, in a randomized dose-optimization trial of patients with chronic phase CML resistant or intolerant to prior imatinib therapy at a median follow-up of approximately 84 months are presented in Table 8.
Drug-related serious adverse reactions (SARs) were reported for 16.7% of adult patients in the randomized trial of patients with newly diagnosed chronic phase CML. Serious adverse reactions reported in ≥ 5% of patients included pleural effusion (5%).
Drug-related SARs were reported for 26.1% of patients treated at the recommended dose of 100 mg once daily in the randomized dose-optimization trial of adult patients with chronic phase CML resistant or intolerant to prior imatinib therapy. Serious adverse reactions reported in ≥ 5% of patients included pleural effusion (10%).
Chronic Myeloid Leukemia (CML)
Adverse reactions (excluding laboratory abnormalities) that were reported in at least 10% of adult patients are shown in Table 6 for newly diagnosed patients with chronic phase CML and Tables 8 and 10 for CML patients with resistance or intolerance to prior imatinib therapy.
a Includes cardiac failure acute, cardiac failure congestive, cardiomyopathy, diastolic dysfunction, ejection fraction decreased and left ventricular dysfunction. | ||||
b Includes erythema, erythema multiforme, rash, rash generalized, rash macular, rash papular, rash pustular, skin exfoliation and rash vesicular. | ||||
c Adverse reaction of special interest with < 10% frequency. | ||||
d Includes conjunctival hemorrhage, ear hemorrhage, ecchymosis, epistaxis, eye hemorrhage, gingival bleeding, hematoma, hematuria, hemoptysis, intra-abdominal hematoma, petechiae, scleral hemorrhage, uterine hemorrhage and vaginal hemorrhage. | ||||
| All Grades | Grade 3/4 | |||
| Dasatinib (n=258) | Imatinib (n=258) | Dasatinib (n=258) | Imatinib (n=258) | |
| Adverse Reaction | Percent (%) of Patients | |||
| Fluid retention | 38 | 45 | 5 | 1 |
| Pleural effusion | 28 | 1 | 3 | 0 |
| Superficial localized edema | 14 | 38 | 0 | < 1 |
| Pulmonary hypertension | 5 | < 1 | 1 | 0 |
| Generalized edema | 4 | 7 | 0 | 0 |
| Pericardial effusion | 4 | 1 | 1 | 0 |
| Congestive heart failure/ cardiac dysfunction a | 2 | 1 | < 1 | < 1 |
| Pulmonary edema | 1 | 0 | 0 | 0 |
| Diarrhea | 22 | 23 | 1 | 1 |
| Musculoskeletal pain | 14 | 17 | 0 | < 1 |
| Rash b | 14 | 18 | 0 | 2 |
| Headache | 14 | 11 | 0 | 0 |
| Abdominal pain | 11 | 8 | 0 | 1 |
| Fatigue | 11 | 12 | < 1 | 0 |
| Nausea | 10 | 25 | 0 | 0 |
| Myalgia | 7 | 12 | 0 | 0 |
| Arthralgia | 7 | 10 | 0 | <1 |
| Hemorrhage c | 8 | 8 | 1 | 1 |
| Gastrointestinal bleeding | 2 | 2 | 1 | 0 |
| Other bleeding d | 6 | 6 | 0 | < 1 |
| CNS bleeding | < 1 | < 1 | 0 | < 1 |
| Vomiting | 5 | 12 | 0 | 0 |
| Muscle spasms | 5 | 21 | 0 | < 1 |
A comparison of cumulative rates of adverse reactions reported in ≥ 10% of patients with minimum follow-up of 1 year and 5 years in a randomized trial of newly diagnosed patients with chronic phase CML treated with dasatinib are shown in Table 7.
a Includes cardiac failure acute, cardiac failure congestive, cardiomyopathy, diastolic dysfunction, ejection fraction decreased and left ventricular dysfunction. | ||||
b Includes erythema, erythema multiforme, rash, rash generalized, rash macular, rash papular, rash pustular, skin exfoliation and rash vesicular. | ||||
| Minimum of 1 Year Follow-up | Minimum of 5 Years Follow-up | |||
| All Grades | Grade 3/4 | All Grades | Grade 3/4 | |
| Adverse Reaction | Percent (%) of Patients | |||
| Fluid retention | 19 | 1 | 38 | 5 |
| Pleural effusion | 10 | 0 | 28 | 3 |
| Superficial localized edema | 9 | 0 | 14 | 0 |
| Pulmonary hypertension | 1 | 0 | 5 | 1 |
| Generalized edema | 2 | 0 | 4 | 0 |
| Pericardial effusion | 1 | < 1 | 4 | 1 |
| Congestive heart failure/cardiac dysfunction a | 2 | < 1 | 2 | < 1 |
| Pulmonary edema | < 1 | 0 | 1 | 0 |
| Diarrhea | 17 | < 1 | 22 | 1 |
| Musculoskeletal pain | 11 | 0 | 14 | 0 |
| Rash b | 11 | 0 | 14 | 0 |
| Headache | 12 | 0 | 14 | 0 |
| Abdominal pain | 7 | 0 | 11 | 0 |
| Fatigue | 8 | < 1 | 11 | < 1 |
| Nausea | 8 | 0 | 10 | 0 |
At 60 months, there were 26 deaths in dasatinib-treated patients (10.1%) and 26 deaths in imatinib-treated patients (10.1%); 1 death in each group was assessed by the investigator as related to study therapy.
a Includes drug eruption, erythema, erythema multiforme, erythrosis, exfoliative rash, generalized erythema, genital rash, heat rash, milia, rash, rash erythematous, rash follicular, rash generalized, rash macular, rash maculopapular, rash papular, rash pruritic, rash pustular, skin exfoliation, skin irritation, urticaria vesiculosa and rash vesicular. | ||
| 100 mg Once Daily | ||
| Chronic (n=165) | ||
| All Grades | Grade 3/4 | |
| Adverse Reaction | Percent (%) of Patients | |
| Fluid retention | 48 | 7 |
| Superficial localized edema | 22 | 0 |
| Pleural effusion | 28 | 5 |
| Generalized edema | 4 | 0 |
| Pericardial effusion | 3 | 1 |
| Pulmonary hypertension | 2 | 1 |
| Headache | 33 | 1 |
| Diarrhea | 28 | 2 |
| Fatigue | 26 | 4 |
| Dyspnea | 24 | 2 |
| Musculoskeletal pain | 22 | 2 |
| Nausea | 18 | 1 |
| Skin rash a | 18 | 2 |
| Myalgia | 13 | 0 |
| Arthralgia | 13 | 1 |
| Infection (including bacterial, viral, fungal and non-specified) | 13 | 1 |
| Abdominal pain | 12 | 1 |
| Hemorrhage | 12 | 1 |
| Gastrointestinal bleeding | 2 | 1 |
| Pruritus | 12 | 1 |
| Pain | 11 | 1 |
| Constipation | 10 | 1 |
Cumulative rates of selected adverse reactions that were reported over time in patients treated with the 100 mg once daily recommended starting dose in a randomized dose-optimization trial of imatinib-resistant or -intolerant patients with chronic phase CML are shown in Table 9.
a Randomized dose-optimization trial results reported in the recommended starting dose of 100 mg once daily (n=165) population. | ||||||
| Minimum of 2 Years Follow-up | Minimum of 5 Years Follow-up | Minimum of 7 Years Follow-up | ||||
| All Grades | Grade 3/4 | All Grades | Grade 3/4 | All Grades | Grade 3/4 | |
| Adverse Reaction | Percent (%) of Patients | |||||
| Diarrhea | 27 | 2 | 28 | 2 | 28 | 2 |
| Fluid retention | 34 | 4 | 42 | 6 | 48 | 7 |
| Superficial edema | 18 | 0 | 21 | 0 | 22 | 0 |
| Pleural effusion | 18 | 2 | 24 | 4 | 28 | 5 |
| Generalized edema | 3 | 0 | 4 | 0 | 4 | 0 |
| Pericardial effusion | 2 | 1 | 2 | 1 | 3 | 1 |
| Pulmonary hypertension | 0 | 0 | 0 | 0 | 2 | 1 |
| Hemorrhage | 11 | 1 | 11 | 1 | 12 | 1 |
| Gastrointestinal bleeding | 2 | 1 | 2 | 1 | 2 | 1 |
a Includes ventricular dysfunction, cardiac failure, cardiac failure congestive, cardiomyopathy, congestive cardiomyopathy, diastolic dysfunction, ejection fraction decreased and ventricular failure. | ||||||
b Includes drug eruption, erythema, erythema multiforme, erythrosis, exfoliative rash, generalized erythema, genital rash, heat rash, milia, rash, rash erythematous, rash follicular, rash generalized, rash macular, rash maculopapular, rash papular, rash pruritic, rash pustular, skin exfoliation, skin irritation, urticaria vesiculosa and rash vesicular. | ||||||
| 140 mg Once Daily | ||||||
| Accelerated (n=157) | Myeloid Blast (n=74) | Lymphoid Blast (n=33) | ||||
| All Grades | Grade 3/4 | All Grades | Grade 3/4 | All Grades | Grade 3/4 | |
| Adverse Reaction | Percent (%) of Patients | |||||
| Fluid retention | 35 | 8 | 34 | 7 | 21 | 6 |
| Superficial localized edema | 18 | 1 | 14 | 0 | 3 | 0 |
| Pleural effusion | 21 | 7 | 20 | 7 | 21 | 6 |
| Generalized edema | 1 | 0 | 3 | 0 | 0 | 0 |
| Pericardial effusion | 3 | 1 | 0 | 0 | 0 | 0 |
| Congestive heart failure/cardiac dysfunction a | 0 | 0 | 4 | 0 | 0 | 0 |
| Pulmonary edema | 1 | 0 | 4 | 3 | 0 | 0 |
| Headache | 27 | 1 | 18 | 1 | 15 | 3 |
| Diarrhea | 31 | 3 | 20 | 5 | 18 | 0 |
| Fatigue | 19 | 2 | 20 | 1 | 9 | 3 |
| Dyspnea | 20 | 3 | 15 | 3 | 3 | 3 |
| Musculoskeletal pain | 11 | 0 | 8 | 1 | 0 | 0 |
| Nausea | 19 | 1 | 23 | 1 | 21 | 3 |
| Skin rash b | 15 | 0 | 16 | 1 | 21 | 0 |
| Arthralgia | 10 | 0 | 5 | 1 | 0 | 0 |
| Infection (including bacterial, viral, fungal and non-specified) | 10 | 6 | 14 | 7 | 9 | 0 |
| Hemorrhage | 26 | 8 | 19 | 9 | 24 | 9 |
| Gastrointestinal bleeding | 8 | 6 | 9 | 7 | 9 | 3 |
| CNS bleeding | 1 | 1 | 0 | 0 | 3 | 3 |
| Vomiting | 11 | 1 | 12 | 0 | 15 | 0 |
| Pyrexia | 11 | 2 | 18 | 3 | 6 | 0 |
| Febrile neutropenia | 4 | 4 | 12 | 12 | 12 | 12 |
Adverse reactions associated with bone growth and development were reported in 5 (5.2%) of pediatric patients with chronic phase CML [see Warnings and Precautions (5.10 )].
Laboratory Abnormalities
Myelosuppression was commonly reported in all patient populations. The frequency of Grade 3 or 4 neutropenia, thrombocytopenia and anemia was higher in patients with advanced phase CML than in chronic phase CML (Tables 12 and 13). Myelosuppression was reported in patients with normal baseline laboratory values as well as in patients with pre-existing laboratory abnormalities.
In patients who experienced severe myelosuppression, recovery generally occurred following dose interruption or reduction; permanent discontinuation of treatment occurred in 2% of adult patients with newly diagnosed chronic phase CML and 5% of adult patients with resistance or intolerance to prior imatinib therapy [see Warnings and Precautions (5.1 )] .
Grade 3 or 4 elevations of transaminases or bilirubin and Grade 3 or 4 hypocalcemia, hypokalemia and hypophosphatemia were reported in patients with all phases of CML but were reported with an increased frequency in patients with myeloid or lymphoid blast phase CML. Elevations in transaminases or bilirubin were usually managed with dose reduction or interruption. Patients developing Grade 3 or 4 hypocalcemia during dasatinib therapy often had recovery with oral calcium supplementation.
Laboratory abnormalities reported in adult patients with newly diagnosed chronic phase CML are shown in Table 12. There were no discontinuations of dasatinib therapy in this patient population due to biochemical laboratory parameters.
CTC grades: neutropenia (Grade 3 ≥ 0.5 to < 1 × 10 9 /L, Grade 4 < 0.5 × 10 9 /L); thrombocytopenia (Grade 3 ≥ 25 to < 50 × 10 9 /L, Grade 4 < 25 × 10 9 /L); anemia (hemoglobin Grade 3 ≥ 65 to < 80 g/L, Grade 4 < 65 g/L); elevated creatinine (Grade 3 > 3 to 6 × upper limit of normal range (ULN), Grade 4 > 6 × ULN); elevated bilirubin (Grade 3 > 3 to 10 × ULN, Grade 4 > 10 × ULN); elevated SGOT or SGPT (Grade 3 > 5 to 20 × ULN, Grade 4 > 20 × ULN); hypocalcemia (Grade 3 < 7 mg/dL to 6 mg/dL, Grade 4 < 6 mg/dL); hypophosphatemia (Grade 3 < 2 mg/dL to 1 mg/dL, Grade 4 < 1 mg/dL); hypokalemia (Grade 3 < 3 to 2.5 mmol/L, Grade 4 < 2.5 mmol/L). | ||
| Dasatinib (n=258) | Imatinib (n=258) | |
| Percent (%) of Patients | ||
| Hematology Parameters | ||
| Neutropenia | 29 | 24 |
| Thrombocytopenia | 22 | 14 |
| Anemia | 13 | 9 |
| Biochemistry Parameters | ||
| Hypophosphatemia | 7 | 31 |
| Hypokalemia | 0 | 3 |
| Hypocalcemia | 4 | 3 |
| Elevated SGPT (ALT) | < 1 | 2 |
| Elevated SGOT (AST) | < 1 | 1 |
| Elevated Bilirubin | 1 | 0 |
| Elevated Creatinine | 1 | 1 |
Laboratory abnormalities reported in patients with CML resistant or intolerant to imatinib who received the recommended starting doses of dasatinib are shown by disease phase in Table 13.
CTC grades: neutropenia (Grade 3 ≥ 0.5 to < 1 × 10 9 /L, Grade 4 < 0.5 × 10 9 /L); thrombocytopenia (Grade 3 ≥ 25 to < 50 × 10 9 /L, Grade 4 < 25 × 10 9 /L); anemia (hemoglobin Grade 3 ≥ 65 to < 80 g/L, Grade 4 < 65 g/L); elevated creatinine (Grade 3 > 3 to 6 × upper limit of normal range (ULN), Grade 4 > 6 × ULN); elevated bilirubin (Grade 3 > 3 to 10 × ULN, Grade 4 > 10 × ULN); elevated SGOT or SGPT (Grade 3 > 5 to 20 × ULN, Grade 4 > 20 × ULN); hypocalcemia (Grade 3 < 7 to 6 mg/dL, Grade 4 < 6 mg/dL); hypophosphatemia (Grade 3 < 2 to 1 mg/dL, Grade 4 < 1 mg/dL); hypokalemia (Grade 3 < 3 to 2.5 mmol/L, Grade 4 < 2.5 mmol/L). | ||||
• Hematology parameters for 100 mg once-daily dosing in chronic phase CML reflects 60-month minimum follow-up. | ||||
| Chronic Phase CML 100 mg Once Daily | Advanced Phase CML 140 mg Once Daily | |||
| (n=165) | Accelerated Phase (n=157) | Myeloid Blast Phase (n=74) | Lymphoid Blast Phase (n=33) | |
| Percent (%) of Patients | ||||
| Hematology Parameters • | ||||
| Neutropenia | 36 | 58 | 77 | 79 |
| Thrombocytopenia | 24 | 63 | 78 | 85 |
| Anemia | 13 | 47 | 74 | 52 |
| Biochemistry Parameters | ||||
| Hypophosphatemia | 10 | 13 | 12 | 18 |
| Hypokalemia | 2 | 7 | 11 | 15 |
| Hypocalcemia | < 1 | 4 | 9 | 12 |
| Elevated SGPT (ALT) | 0 | 2 | 5 | 3 |
| Elevated SGOT (AST) | < 1 | 0 | 4 | 3 |
| Elevated Bilirubin | < 1 | 1 | 3 | 6 |
| Elevated Creatinine | 0 | 2 | 8 | 0 |
Among adult patients with chronic phase CML with resistance or intolerance to prior imatinib therapy, cumulative Grade 3 or 4 cytopenias were similar at 2 years and 5 years including: neutropenia (36% vs. 36%), thrombocytopenia (23% vs. 24%) and anemia (13% vs. 13%).
Philadelphia Chromosome-Positive Acute Lymphoblastic Leukemia (Ph+ ALL) in Adults
A total of 135 adult patients with Ph+ ALL were treated with dasatinib in clinical studies. The median duration of treatment was 3 months (range 0.03 to 31 months). The safety profile of patients with Ph+ ALL was similar to those with lymphoid blast phase CML. The most frequently reported adverse reactions included fluid retention events, such as pleural effusion (24%) and superficial edema (19%) and gastrointestinal disorders, such as diarrhea (31%), nausea (24%) and vomiting (16%). Hemorrhage (19%), pyrexia (17%), rash (16%) and dyspnea (16%) were also frequently reported. Serious adverse reactions reported in ≥ 5% of patients included pleural effusion (11%), gastrointestinal bleeding (7%), febrile neutropenia (6%) and infection (5%).
Additional Pooled Data from Clinical Trials
The following additional adverse reactions were reported in adult patients in dasatinib CML clinical studies and adult patients in Ph+ ALL clinical studies at a frequency of ≥ 10%, 1% to < 10%, 0.1% to < 1%, or < 0.1%. These adverse reactions are included based on clinical relevance.
Gastrointestinal Disorders: 1% to < 10% – mucosal inflammation (including mucositis/stomatitis), dyspepsia, abdominal distension, constipation, gastritis, colitis (including neutropenic colitis), oral soft tissue disorder; 0.1% to < 1% – ascites, dysphagia, anal fissure, upper gastrointestinal ulcer, esophagitis, pancreatitis, gastroesophageal reflux disease; < 0.1% – protein losing gastroenteropathy, ileus, acute pancreatitis, anal fistula.
General Disorders and Administration-Site Conditions: ≥ 10% – peripheral edema, face edema; 1% to < 10% – asthenia, chest pain, chills; 0.1% to < 1% – malaise, other superficial edema, peripheral swelling; < 0.1% – gait disturbance.
Skin and Subcutaneous Tissue Disorders: 1% to < 10% – alopecia, acne, dry skin, hyperhidrosis, urticaria, dermatitis (including eczema); 0.1% to < 1% – pigmentation disorder, skin ulcer, bullous conditions, photosensitivity, nail disorder, neutrophilic dermatosis, panniculitis, palmar-plantar erythrodysesthesia syndrome, hair disorder; < 0.1% – leukocytoclastic vasculitis, skin fibrosis.
Respiratory, Thoracic and Mediastinal Disorders: 1% to < 10% – lung infiltration, pneumonitis, cough; 0.1% to < 1% – asthma, bronchospasm, dysphonia, pulmonary arterial hypertension; < 0.1% – acute respiratory distress syndrome, pulmonary embolism.
Nervous System Disorders: 1% to < 10% – neuropathy (including peripheral neuropathy) , dizziness, dysgeusia, somnolence; 0.1% to < 1% – amnesia, tremor, syncope, balance disorder; < 0.1% – convulsion, cerebrovascular accident, transient ischemic attack, optic neuritis, VIIth nerve paralysis, dementia, ataxia.
Blood and Lymphatic System Disorders: 0.1% to < 1% – lymphadenopathy, lymphopenia; < 0.1% – aplasia pure red cell.
Musculoskeletal and Connective Tissue Disorders: 1% to < 10% – muscular weakness, musculoskeletal stiffness; 0.1% to < 1% – rhabdomyolysis, tendonitis, muscle inflammation, osteonecrosis, arthritis; < 0.1% – epiphyses delayed fusion (reported at 1% to < 10% in the pediatric studies), growth retardation (reported at 1% to < 10% in the pediatric studies).
Investigations: 1% to < 10% – weight increased, weight decreased; 0.1% to < 1% – blood creatine phosphokinase increased, gamma-glutamyltransferase increased.
Infections and Infestations: 1% to < 10% – pneumonia (including bacterial, viral and fungal), upper respiratory tract infection/inflammation, herpes virus infection, enterocolitis infection, sepsis (including fatal outcomes [0.2%]).
Metabolism and Nutrition Disorders: 1% to < 10% – appetite disturbances, hyperuricemia; 0.1% to < 1% – hypoalbuminemia, tumor lysis syndrome, dehydration, hypercholesterolemia; < 0.1% – diabetes mellitus.
Cardiac Disorders: 1% to < 10% – arrhythmia (including tachycardia), palpitations; 0.1% to < 1% – angina pectoris, cardiomegaly, pericarditis, ventricular arrhythmia (including ventricular tachycardia), electrocardiogram T-wave abnormal, troponin increased; < 0.1% – cor pulmonale, myocarditis, acute coronary syndrome, cardiac arrest, electrocardiogram PR prolongation, coronary artery disease, pleuropericarditis.
Eye Disorders: 1% to < 10% – visual disorder (including visual disturbance, vision blurred and visual acuity reduced), dry eye; 0.1% to < 1% – conjunctivitis, visual impairment, lacrimation increased, < 0.1% – photophobia.
Vascular Disorders: 1% to < 10% – flushing, hypertension; 0.1% to < 1% – hypotension, thrombophlebitis, thrombosis; < 0.1% – livedo reticularis, deep vein thrombosis, embolism.
Psychiatric Disorders: 1% to < 10% – insomnia, depression; 0.1% to < 1% – anxiety, affect lability, confusional state, libido decreased.
Pregnancy, Puerperium and Perinatal Conditions: < 0.1% – abortion.
Reproductive System and Breast Disorders: 0.1% to < 1% – gynecomastia, menstrual disorder.
Injury, Poisoning and Procedural Complications: 1% to < 10% – contusion.
Ear and Labyrinth Disorders: 1% to < 10% – tinnitus; 0.1% to < 1% – vertigo, hearing loss.
Hepatobiliary Disorders: 0.1% to < 1% – cholestasis, cholecystitis, hepatitis.
Renal and Urinary Disorders: 0.1% to < 1% – urinary frequency, renal failure, proteinuria; < 0.1% – renal impairment.
Immune System Disorders: 0.1% to < 1% – hypersensitivity (including erythema nodosum).
Endocrine Disorders: 0.1% to < 1% – hypothyroidism; < 0.1% – hyperthyroidism, thyroiditis.
Pediatric use information is approved for Bristol-Myers Squibb Company's Sprycel (dasatinib) tablets. However, due to Bristol-Myers Squibb Company's marketing exclusivity rights, this drug product is not labeled with that pediatric information.
Postmarketing Experience
The following additional adverse reactions have been identified during post approval use of dasatinib. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Infections: hepatitis B virus reactivation
Cardiac disorders: atrial fibrillation/atrial flutter
Respiratory, thoracic and mediastinal disorders: interstitial lung disease, chylothorax
Skin and subcutaneous tissue disorders: Stevens-Johnson syndrome
Renal and urinary disorders: nephrotic syndrome
Blood and lymphatic system disorders: thrombotic microangiopathy
Hepatobiliary disorders: hepatotoxicity
Pediatric use information is approved for Bristol-Myers Squibb Company's Sprycel (dasatinib) tablets. However, due to Bristol-Myers Squibb Company's marketing exclusivity rights, this drug product is not labeled with that pediatric information.
DRUG INTERACTIONS
Effect of Other Drugs on Dasatinib
Strong CYP3A4 Inhibitors
The coadministration with strong CYP3A inhibitors may increase dasatinib concentrations [see Clinical Pharmacology (12.3 )] . Increased dasatinib concentrations may increase the risk of toxicity. Avoid concomitant use of strong CYP3A4 inhibitors. If concomitant administration of a strong CYP3A4 inhibitor cannot be avoided, consider a dasatinib dose reduction [see Dosage and Administration (2.5 )] .
Strong CYP3A4 Inducers
The coadministration of dasatinib with strong CYP3A inducers may decrease dasatinib concentrations [see Clinical Pharmacology (12.3 )] . Decreased dasatinib concentrations may reduce efficacy. Consider alternative drugs with less enzyme induction potential. If concomitant administration of a strong CYP3A4 inducer cannot be avoided, consider a dasatinib dose increase.
Gastric Acid Reducing Agents
The coadministration of dasatinib with a gastric acid reducing agent may decrease the concentrations of dasatinib. Decreased dasatinib concentrations may reduce efficacy. Do not administer H 2 antagonists or proton pump inhibitors with dasatinib. Consider the use of antacids in place of H 2 antagonists or proton pump inhibitors. Administer the antacid at least 2 hours prior to or 2 hours after the dose of dasatinib. Avoid simultaneous administration of dasatinib with antacids.
DESCRIPTION
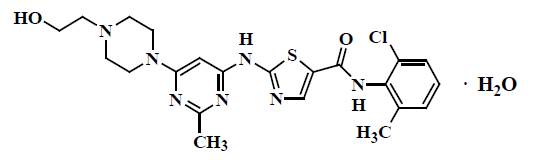
Dasatinib is a kinase inhibitor. The chemical name for dasatinib is N-(2-chloro-6-methylphenyl)-2-[[6-[4-(2-hydroxyethyl)-1-piperazinyl]-2-methyl-4-pyrimidinyl]amino]-5-thiazolecarboxamide, monohydrate. The molecular formula is C 22 H 26 ClN 7 O 2 S • H 2 O, which corresponds to a molecular weight of 506.02 g/mol (monohydrate). The anhydrous free base has a molecular weight of 488.01 g/mol.
Dasatinib has the following chemical structure:
Dasatinib is a white to off-white powder. The drug substance is insoluble in water and slightly soluble in ethanol and methanol.
Each dasatinib tablet intended for oral administration contains 20 mg, 50 mg, 70 mg, 80 mg, 100 mg or 140 mg of dasatinib. In addition, each tablet contains the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, glyceryl monocaprylocaprate type 1, hydroxypropyl cellulose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyvinyl alcohol-partially hydrolyzed, sodium lauryl sulfate, talc and titanium dioxide.
CLINICAL PHARMACOLOGY
Mechanism of Action
Dasatinib, at nanomolar concentrations, inhibits the following kinases: BCR-ABL, SRC family (SRC, LCK, YES, FYN), c-KIT, EPHA2 and PDGFRβ. Based on modeling studies, dasatinib is predicted to bind to multiple conformations of the ABL kinase.
In vitro , dasatinib was active in leukemic cell lines representing variants of imatinib mesylate-sensitive and resistant disease. Dasatinib inhibited the growth of chronic myeloid leukemia (CML) and acute lymphoblastic leukemia (ALL) cell lines overexpressing BCR-ABL. Under the conditions of the assays, dasatinib could overcome imatinib resistance resulting from BCR-ABL kinase domain mutations, activation of alternate signaling pathways involving the SRC family kinases (LYN, HCK) and multi-drug resistance gene overexpression.
Pharmacodynamics
Cardiac Electrophysiology
Of 2,440 patients treated with dasatinib at all doses tested in clinical trials, 16 patients (< 1%) had QTc prolongation reported as an adverse reaction. Twenty-two patients (1%) experienced a QTcF > 500 ms. In 865 patients with leukemia treated with dasatinib 70 mg BID in five Phase 2 studies, the maximum mean changes in QTcF (90% upper bound CI) from baseline ranged from 7 ms to 13.4 ms.
An analysis of the data from five Phase 2 studies in patients (70 mg BID) and a Phase 1 study in healthy subjects (100 mg single dose) suggests that there is a maximum increase of 3 milliseconds to 6 milliseconds in Fridericia corrected QTc interval from baseline for subjects receiving therapeutic doses of dasatinib, with associated upper 95% confidence intervals < 10 msec.
Pharmacokinetics
The pharmacokinetics of dasatinib exhibits dose proportional increases in AUC and linear elimination characteristics over the dose range of 15 mg/day (0.15 times the lowest approved recommended dose) to 240 mg/day (1.7 times the highest approved recommended dose).
At 100 mg QD, the maximum concentration at steady state (C max ) is 82.2 ng/mL (CV% 69%), area under the plasma drug concentration time curve (AUC) is 397 ng/mL • hr (CV% 55%). The clearance of dasatinib is found to be time-invariant. When administered to adult healthy subjects as dispersed tablets in juice, the adjusted geometric mean ratio was 0.97 (90% CI: 0.85, 1.10) for C max and 0.84 (90% CI: 0.78, 0.91) for AUC as compared to intact tablets.
Absorption
The maximum plasma concentrations (C max ) of dasatinib are observed between 0.5 hours and 6 hours (T max ) following oral administration.
Food Effect
A high-fat meal increased the mean AUC of dasatinib following a single dose of 100 mg by 14%. The total calorie content of the high-fat meal was 985 kcal. The calories derived from fat, carbohydrates and protein were 52%, 34% and 14% for the high-fat meal.
Distribution
The apparent volume of distribution is 2,505 L (CV% 93%).
Binding of dasatinib to human plasma proteins in vitro was approximately 96% and of its active metabolite was 93%, with no concentration dependence over the range of 100 ng/mL to 500 ng/mL.
Dasatinib is a P-gp substrate in vitro .
Elimination
The mean terminal half-life of dasatinib is 3 hours to 5 hours. The mean apparent oral clearance is 363.8 L/hr (CV% 81.3%).
Metabolism
Dasatinib is metabolized in humans, primarily by CYP3A4. CYP3A4 is the primary enzyme responsible for the formation of the active metabolite. Flavin-containing monooxygenase 3 (FMO-3) and uridine diphosphate-glucuronosyltransferase (UGT) enzymes are also involved in the formation of dasatinib metabolites.
The exposure of the active metabolite, which is equipotent to dasatinib, represents approximately 5% of the AUC of dasatinib. The active metabolite of dasatinib is unlikely to play a major role in the observed pharmacology of the drug. Dasatinib also has several other inactive oxidative metabolites.
Excretion
Elimination is primarily via the feces. Following a single radiolabeled dose of oral dasatinib, 4% of the administered radioactivity was recovered in the urine and 85% in the feces within 10 days. Unchanged dasatinib accounted for 0.1% of the administered dose in the urine and 19% of the administered dose in the feces with the remainder of the dose being metabolites.
Specific Populations
Age (15 to 86 years old), sex, and renal impairment (creatinine clearance 21.6 mL/min to 342.3 mL/min as estimated by Cockcroft Gault) have no clinically relevant effect on the pharmacokinetics of dasatinib.
Pediatric Patients
Dasatinib clearance and volume of distribution change with body weight in pediatric patients. Dasatinib has not been studied in patients < 1 year old.
Patients with Hepatic Impairment
Compared to subjects with normal liver function, patients with moderate hepatic impairment (Child Pugh B) had decreases in mean C max by 47% and mean AUC by 8%. Patients with severe hepatic impairment (Child Pugh C) had decreases in mean C max by 43% and in mean AUC by 28% compared to the subjects with normal liver function.
Drug Interaction Studies
Cytochrome P450 Enzymes
The coadministration of ketoconazole (strong CYP3A4 inhibitor) twice daily increased the mean C max of dasatinib by 4-fold and the mean AUC of dasatinib by 5-fold following a single oral dose of 20 mg.
The coadministration of rifampin (strong CYP3A4 inducer) once daily decreased the mean C max of dasatinib by 81% and the mean AUC of dasatinib by 82%.
Dasatinib is a time-dependent inhibitor of CYP3A4. Dasatinib does not inhibit CYP1A2, 2A6, 2B6, 2C8, 2C9, 2C19, 2D6 or 2E1. Dasatinib does not induce CYP enzymes.
Gastric Acid Reducing Agents
The administration of 30 mL of aluminum hydroxide/magnesium hydroxide 2 hours prior to a single dose of dasatinib was associated with no relevant change in the mean AUC of dasatinib; however, the mean C max of dasatinib was increased by 26%.
The simultaneous administration of 30 mL of aluminum hydroxide/magnesium hydroxide with a single dose of dasatinib was associated with a 55% reduction in the mean AUC of dasatinib and a 58% reduction in the mean C max of dasatinib.
The administration of a single dose of dasatinib 10 hours following famotidine (H 2 antagonist) reduced the mean AUC of dasatinib by 61% and the mean C max of dasatinib by 63%.
The administration of a single 100 mg dose of dasatinib 22 hours following a 40 mg dose of omeprazole (proton pump inhibitor) at steady state reduced the mean AUC of dasatinib by 43% and the mean C max of dasatinib by 42%.
Transporters
Dasatinib is not an inhibitor of P-gp in vitro .
Pediatric use information is approved for Bristol-Myers Squibb Company's Sprycel (dasatinib) tablets. However, due to Bristol-Myers Squibb Company's marketing exclusivity rights, this drug product is not labeled with that pediatric information.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2-year carcinogenicity study, rats were administered oral doses of dasatinib at 0.3 mg/kg/day, 1 mg/kg/day and 3 mg/kg/day. The highest dose resulted in a plasma drug exposure (AUC) level approximately 60% of the human exposure at 100 mg once daily. Dasatinib induced a statistically significant increase in the combined incidence of squamous cell carcinomas and papillomas in the uterus and cervix of high-dose females and prostate adenoma in low-dose males.
Dasatinib was clastogenic when tested in vitro in Chinese hamster ovary cells, with and without metabolic activation. Dasatinib was not mutagenic when tested in an in vitro bacterial cell assay (Ames test) and was not genotoxic in an in vivo rat micronucleus study.
Dasatinib did not affect mating or fertility in male and female rats at plasma drug exposure (AUC) similar to the human exposure at 100 mg daily. In repeat dose studies, administration of dasatinib resulted in reduced size and secretion of seminal vesicles and immature prostate, seminal vesicle and testis. The administration of dasatinib resulted in uterine inflammation and mineralization in monkeys and cystic ovaries and ovarian hypertrophy in rodents.
CLINICAL STUDIES
Newly Diagnosed Chronic Phase CML in Adults
DASISION (Dasatinib vs Imatinib Study in Treatment-Naive Chronic Myeloid Leukemia Patients) (NCT00481247) was an open-label, multicenter, international, randomized trial conducted in adult patients with newly diagnosed chronic phase CML. A total of 519 patients were randomized to receive either dasatinib 100 mg once daily or imatinib 400 mg once daily. Patients with a history of cardiac disease were included in this trial except those who had a myocardial infarction within 6 months, congestive heart failure within 3 months, significant arrhythmias or QTc prolongation. The primary endpoint was the rate of confirmed complete cytogenetic response (CCyR) within 12 months. Confirmed CCyR was defined as a CCyR noted on two consecutive occasions (at least 28 days apart).
Median age was 46 years in the dasatinib group and 49 years in the imatinib groups, with 10% and 11% of patients ≥ 65 years of age, respectively. There were slightly more male than female patients in both groups (59% vs. 41%). Fifty-three percent of all patients were Caucasian and 39% were Asian. At baseline, the distribution of Hasford scores was similar in the dasatinib and imatinib treatment groups (low risk: 33% and 34%; intermediate risk: 48% and 47%; high risk: 19% and 19%, respectively). With a minimum of 12 months follow-up, 85% of patients randomized to dasatinib and 81% of patients randomized to imatinib were still on study.
With a minimum of 24 months follow-up, 77% of patients randomized to dasatinib and 75% of patients randomized to imatinib were still on study and with a minimum of 60 months follow-up, 61% and 62% of patients, respectively, were still on treatment at the time of study closure.
Efficacy results are summarized in Table 16.
a Confirmed CCyR is defined as a CCyR noted on two consecutive occasions at least 28 days apart. | ||
b Major molecular response (at any time) was defined as BCR-ABL ratios ≤ 0.1% by RQ-PCR in peripheral blood samples standardized on the International scale. These are cumulative rates representing minimum follow up for the time frame specified. | ||
• Adjusted for Hasford score and indicated statistical significance at a pre-defined nominal level of significance. | ||
CI = confidence interval. | ||
| Dasatinib (n=259) | Imatinib (n=260) | |
| Confirmed CCyR a | ||
| Within 12 months (95% CI) | 76.8% (71.2 to 81.8) | 66.2% (60.1 to 71.9) |
| P-value | 0.007 • | |
| Major Molecular Response b | ||
| 12 months (95% CI) | 52.1% (45.9 to 58.3) | 33.8% (28.1 to 39.9) |
| P-value | <0.0001 | |
| 60 months (95% CI) | 76.4% (70.8 to 81.5) | 64.2% (58.1 to 70.1) |
The confirmed CCyR within 24 months, 36 months and 60 months for dasatinib versus imatinib arms were 80% versus 74%, 83% versus 77%, and 83% versus 79%, respectively. The MMR at 24 months and 36 months for dasatinib versus imatinib arms were 65% versus 50% and 69% versus 56%, respectively.
After 60 months follow-up, median time to confirmed CCyR was 3.1 months in 215 dasatinib responders and 5.8 months in 204 imatinib responders. Median time to MMR after 60 months follow-up was 9.3 months in 198 dasatinib responders and 15 months in 167 imatinib responders.
At 60 months, 8 patients (3%) on the dasatinib arm progressed to either accelerated phase or blast crisis while 15 patients (6%) on the imatinib arm progressed to either accelerated phase or blast crisis.
The estimated 60-month survival rates for dasatinib- and imatinib-treated patients were 90.9% (CI: 86.6% to 93.8%) and 89.6% (CI: 85.2% to 92.8%), respectively. Based on data 5 years after the last patient was enrolled in the trial, 83% and 77% of patients were known to be alive in the dasatinib and imatinib treatment groups, respectively, 10% were known to have died in both treatment groups and 7% and 13% had unknown survival status in the dasatinib and imatinib treatment groups, respectively.
At 60 months follow-up in the dasatinib arm, the rate of MMR at any time in each risk group determined by Hasford score was 90% (low risk), 71% (intermediate risk) and 67% (high risk). In the imatinib arm, the rate of MMR at any time in each risk group determined by Hasford score was 69% (low risk), 65% (intermediate risk) and 54% (high risk).
BCR-ABL sequencing was performed on blood samples from patients in the newly diagnosed trial who discontinued dasatinib or imatinib therapy. Among dasatinib-treated patients the mutations detected were T315I, F317I/L and V299L.
Dasatinib does not appear to be active against the T315I mutation, based on in vitro data.
Imatinib-Resistant or -Intolerant CML or Ph+ ALL in Adults
The efficacy and safety of dasatinib were investigated in adult patients with CML or Ph+ ALL whose disease was resistant to or who were intolerant to imatinib: 1,158 patients had chronic phase CML, 858 patients had accelerated phase, myeloid blast phase or lymphoid blast phase CML and 130 patients had Ph+ ALL. In a clinical trial in chronic phase CML, resistance to imatinib was defined as failure to achieve a complete hematologic response (CHR; after 3 months), major cytogenetic response (MCyR; after 6 months) or complete cytogenetic response (CCyR; after 12 months) or loss of a previous molecular response (with concurrent ≥ 10% increase in Ph+ metaphases), cytogenetic response or hematologic response. Imatinib intolerance was defined as inability to tolerate 400 mg or more of imatinib per day or discontinuation of imatinib because of toxicity.
Results described below are based on a minimum of 2 years follow-up after the start of dasatinib therapy in patients with a median time from initial diagnosis of approximately 5 years. Across all studies, 48% of patients were women, 81% were white, 15% were black or Asian, 25% were 65 years of age or older and 5% were 75 years of age or older. Most patients had long disease histories with extensive prior treatment, including imatinib, cytotoxic chemotherapy, interferon and stem cell transplant. Overall, 80% of patients had imatinib-resistant disease and 20% of patients were intolerant to imatinib. The maximum imatinib dose had been 400 mg/day to 600 mg/day in about 60% of the patients and > 600 mg/day in 40% of the patients.
The primary efficacy endpoint in chronic phase CML was MCyR, defined as elimination (CCyR) or substantial diminution (by at least 65%, partial cytogenetic response) of Ph+ hematopoietic cells. The primary efficacy endpoint in accelerated phase, myeloid blast phase, lymphoid blast phase CML and Ph+ ALL was major hematologic response (MaHR), defined as either a CHR or no evidence of leukemia (NEL).
Chronic Phase CML
Dose-Optimization Trial: A randomized, open-label trial (NCT00123474) was conducted in adult patients with chronic phase CML to evaluate the efficacy and safety of dasatinib administered once daily compared with dasatinib administered twice daily. Patients with significant cardiac diseases, including myocardial infarction within 6 months, congestive heart failure within 3 months, significant arrhythmias or QTc prolongation were excluded from the trial. The primary efficacy endpoint was MCyR in patients with imatinib-resistant CML. A total of 670 patients, of whom 497 had imatinib-resistant disease, were randomized to the dasatinib 100 mg once-daily, 140 mg once-daily, 50 mg twice-daily or 70 mg twice-daily group. Median duration of treatment was 22 months.
Efficacy was achieved across all dasatinib treatment groups with the once-daily schedule demonstrating comparable efficacy (non-inferiority) to the twice-daily schedule on the primary efficacy endpoint (difference in MCyR 1.9%; 95% CI [−6.8% to 10.6%]); however, the 100-mg once-daily regimen demonstrated improved safety and tolerability.
Efficacy results are presented in Tables 17 and 18 for adult patients with chronic phase CML who received the recommended starting dose of 100 mg once daily.
a CHR (response confirmed after 4 weeks): WBC ≤ institutional ULN, platelets < 450,000/mm 3 , no blasts or promyelocytes in peripheral blood, < 5% myelocytes plus metamyelocytes in peripheral blood, basophils in peripheral blood < 20%, and no extramedullary involvement. | |
b MCyR combines both complete (0% Ph+ metaphases) and partial (> 0% to 35%) responses. | |
| All Patients | 100 mg Once Daily (n=167) |
| Hematologic Response Rate % (95% CI) | |
| CHR a | 92% (86 to 95) |
| Cytogenetic Response Rate % (95% CI) | |
| MCyR b | 63% (56 to 71) |
| CCyR | 50% (42 to 58) |
a Results reported in recommended starting dose of 100 mg once daily. | |||
b Major molecular response criteria: Defined as BCR-ABL/control transcripts ≤ 0.1% by RQ-PCR in peripheral blood samples. | |||
| Minimum Follow-up Period | |||
| 2 Years | 5 Years | 7 Years | |
| Major Molecular Response b % (n/N) | |||
| All Patients Randomized | 34% (57/167) | 43% (71/167) | 44% (73/167) |
| Imatinib-Resistant Patients | 33% (41/124) | 40% (50/124) | 41% (51/124) |
| Imatinib-Intolerant Patients | 37% (16/43) | 49% (21/43) | 51% (22/43) |
Based on data 7 years after the last patient was enrolled in the trial, 44% were known to be alive, 31% were known to have died and 25% had an unknown survival status.
By 7 years, transformation to either accelerated or blast phase occurred in nine patients on treatment in the 100 mg once-daily treatment group.
Advanced Phase CML and Ph+ ALL
Dose-Optimization Trial: One randomized open-label trial (NCT00123487) was conducted in patients with advanced phase CML (accelerated phase CML, myeloid blast phase CML or lymphoid blast phase CML) to evaluate the efficacy and safety of dasatinib administered once daily compared with dasatinib administered twice daily. The primary efficacy endpoint was MaHR. A total of 611 patients were randomized to either the dasatinib 140 mg once-daily or 70 mg twice-daily group. Median duration of treatment was approximately 6 months for both treatment groups. The once-daily schedule demonstrated comparable efficacy (non-inferiority) to the twice-daily schedule on the primary efficacy endpoint; however, the 140-mg once-daily regimen demonstrated improved safety and tolerability.
Response rates for patients in the 140 mg once-daily group are presented in Table 19.
a Hematologic response criteria (all responses confirmed after 4 weeks): Major hematologic response: (MaHR) = complete hematologic response (CHR) + no evidence of leukemia (NEL). CHR: WBC ≤ institutional ULN, ANC ≥ 1,000/mm 3 , platelets ≥ 100,000/mm 3 , no blasts or promyelocytes in peripheral blood, bone marrow blasts ≤ 5%, < 5% myelocytes plus metamyelocytes in peripheral blood, basophils in peripheral blood < 20% and no extramedullary involvement. | ||||
NEL: same criteria as for CHR but ANC ≥ 500/mm 3 and < 1000/mm 3 or platelets ≥ 20,000/mm 3 and ≤ 100,000/mm 3 . | ||||
b MCyR combines both complete (0% Ph+ metaphases) and partial (> 0% to 35%) responses. | ||||
CI = confidence interval ULN = upper limit of normal range. | ||||
| 140 mg Once Daily | ||||
| Accelerated (n=158) | Myeloid Blast (n=75) | Lymphoid Blast (n=33) | Ph+ ALL (n=40) | |
| MaHR a | 66% | 28% | 42% | 38% |
| (95% CI) | (59 to 74) | (18 to 40) | (26 to 61) | (23 to 54) |
| CHR a | 47% | 17% | 21% | 33% |
| (95% CI) | (40 to 56) | (10 to 28) | (9 to 39) | (19 to 49) |
| NEL a | 19% | 11% | 21% | 5% |
| (95% CI) | (13 to 26) | (5 to 20) | (9 to 39) | (1 to 17) |
| MCyR b | 39% | 28% | 52% | 70% |
| (95% CI) | (31 to 47) | (18 to 40) | (34 to 69) | (54 to 83) |
| CCyR | 32% | 17% | 39% | 50% |
| (95% CI) | (25 to 40) | (10 to 28) | (23 to 58) | (34 to 66) |
In the dasatinib 140 mg once-daily group, the median time to MaHR was 1.9 months (min-max: 0.7 to 14.5) for patients with accelerated phase CML, 1.9 months (min-max: 0.9 to 6.2) for patients with myeloid blast phase CML and 1.8 months (min-max: 0.9 to 2.8) for patients with lymphoid blast phase CML.
In patients with myeloid blast phase CML, the median duration of MaHR was 8.1 months (min-max: 2.7 to 21.1) and 9.0 (min-max: 1.8 to 23.1) months for the 140 mg once-daily group and the 70 mg twice-daily group, respectively. In patients with lymphoid blast phase CML, the median duration of MaHR was 4.7 months (min-max: 3 to 9) and 7.9 months (min-max: 1.6 to 22.1) for the 140 mg once-daily group and the 70 mg twice-daily group, respectively. In patients with Ph+ ALL who were treated with dasatinib 140 mg once-daily, the median duration of MaHR was 4.6 months (min-max: 1.4 to 10.2). The medians of progression-free survival for patients with Ph+ ALL treated with dasatinib 140 mg once-daily and 70 mg twice-daily were 4.0 months (min-max: 0.4 to 11.1) and 3.1 months (min-max: 0.3 to 20.8), respectively.
Pediatric use information is approved for Bristol-Myers Squibb Company's Sprycel (dasatinib) tablets. However, due to Bristol-Myers Squibb Company's marketing exclusivity rights, this drug product is not labeled with that pediatric information.
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Dasatinib Tablets, 20 mg are white to off-white, round, film-coated tablets, debossed with '1741' on one side and plain on the other side and are supplied as follows:
NDC 70710-1741-6 in bottle of 60 tablets with child-resistant closure
Dasatinib Tablets, 50 mg are white to off-white, oval, film-coated tablets, debossed with '1742' on one side and plain on the other side and are supplied as follows:
NDC 70710-1742-6 in bottle of 60 tablets with child-resistant closure
Dasatinib Tablets, 70 mg are white to off-white, round, film-coated tablets, debossed with '1743' on one side and plain on the other side and are supplied as follows:
NDC 70710-1743-6 in bottle of 60 tablets with child-resistant closure
Dasatinib Tablets, 80 mg are white to off-white, oblong, film-coated tablets, debossed with '1744' on one side and plain on the other side and are supplied as follows:
NDC 70710-1744-3 in bottle of 30 tablets with child-resistant closure
Dasatinib Tablets, 100 mg are white to off-white, oval, film-coated tablets, debossed with '1745' on one side and plain on the other side and are supplied as follows:
NDC 70710-1745-3 in bottle of 30 tablets with child-resistant closure
Dasatinib Tablets, 140 mg are white to off-white, round, film-coated tablets, debossed with '1746' on one side and plain on the other side and are supplied as follows:
NDC 70710-1746-3 in bottle of 30 tablets with child-resistant closure
Storage
Dasatinib tablets should be stored at 20°C to 25°C (68ºF to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Handling and Disposal
Dasatinib is an antineoplastic product. Follow special handling and disposal procedures. 1
Personnel who are pregnant should avoid exposure to crushed or broken tablets.
Dasatinib tablets consist of a core tablet, surrounded by a film coating to prevent exposure of healthcare professionals to the active substance. The use of latex or nitrile gloves for appropriate disposal when handling tablets that are inadvertently crushed or broken is recommended, to minimize the risk of dermal exposure.
Mechanism of Action
Dasatinib, at nanomolar concentrations, inhibits the following kinases: BCR-ABL, SRC family (SRC, LCK, YES, FYN), c-KIT, EPHA2 and PDGFRβ. Based on modeling studies, dasatinib is predicted to bind to multiple conformations of the ABL kinase.
In vitro , dasatinib was active in leukemic cell lines representing variants of imatinib mesylate-sensitive and resistant disease. Dasatinib inhibited the growth of chronic myeloid leukemia (CML) and acute lymphoblastic leukemia (ALL) cell lines overexpressing BCR-ABL. Under the conditions of the assays, dasatinib could overcome imatinib resistance resulting from BCR-ABL kinase domain mutations, activation of alternate signaling pathways involving the SRC family kinases (LYN, HCK) and multi-drug resistance gene overexpression.